Abstract
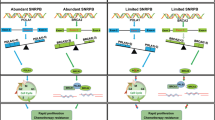
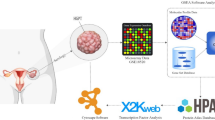
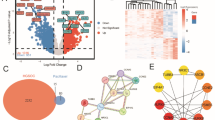
Apoptosis of ovarian granular cells is closely related with weakening fertility of women. Hence, resisting apoptosis of human ovarian granular cells (KGN cells) is of important significance. According to studies, detection of oxidation indicators such as ROS, NO, MDA and SOD, DAPI fluorescence staining experiment, mRNA and Western Blot test of Cleaved-caspase-3, Caspase-3, Bax and Bcl-2 demonstrate that small peptides from Periplaneta americana (SPPA) can improve H2O2-induced apoptosis of KGN cells. While SPPA has been reported to possess antioxidant potential, its specific role and molecular mechanisms within ovarian granulosa cells-particularly regarding its involvement in the miRNA regulatory network have not yet been thoroughly investigated. Therefore, high-throughput sequencing of miRNAs was employed to identify differentially expressed miRNAs (DEMs) that are associated with the regulatory mechanisms through which SPPA inhibits H2O2-induced apoptosis in KGN cells. Experiments were divided into three groups, namely, the control group, H2O2 group and H2O2 + SPPA group. A total of 75 DEMs were screened by comparing the control group and the H2O2 group, and 30 DEMs were screened by comparing the H2O2 group and H2O2 + SPPA group. It is important to note that 8 overlapping DEMs were identified upregulating in H2O2 exposure, but downregulating in SPPA repair. Another 5 overlapping DEMs were identified downregulating in H2O2 exposure, but upregulating in SPPA repair. 3534 target genes of the aforementioned 13 DEMs have significant enrichment in multiple KEGG pathways. Among them, hsa-miR-103a-3p, NovelmiRNA-214 and NovelmiRNA-383 play significant roles in SPPA resistance process of cell apoptosis. 5 DEMs were selected for florescence quantitative PCR (qPCR) verification and the expression level was consistent with sequencing results. Finally, a control network of SPPA resistance against the H2O2-induced KGN cell apoptosis was built based on the target genes screened by the miRNA-seq technology. This study provides a direction and some references to screen and suggest miRNAs that may be involved in the anti-apoptotic process of SPPA.
Similar content being viewed by others
Data availability
The miRNA-seq raw data is deposited on the Sequence Read Archive (SRA) database at https://www.ncbi.nlm.nih.gov/bioproject, BioProject: PRJNA837735.
References
Golezar, S., Ramezani, T. F., Khazaei, S., Ebadi, A. & Keshavarz, Z. The global prevalence of primary ovarian insufficiency and early menopause: a meta-analysis. Climacteric 22(4), 403–411 (2019).
Liang, Q. X. et al. Ablation of beta subunit of protein kinase ck2 in mouse oocytes causes follicle atresia and premature ovarian failure. Cell. Death Dis. 9(5), 508 (2018).
Hussein, M. R. Apoptosis in the ovary: molecular mechanisms. Hum. Reprod. Update. 11(2), 162–177 (2005).
Tiwari, M. et al. Apoptosis in mammalian oocytes: a review. Apoptosis 20(8), 1019–1025 (2015).
Agarwal, A., Gupta, S. & Sharma, R. K. Role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 3, 28 (2005).
Zhang, H. et al. Periplaneta americana extract used in patients with systemic inflammatory response syndrome. World J. Emerg. Med. 7(1), 50–54 (2016).
Zhao, B. et al. Periplaneta americana extract promotes hard palate mucosal wound healing via the pi3k/akt signaling pathway in male mice. Arch. Oral Biol. 158, 105856 (2024).
Kong, C. et al. Periplaneta americana peptide decreases apoptosis of pig-ovary granulosa cells induced by h(2) o(2) through foxo1. Reprod. Domest. Anim. 56(11), 1413–1424 (2021).
Fu, R. et al. Small peptides from periplaneta americana inhibits oxidative stress-induced kgn cell apoptosis by regulating mitochondrial function through bcl2l13. Reprod. Sci. 30(2), 473–486 (2023).
Wang, Q. et al. The protective effect of small peptides from periplaneta americana on hydrogen peroxide-induced apoptosis of granular cells. Vitro Cell. Dev. Biol. -Anim. 57(6), 610–619 (2021).
Bofill-De, R. X. et al. Structural differences between pri-mirna paralogs promote alternative drosha cleavage and expand target repertoires. Cell. Rep. 26(2), 447–459 (2019).
Christenson, L. K. Microrna control of ovarian function. Anim. Reprod. 7(3), 129–133 (2010).
Zhang, J. et al. Downregulation of mir-192 alleviates oxidative stress-induced porcine granulosa cell injury by directly targeting acvr2a. Cells 11(15) (2022).
He, H. et al. Mirna sequencing analysis of healthy and atretic follicles of chickens revealed that mir-30a-5p inhibits granulosa cell death via targeting beclin1. J. Anim. Sci. Biotechnol. 13(1), 55 (2022).
Liu, L. et al. Chi-mir-130b-3p regulates the zea-induced oxidative stress damage through the keap1/nrf2 signaling pathway by targeting sesn2 in goat gcs. FASEB J. 37(11), e23212 (2023).
Sun, Z. et al. Cap-mirseq: a comprehensive analysis pipeline for microrna sequencing data. BMC Genom. 15(1), 423 (2014).
Kume, S. et al. Silent information regulator 2 (sirt1) attenuates oxidative stress-induced mesangial cell apoptosis via p53 deacetylation. Free Radic Biol. Med. 40(12), 2175–2182 (2006).
Li, L. C. et al. Autophagy induced by hydrogen peroxide in porcine ovarian granulosa cells and its effect on apoptosis. J. Nanjing Agricultural Univ. 39(05), 814–818 (2016).
Xiang, L., Yao, G., Li, R. X., Zhang, J. & Huang, Y. Z. Research progress on the establishment methods of animal models of premature ovarian failure. Chin. J. Pharm. 50(05), 386–389 (2015).
Massin, N. et al. Evaluation of different markers of the ovarian reserve in patients presenting with premature ovarian failure. Mol. Cell. Endocrinol. 282(1–2), 95–100 (2008).
Weng, Q. et al. Oxidative stress induces mouse follicular granulosa cells apoptosis via jnk/foxo1 pathway. PLoS One. 11(12), e167869 (2016).
Liu, Z. Q. et al. Expression of puma in follicular granulosa cells regulated by foxo1 activation during oxidative stress. Reprod. Sci. 22(6), 696–705 (2015).
Shen, M. et al. Involvement of the up-regulated foxo1 expression in follicular granulosa cell apoptosis induced by oxidative stress. J. Biol. Chem. 287(31), 25727–25740 (2012).
Zhang, M. et al. Mir-181a increases foxo1 acetylation and promotes granulosa cell apoptosis via sirt1 downregulation. Cell. Death Dis. 8(10), e3088 (2017).
Engedal, N. et al. From oxidative stress damage to pathways, networks, and autophagy via micrornas. Oxid. Med. Cell. Longev. 2018, 4968321 (2018).
Lewinska, A. et al. Reduced levels of methyltransferase dnmt2 sensitize human fibroblasts to oxidative stress and dna damage that is accompanied by changes in proliferation-related mirna expression. Redox Biol. 14, 20–34 (2018).
Morita, Y. et al. Resveratrol promotes expression of sirt1 and star in rat ovarian granulosa cells: an implicative role of sirt1 in the ovary. Reprod. Biol. Endocrinol. 10, 14 (2012).
Sohel, M. M. H. et al. Oxidative stress modulates the expression of apoptosis-associated micrornas in bovine granulosa cells in vitro. Cell. Tissue Res. 376(2), 295–308 (2019).
Liu, S. Y. Mechanism study of Periplaneta Americana regulating mir-29a/pten/pi3k to intervene in Ulcerative Colitis in mice. Guangzhou Univ. Chin. Medicine (2023).
Penna, E., Orso, F. & Taverna, D. Mir-214 as a key hub that controls cancer networks: small player, multiple functions. J. Invest. Dermatol. 135(4), 960–969 (2015).
Yang, S. et al. Mirna-214 suppresses oxidative stress in diabetic nephropathy via the ros/akt/mtor signaling pathway and uncoupling protein 2. Exp. Ther. Med. 17(5), 3530–3538 (2019).
Chen, W. et al. Microrna-214 protects l6 skeletal myoblasts against hydrogen peroxide-induced apoptosis. Free Radic Res. 54(2–3), 162–172 (2020).
Wang, F., Liu, M., Li, X. & Tang, H. Mir-214 reduces cell survival and enhances cisplatin-induced cytotoxicity via down-regulation of bcl2l2 in cervical cancer cells. FEBS Lett. 587(5), 488–495 (2013).
Yang, L., Zhang, L., Lu, L. & Wang, Y. Mir-214-3p regulates multi-drug resistance and apoptosis in retinoblastoma cells by targeting abcb1 and xiap. OncoTargets Ther. 13, 803–811 (2020).
Zhang, J., Su, B., Gong, C., Xi, Q. & Chao, T. Mir-214 promotes apoptosis and sensitizes breast cancer cells to doxorubicin by targeting the rfwd2-p53 cascade. Biochem. Biophys. Res. Commun. 478(1), 337–342 (2016).
Huang, Q. et al. Effects of miR-103a-3p on proliferation and apoptosis of ovarian granulosa cells. J. Xiangnan Univ. (Medical Edition). 24(04), 1–6 (2022).
Zhou, Y. P. & Xia, Q. Inhibition of mir-103a-3p suppresses lipopolysaccharide-induced sepsis and liver injury by regulating fbxw7 expression. Cell. Biol. Int. 44(9), 1798–1810 (2020).
Zhang, Q. et al. Roles of mir-10a-5p and mir-103a-3p, regulators of bdnf expression in follicular fluid, in the outcomes of ivf-et. Front. Endocrinol. 12, 637384 (2021).
Cheng, M. & Wang, Y. Downregulation of hmgb1 by mir-103a-3p promotes cell proliferation, alleviates apoptosis and in flammation in a cell model of osteoarthritis. Iran. J. Biotechnol. 18(1), e2255 (2020).
Benco, A. et al. Involvement of the transcription factor stat1 in the regulation of porcine ovarian granulosa cell functions treated and not treated with ghrelin. Reproduction 138(3), 553–560 (2009).
Jafarzadeh, A. et al. Microrna-383: a tumor suppressor mirna in human cancer. Front. Cell. Dev. Biol. 10, 955486 (2022).
Gu, B., Wang, J., Song, Y., Wang, Q. & Wu, Q. Microrna-383 regulates cell viability and apoptosis by mediating wnt/beta-catenin signaling pathway in non-small cell lung cancer. J. Cell. Biochem. 120(5), 7918–7926 (2019).
Li, Z. M. & Li, W. Mir-383 suppresses proliferation of mouse follicular granulosa cells by downregulating cyclin-related proteins. J. Cell. Mol. Immunol. 35(06), 518–525 (2019).
Liu, Y., Wang, Z., Tang, Z., Fu, Y. & Wang, L. Mirna-383-5p functions as an anti-oncogene in glioma through the akt/mtor signaling pathway by targeting vegfa. Curr. Cancer Drug Targets. 24(4), 463–475 (2024).
Li, Y. Research on the mechanism of mir-383-5p regulating apoptosis in ovarian granulosa cells. Hebei Med. University, (2022).
Zhang, H. B. et al. Identified variably spliced genes in resveratrol-induced apoptosis of porcine ovarian granulosa cells through RNA-seq screening. China Anim. Husb. Magazine. 58(08), 36–44 (2022).
Zhang, C. et al. Mirnas expression profiling of rat ovaries displaying pcos with insulin resistance. Arch. Gynecol. Obstet. 302(5), 1205–1213 (2020).
Lin, J. et al. Cytochrome p450 family proteins as potential biomarkers for ovarian granulosa cell damage in mice with premature ovarian failure. Int. J. Clin. Exp. Pathol. 11(8), 4236–4246 (2018).
Tosca, L., Chabrolle, C. & Dupont, J. [Ampk: a link between metabolism and reproduction?]. M S-Med Sci. 24(3), 297–300 (2008).
Brunk, U. T., Neuzil, J. & Eaton, J. W. Lysosomal involvement in apoptosis. Redox Rep. 6(2), 91–97 (2001).
Guicciardi, M. E., Leist, M. & Gores, G. J. Lysosomes in cell death. Oncogene 23(16), 2881–2890 (2004).
Acknowledgements
We would like to thank Suzhou Jinweizhi Biotechnology Co., Ltd for miRNA-seq data analysis. Additionally, thanks to figdraw (https://www.figdraw.com/static/index.html#/) for providing the drawing platform and everyone who was involved in this study.
Funding
This work was supported by National Natural Science Foundation of China (NO. 32260865); Yunnan Province Young and Middle-aged Academic and Technical Leaders Reserve Talents Project (NO. 202305AC160037), Wang Junjun Expert Workstation Project of Yunnan Province (NO. 202305AF150083); Basic Research Program of Yunnan Provincial Department of Science and Technology (NO. 202101BA070001-202).
Author information
Authors and Affiliations
Contributions
Linjie Xu: conceptualization, data curation, methodology, project administration, resources, software, supervision, validation, writing-original draft. Rong Jiang: data curation, formal analysis, investigation, methodology, resources, software, validation, writing-original draft. Jingjing Su: data curation, investigation, resources, visualization, writing-review & editing. Ruixian Sun: formal analysis, methodology, resources, supervision, writing-original draft. Lilian Yang: formal analysis, project administration, resources. Yunping Tang: formal analysis, resources. Shiyan Sui: conceptualization, funding acquisition, project administration, resources, supervision, validation, writing-review & editing. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The authors confirm that this study received approval from the relevant institutional and/or national research ethics committee, specifically the Medical Ethics Committee of Dali University, under the ethics approval number MECDU-202110-4.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, L., Jiang, R., Su, J. et al. Analysis of the resistance of small peptides from Periplaneta americana to H2O2-induced apoptosis in KGN Cells based on miRNA-seq. Sci Rep (2026). https://doi.org/10.1038/s41598-026-41839-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-41839-y