Abstract
Patients with diminished ovarian reserve (DOR) often exhibit depression, which may aggravate the disease by affecting spontaneous regional activity in the brain. However, the differences of brain activity between DOR patients with and without depression are unclear. Eighty-five DOR patients including 42 depressive and 43 non-depressive patients, as well as 44 healthy controls (HC), were enrolled. Resting-state functional magnetic resonance imaging data were obtained and preprocessed to calculate the measures of fractional amplitude of low-frequency fluctuation (fALFF) and regional homogeneity (ReHo), evaluating the differences of spontaneous regional brain activity between groups. In addition, relationships between fALFF, ReHo values of altered brain regions and scores of 17-item Hamilton Depression Rating Scale (HAMD-17) were evaluated. Receiver operating characteristic (ROC) curves were also used to explore the suitability of the altered brain regions as potential neuroimaging biomarkers for evaluating the level of depression in DOR patients. Compared with HC, DOR patients with depression showed decreased intensity and concordance of regional brain activity especially in the frontal regions while DOR patients without depression exhibited decreased brain activity in the frontal, parietal regions and increased concordance of activity in the parietal, temporal regions. In addition, compared with non-depressive DOR patients, depressive patients displayed decreased brain activity in the frontal, temporal and parietal regions. DOR patients with moderate depression demonstrated decreased brain activity in the frontal and parietal regions when compared to patients with mild depression. Moreover, negative relationships were found between HAMD-17 scores and fALFF values of the right opercular part of inferior frontal gyrus, precuneus and postcentral gyrus, as well as ReHo values in the right middle cingulate gyrus and supplementary motor area. Moreover, ROC analysis revealed that both altered fALFF and ReHo values of impaired regions might be helpful for evaluating the level of depression in DOR patients. The accompanied depression in DOR patients might be associated with decreased intensity and concordance of brain activity in the frontal, temporal and parietal regions. In depressive DOR patients, the worse depression might be related to decreased brain activity in the frontal and parietal regions. These findings might provide new insights into the pathological mechanism underlying DOR with depression.
Similar content being viewed by others
Introduction
Diminished ovarian reserve (DOR) refers to the gradual decline of ovarian function, which is a common pathological condition in reproductive-age women (typically in their mid to late 30s)1. It is characterized by decreased ability of ovaries to produce eggs, resulting in decreased number of follicles in the ovary and declined quality of oocytes2. DOR clinically manifests as hypogonadism including hypomenorrhea, oligomenorrhea, amenorrhea, galactorrhea and infertility3. Therefore, DOR is considered as the main cause of female infertility4. It was estimated that 10% women were diagnosed with DOR in an infertility clinic of USA and 32% women carried vitro fertilization (IVF) were diagnosed with DOR stated by the US-based national Society for Assisted Reproductive Technology system5,6. In China, the infertility rate increased from 3% 20 years ago to 15%, and female infertility caused by DOR accounts for 10% of childbearing couples7,8. DOR patients have three related but distinctly different clinical outcomes including decreased oocyte quality, declined oocyte quantity and poor reproductive potential9. However, there is currently no uniformly accepted medical definition of DOR, which was reported by the American Reproductive Medicine Association (ASRM) Practice Committee in 201210. There are multifactorial etiology factors that cause DOR, including genetics, environmental factors, lifestyle factors such as smoking, immune factors and other destructive factors11. However, the exact pathogenesis of DOR is still unclear.
The female reproductive system is very sensitive to depression, which can reduce the number of primordial, primary and secondary follicles, as well as the number, and diameter of oocyte12. Life stress including stress-induced depression played a key role in the reduction of ovarian reserve13. It was reported that women diagnosed with DOR had higher infertility distress and experienced emotional distress, which increased their risk for psychological distress14. Therefore, depression is also considered as an important psychological factor causing DOR14. Depression exerts effects on the female reproductive system by activating the hypothalamic-pituitary-adrenal (HPA) axis15,16. In addition, the dysfunction of female reproductive system is often observed in patients with depression17. The development of depression has been found to be associated with abnormal activity of brain regions involved in the regulation of emotion, which are considered as the central pathological mechanisms underlying depression18,19. Resting-state functional magnetic resonance imaging (rs-fMRI) is a widely used and noninvasive method to evaluate the changes of brain activity in depressive patients20. Amplitude of low-frequency fluctuation (ALFF), fractional ALFF (fALFF) and regional homogeneity (ReHo) are the three major parameters for measuring the magnitude and concordance of regional spontaneous neural activity based on rs-fMRI data21,22. In patients with depression, both altered fALFF and ReHo values have been identified in brain regions of the neural circuits or networks associated with emotional recognition, processing and regulation23,24.
However, it remains unclear whether the intensity and consistency of brain activity are impaired in DOR patients as a whole, and what the rs-fMRI characteristics of DOR patients are compared to HC. More importantly, the differences in central mechanisms between DOR patients with and without depression are still unknown. In this study, we hypothesized that DOR patients with depression might exhibit decreased intensity and consistency of activity in brain regions implicated in the emotional regulation when compared with those without depression. Therefore, DOR patients with depression and without depression, as well as healthy controls (HC) were enrolled in this study. Both fALFF and ReHo values were calculated in the whole brain and these measures were compared between groups to detect the central neural mechanisms underlying DOR with depression and without depression. Moreover, associations between fALFF, ReHo values of abnormal brain regions and the level of depression were evaluated. Furthermore, we used receiver operating characteristic (ROC) curves to explore whether the altered brain regions could serve as potential neuroimaging biomarkers for evaluating the level of depression in DOR patients, which might help find more objective indicators to assist in identifying and evaluating depression in DOR patients compared with scales.
Materials and methods
Participants
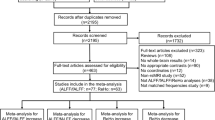
The present study was approved by the Medical Research Ethics Committee of The First Affiliated Hospital of Henan University of Chinese Medicine. This study was performed in accordance with the Declaration of Helsinki. In addition, all participants provided written informed consents prior to their participation in this study. The flow chart of this study was displayed in Fig. 1. DOR patients were acquired from the Department of Gynaecology and Obstetrics in The First Affiliated Hospital of Henan University of Chinese Medicine. The diagnosis of DOR was made according to the Bologna criteria (follicle-stimulating hormone, FSH: 10-40IU/L; luteinizing hormone, LH: 10-20IU/L; AMH, anti-Müllerian hormone: 0.5-1.1ng/mL; antral follicle count, AFC ≤ 6)25. The diagnosis of depression was made according to the Diagnostic and Statistical Manual of Mental Disorders-fifth Edition (DSM-5) criteria and the level of depression was evaluated by 17-item Hamilton Depression Rating Scale (HAMD-17). Age- and education-matched HC were recruited through community by local advertisements.
The flow chart of this study. MRI: magnetic resonance imaging; MIN: Montreal Institute of Neurology; fALFF: fractional amplitude of low-frequency fluctuation; ReHo: regional homogeneity.
The inclusion criteria for all participants were as follows: Han nationality in China, right-handed, aged 25–40 years old, more than 9 years of education level, Hamilton Anxiety Rating Scale (HAMA) scores less than 7, eliminating the interference of anxiety. In addition, DOR patients should also meet the following inclusion criteria: depression occurred after DOR, the duration of depression less than 6 months, with no history of treatment of depression and DOR. Patients with HAMD-17 scores more than 7 were classified as the group of DOR patients with depression. Patients with HAMD-17 scores less than 7 were classified as the group of DOR patients without depression. Moreover, HC should also meet the following inclusion criteria: no history of DOR, did not meet the criteria of any DSM-5 axis I disorder or personality disorders according to the Structured Clinical Interview for DSM-5 Nonpatient Edition (SCID-5, Chinese version), HAMD-17 scores less than 7.
The exclusion criteria for all participants were as follows: (1) depression caused by other organic, psychotic disorders or drugs; (2) combined with other psychiatric disorders; (3) severe somatic disease; (4) in lactating, menstruating or pregnant stage, or use of contraceptive drugs; (5) alcohol or substance abuse or dependence; (6) macroscopic abnormalities in brain structure; (7) any other contraindication for MRI scanning.
Based on the inclusion and exclusion criteria, a total of 42 DOR patients with depression, 43 DOR patients without depression and 44 HC were enrolled in this study26. The depressive DOR patients were classified into subgroups based on the HAMD-17 scores according to standardized criteria27: a mild depression group (HAMD-17 scores: 8–16) and a moderate depression group (HAMD-17 scores: 17–23). The demographic and clinical characteristics of the three groups were presented in Table 1.
MRI data acquisition
Considering that rs-fMRI allows easier data collection and it identifies underlying intrinsic brain activity that is not confounded by differences in task performance or strategy differences when compared with task-based fMRI, therefore, rs-fMRI data were acquired from all these participants in this study. MRI data were obtained by a 3.0 Tesla MRI scanner (Ingenia, Philips). During MRI scanning, all participants were instructed to minimize movement, keep eyes closed, stay awake and think of nothing in particular. T1-weighted 3D structural images were obtained with the following parameters: repetition time (TR)=9ms; echo time (TE) = 2.48ms; slice thickness=1 mm; field of view (FOV) = 200 × 200mm2; matrix = 200 × 200; flip angle (FA) = 9°. The rs-MRI data were obtained with the following parameters: TR=3000ms; TE = 40ms; slice thickness=3 mm; FOV = 240 × 240mm2; matrix = 80 × 80; FA = 90°; time points = 240; acquisition time=12 min.
MRI data preprocessing
The software of Data Processing Assistant for Resting-State fMRI (DPARSF)28 was applied to preprocess MRI data based on MATLAB platform. The preprocessing course consisted of the following steps: removal of the initial 10 time point for signal stabilization and subject adaptation; slice timing correction for acquisition delay between slices; head motion correction and subjects with head motion more than 2.5 mm of translation or rotation more than 2.5 were excluded; reorient T1-weighted structural and functional images; remove scalp, skull and segmentation of structural data; co-registration of functional images to T1-weighted structural images; spatial normalization to the standard Montreal Neurological Institute (MNI) brain space and resampled to 3 mm cubic voxels.
fALFF and ReHo calculation
The calculation process of fALFF values were follows: spatial smoothing using Gaussian kernel (full-width at half maximum = 4 mm); linear detrending; regressing out nuisance covariates including Fristion’s 24-parameter regression, white matter, CSF and global mean signals; converting the time courses into the frequency domain by Fast Fourier Transform (FFT); ALFF value was obtained by calculating and summing the square root of the power spectrum across 0.01–0.08 Hz; fALFF value was obtained by dividing the power spectrum of low-frequency (0.01–0.08 Hz) to that of the entire frequency range; finally, fALFF values were normalized to zfALFF by Fisher’s r-to-z transformation.
The calculation process of ReHo values were follows: linear detrending; temporal bandpass filtering; regressing out nuisance covariates including Fristion’s 24-parameter regression, white matter, CSF and global mean signals; ReHo values were obtained by calculating Kendall’s Coefficient of Concordance (KCC, also called ReHo) of the time series of a given voxel with those of its nearest neighbors on a voxel-wise basis; ReHo values were transformed into z-values using Fisher’s r-to-z transformation.
Statistical analysis
Firstly, differences in demographic and clinical data among the three groups (DOR with depression, DOR without depression, HC) were assessed using one-way analysis of variance (ANOVA) with least-significant difference (LSD) post-hoc tests based on the software of Statistical Package for the Social Sciences (SPSS). The statistical significance level was set at P < 0.05 (uncorrected).
Secondly, group comparisons of fALFF and ReHo values in this study were conducted at three levels: (a) Overall DOR Effect: To investigate the effect of DOR, all DOR patients (n = 85, combining depressed and non-depressed subgroups) were compared against the HC group (HC, n = 44) using two-sample t-tests for fALFF and ReHo values. (b) Effect of Depressive State (within DOR): To explore the specific effect of depressive state (independent of DOR itself) within the context of DOR, we compared the DOR with depression group (n = 42) with the DOR without depression group (n = 43), also using two-sample t-tests. (c) Effect of Depression Severity: Within the DOR with depression group, we compared the moderate depression subgroup with the mild depression subgroup. Group differences of fALFF and ReHo values were compared using a general linear model (two-sample t-test with covariates) implemented in the software of Resting-State fMRI Data Analysis Toolkit (REST) based on MATLAB platform29. We included age, BMI, education level, HAMA (scores) and hormonal indices including FSH, LH, AMH and AFC as nuisance covariates to isolate the effect of the depressive state. The significance threshold was set at P < 0.001 (a minimum cluster size of 6 voxels), corrected for multiple comparisons using the AlphaSim program in REST software29.
Moreover, the relationships between fALFF, ReHo values of impaired brain regions and HAMD-17 scores were explored by the method of Pearson correlation analysis. The statistical significance level was set at P < 0.05 (uncorrected).
Finally, ROC analysis was used to explore the suitability of the altered brain regions as potential neuroimaging biomarkers. For the combined model, a logistic regression model was fitted using the fALFF (or ReHo) values of the significant regions as independent variables to predict the probability of group membership, and this probability was used to generate the ROC curve. The statistical significance level was set at P < 0.05 (uncorrected).
Results
Comparison of demographic and clinical data between DOR patients with, without depression and HC
One-way ANOVA results showed no significant differences among the three participant groups in age, years of education, BMI, or anxiety scale scores (all P > 0.05). Post-hoc LSD tests revealed that: (1) for depression scores, the DOR with depression group scored significantly higher than both the DOR without depression group and the healthy control group (both P < 0.001), with no difference between the latter two groups; (2) regarding hormone levels and follicle counts, there were no significant differences between the two DOR subgroups (all P > 0.05), but both subgroups showed highly significant differences compared to the healthy control group (all P < 0.001), manifested as significantly elevated follicle-stimulating hormone and luteinizing hormone levels, and significantly decreased anti-Müllerian hormone levels and antral follicle counts. (Table 1)
Comparison of fALFF values between DOR patients with, without depression and HC
In comparison with HC, declined fALFF values were discovered in the left precuneus, right middle frontal gyrus, inferior temporal gyrus and angular gyrus of DOR patients with depression (Table 2; Fig. 2).
Comparison of fALFF values between DOR patients with, without depression and HC. A DOR with depression vs. HC; B DOR with no depression vs. HC; C DOR with vs. without depression; D DOR with moderate vs. mild depression. Blue clump: brain region with decreased fALFF values; Red clump: brain region with increased fALFF values. DOR: diminished ovarian reserve; HC: healthy controls. fALFF: fractional amplitude of low-frequency fluctuation. The significance threshold was set at P < 0.001 (a minimum cluster size of 6 voxels), corrected for multiple comparisons using the AlphaSim program in REST software. Functionally decreased and increased areas were depicted in blue and red, respectively. The corresponding T-values for these regions were presented in Table 2.
Compared with HC, DOR patients without depression displayed decreased fALFF values in the left precuneus, right middle occipital gyrus and increased fALFF values in the left anterior cingulate gyrus (Table 2; Fig. 2).
In addition, DOR patients with depression had decreased fALFF values in the left medial orbital superior frontal gyrus, middle frontal gyrus, anterior cingulate gyrus, inferior temporal gyrus and right superior frontal gyrus, middle frontal gyrus, anterior cingulate gyrus, middle temporal gyrus, inferior temporal gyrus compared to those without depression (Table 2; Fig. 2).
Moreover, decreased fALFF values were identified in the right opercular part of inferior frontal gyrus, precuneus and postcentral gyrus of DOR with moderate depression compared to those with mild depression (Table 2; Fig. 2).
In this study, fALFF values were re-analyzed using the more rigorous Gaussian Random Field (GRF) correction (voxel-level P < 0.001, cluster-level P < 0.05) (Stable 1). The right middle frontal gyrus was the core finding between DOR with Depression and HC while the core finding between DOR without Depression and HC was the left precuneus and anterior cingulate gyrus. In addition, the left anterior cingulate gyrus and right superior frontal gyrus were the most critical and reliable brain regions distinguishing depressed from non-depressed DOR patients. Moreover, the difference in depression severity within DOR patients was primarily linked to the precuneus.
Comparison of ReHo values between DOR patients with, without depression and HC
Compared with HC, DOR patients with depression demonstrated decreased ReHo values in the left superior frontal gyrus, orbital part of inferior frontal gyrus, triangular part of inferior frontal gyrus, anterior cingulate gyrus, inferior temporal gyrus, right medial orbital superior frontal gyrus, superior frontal gyrus and increased ReHo values in the right fusiform gyrus (Table 3; Fig. 3).
Comparison of ReHo values between DOR patients with, without depression and HC. A DOR with depression vs. HC; B DOR with no depression vs. HC; C DOR with vs. without depression; D DOR with moderate vs. mild depression. Blue clump: brain region with decreased ReHo values; Red clump: brain region with increased ReHo values. DOR: diminished ovarian reserve; HC: healthy controls. ReHo: regional homogeneity. The significance threshold was set at P < 0.001 (a minimum cluster size of 6 voxels), corrected for multiple comparisons using the AlphaSim program in REST software. Functionally decreased and increased areas were depicted in blue and red, respectively. The corresponding T-values for these regions were presented in Table 3.
DOR patients without depression had decreased ReHo values in the left medial orbital superior frontal gyrus, precuneus, inferior parietal gyrus and increased ReHo values in the left postcentral gyrus, middle temporal gyrus, inferior temporal gyrus, right postcentral gyrus and middle temporal gyrus when compared with HC (Table 3; Fig. 3).
In addition, DOR patients with depression exhibited decreased ReHo values in the left superior frontal gyrus, middle frontal gyrus, orbital part of inferior frontal gyrus, opercular part of inferior frontal gyrus, anterior cingulate gyrus, temporal pole of superior temporal gyrus, middle temporal gyrus, right middle frontal gyrus, precuneus, postcentral gyrus, supplementary motor area and increased ReHo values in the right supramarginal gyrus when compared with those without depression (Table 3; Fig. 3).
Moreover, DOR with moderate depression showed decreased ReHo values in the right middle cingulate gyrus and supplementary motor area when compared with those with mild depression (Table 3; Fig. 3).
In this study, ReHo values were re-analyzed using the more rigorous GRF correction (voxel-level P < 0.001, cluster-level P < 0.05) (Stable 2). The left triangular part of the inferior frontal gyrus was consistently identified between DOR with depression and HC. This region, part of Broca’s area, was crucial for cognitive control, speech production, and emotional regulation. Its reduced ReHo suggested impaired local processing efficiency in this key cognitive-emotional hub in depressed DOR patients. The most critical comparison between DOR with and without depression highlighted a network involving the left middle frontal gyrus, left orbital and opercular parts of the inferior frontal gyrus, and the left middle temporal gyrus. The survival of these specific regions underscored their central role in differentiating the depressed state within the DOR population. This pointed to a core disruption in a fronto-temporal circuit governing executive function, emotional processing, and semantic cognition. The left precuneus showed reduced ReHo in non-depressed DOR patients compared to HC in both tables. This reinforced the finding from the fALFF analysis and suggested that DOR itself might be associated with an alteration in this key hub of the default mode network (DMN), possibly representing a stable neural trait or even a protective adaptation. Numerous small clusters in the cingulate cortex, supplementary motor area, postcentral gyrus, and other parts of the frontal and temporal lobes disappeared. This included all findings between DOR with moderate and mild depression. This indicated that the ReHo correlates of depression severity within DOR were less robust than the correlates of the depressed state itself. The complex, overlapping patterns of increased and decreased ReHo in the DOR with no depression and HC were distilled down to two primary findings including the left precuneus and the right postcentral gyrus. This suggested that the other alterations might be statistical noise or represent subtle, non-core aspects of the DOR phenotype without comorbid depression.
Relationships between fALFF, ReHo values and HAMD-17 scores
Negative relationships were found between HAMD-17 scores and fALFF values of the right opercular part of inferior frontal gyrus (r=-0.42; P = 0.0063), precuneus (r=-0.40; P = 0.0085) and postcentral gyrus (r=-0.44; P = 0.0036), as well as ReHo values in the right middle cingulate gyrus (r=-0.47; P = 0.0015) and supplementary motor area (r=-0.39; P = 0.012) (Fig. 4).
Relationships between fALFF, ReHo values and HAMD-17 scores. HAMD-17: 17-item Hamilton Depression Rating Scale; fALFF: fractional amplitude of low-frequency fluctuation; ReHo: regional homogeneity.
ROC analysis of altered brain regions
ROC analysis indicated that fALFF values of the right opercular part of inferior frontal gyrus, precuneus, postcentral gyrus, as well as the combined model of fALFF values had preferable ability for evaluating the level of depression in DOR patients (Table 4; Fig. 5A-B). In addition, ReHo values of the right middle cingulate gyrus, supplementary motor area and the combined model of ReHo values also could effectively evaluate the level of depression in DOR patients (Table 4; Fig. 5C-D).
0Receiver operator characteristic (ROC) curves of altered brain regions for evaluating the level of depression in DOR patients. A, B: ROC analysis of fALFF values in evaluating the level of depression in DOR patients; C, D: ROC analysis of ReHo values in evaluating the level of depression in DOR patients. fALFF: fractional amplitude of low-frequency fluctuation; ReHo: regional homogeneity. AUC: area under the curve.
In addition, k-fold cross-validation ROC analysis results (k = 10) had been provided in the Supplementary materials (SFigure 1 and SFigure 2), which provide more realistic and generalizable estimate of the model’s diagnostic power. We noted that while the cross-validated AUCs were slightly lower, as expected, they remained statistically significant and upheld our primary conclusion that the neuroimaging metrics had meaningful discriminatory power.
Moreover, ROC analysis indicated that both the combined model of fALFF and ReHo values had preferable ability for distinguishing DOR patients without depression from HC (Fig. 6).
Receiver operator characteristic (ROC) curves of altered brain regions for distinguishing DOR patients without depression from HC. DOR: diminished ovarian reserve; HC: healthy controls.
Discussion
To the best of our knowledge, this is the first study to explore the distinct central pathogenesis of depressive and non-depressive DOR based on rs-fMRI data with the measures of fALFF and ReHo. The findings showed that depressive DOR might be related to decreased intensity and concordance of brain activity in the frontal regions while non-depressive DOR might be associated with decreased brain activity in the frontal and parietal regions, as well as increased concordance of activity in the parietal and temporal regions. In addition, decreased brain activity of the frontal, temporal and parietal regions might be involved in the development of depression in DOR patients. Decreased brain activity of the frontal and parietal regions might lead to worse depression in depressive DOR patients. Moreover, negative relationships were found between HAMD-17 scores and fALFF, ReHo values of abnormal brain regions. ROC curve analysis indicated that both altered fALFF and ReHo values of impaired regions might be helpful for evaluating the level of depression in DOR patients. It was important to note that our initial ROC analysis was susceptible to overfitting, as it was performed on the same dataset used to identify the significant brain regions. To address this, we implemented a 10-fold cross-validation scheme, which provided more conservative and generalizable estimate of the classification accuracy. The fact that our cross-validated AUCs remained significant strengthens confidence in the potential of these biomarkers.
The superior discriminatory power of the combined fALFF/ReHo model in identifying non-depressed DOR patients holds significant clinical promise. It suggests that a multi-parametric neuroimaging signature could serve as an objective biomarker for DOR, complementing existing endocrine measures. Crucially, by demonstrating efficacy in a depression-free cohort, our model captures the core neuropathophysiology of DOR, independent of the confounding effects of mood disorders. This paves the way for developing auxiliary diagnostic tools that can aid in the precise subtyping of DOR, particularly in complex cases with psychiatric comorbidity. Furthermore, validating this signature in longitudinal studies could unlock its potential for early identification of at-risk individuals and for objectively monitoring disease progression or therapeutic response.
While our study utilized fALFF and ReHo to identify local aberrations in neural activity and synchronization in DOR patients with depression, it is important to acknowledge that these are relatively static, region-centric metrics. The complex pathophysiology of depression, particularly in the context of endocrine dysfunction, likely involves large-scale alterations in brain network organization and dynamics that extend beyond localized changes. Advanced analytical frameworks could provide a more holistic characterization of these disturbances. For instance, graph theory can quantify the brain’s network efficiency, delineating how well it balances specialized processing (segregation) with global information integration30. Furthermore, the brain is a dynamic system, and its time-varying functional connections may be particularly relevant to the fluctuating nature of depressive symptoms. Dynamic functional connectivity (dFC) analyses can capture these transient brain states, offering insights into the temporal instability of neural networks that static methods might obscure31,32. Finally, information-theoretic measures could probe the complexity and predictability of neural signals, potentially revealing a loss of nuanced brain dynamics associated with depressive states32.
Previous studies demonstrated that patients with ovarian dysfunction often commonly suffered from depression when compared with HC33,34,35. A previous rs-fMRI study showed that both abnormal brain activity and functional connectivity were detected at the regional and network levels in patients with polycystic ovary syndrome (PCOS)36. Compared with HC, decreased intensity of spontaneous regional brain activity were identified in the left middle frontal gyrus, posterior cingulate gyrus and right middle occipital gyrus of PCOS patients by the measure of ALFF36. In addition, PCOS patients showed increased ALFF values in the left inferior temporal gyrus and decreased ALFF values in the left superior frontal gyrus and inferior occipital gyrus, as well as decreased functional connectivity, which indicated that PCOS could cause abnormal brain activity37. Moreover, PCOS patients exhibited higher activation in the right superior parietal lobe and inferior parietal lobe during cognitive task condition, which suggested that PCOS might affect executive functioning of patients38. Increased brain activation was also found in the right orbitofrontal cortex of PCOS patients, which might be associated with the enhanced sympathoexcitation and insulin sensitivity of patients39. However, no studies have explored the changes of brain activity in DOR patients and those with depression by the technique of rs-fMRI.
In this study, in comparison with HC, DOR patients without depression showed decreased fALFF values in the left precuneus, right middle occipital gyrus and increased fALFF values in the left anterior cingulate gyrus. The precuneus is a part of superior parietal lobule and it is a core region of DMN involved in emotion and cognition regulation. The middle occipital gyrus is implicated in visual attention and sensory processing, which is also associated with cognition regulation40,41,42. The anterior cingulate gyrus is an important region located in the cognitive control network related to negative affect and cognitive control43,44. Depressive PCOS was associated with the impaired emotional and executive functions in the anterior cingulate gyrus45. In non-depressive DOR patients, decreased intensity of brain activation of the left precuneus and right middle occipital gyrus reflected less efficient emotion and cognition processing, which required greater compensatory activation of the left anterior cingulate gyrus to facilitate affect and cognitive control. Moreover, decreased consistency of brain activity was revealed in the DMN including left medial orbital superior frontal gyrus, precuneus, inferior parietal gyrus and increased consistency of brain activity was identified in the DMN and sensory-motor areas including the left postcentral gyrus, middle temporal gyrus, inferior temporal gyrus, right postcentral gyrus, middle temporal gyrus in this study. The medial orbital superior frontal gyrus, precuneus and inferior parietal gyrus are important regions of DMN, which often exhibit abnormal activation in patients with depression and cognitive impairment46,47,48. The decreased activity consistency of these regions might indicate that DOR patients were more likely to suffer from depression and cognitive dysfunction. The middle temporal gyrus is related to emotion generation and processing49,50 and the inferior temporal gyrus is associated with visual processing, which is an important part of cognitive processing51. The increased activity consistency of these two regions were hypothesized as the compensatory mechanism to maintain normal emotional and cognitive performance.
Compared with HC, DOR patients with depression showed a wide range of brain regions with decreased brain activity, particularly in the DMN and attention network. Altered ALFF values were detected in the DMN, which might be the core neuropathological changes in medication-free patients with major depressive disorder52. In addition, altered brain function was found in the DMN with the measures of ALFF and ReHo, which were related to the heterogeneous clinical performance and neurocognitive impairment of first-episode, drug naïve major depressive disorder53. Moreover, hypoconnectivity and decreased homogeneity was revealed in the attention network, which might be a useful neuroimaging biomarker for differentiating depressive patients from HC54,55. The findings of this study were consisted with previous studies, and the conjoint alterations of fALFF and ReHo in the DMN and attention network might represent the central neuropathological mechanism underlying DOR with depression. Compared to DOR patients without depression, those with depression demonstrated that brain regions with decreased intensity of activity were mainly located in the DMN and attention network while brain regions with decreased consistency of activity were primarily distributed in the DMN, attention network and sensory-motor areas. Therefore, abnormal brain activity in the DMN, attention network and sensory-motor areas might be associated with the development of depression condition in DOR patients.
In addition, the innovative discovery of this study was that DOR with moderate depression showed decreased activity intensity in the right opercular part of inferior frontal gyrus, precuneus, postcentral gyrus and decreased activity homogeneity of in the right middle cingulate gyrus and supplementary motor area when compared to those with mild depression. The decreased activation of the inferior frontal gyrus was considered as a risk indicator for clinical depression56. The reduced fALFF values of right precuneus were associated with number of depressive episodes, which indicated that the DMN played a vital role in the pathology of recurrent depression57. The level of subclinical depressive symptom was associated with functional connectivity of the postcentral gyrus in patients with remission of major depressive disorder58. The right middle cingulate gyrus showed negative associations with the disease duration in major depressive disorder patients59. The function of the supplementary motor area was related to the neurocognition of patients with major depressive disorder60. In this study, we speculated that depression might be more serious as brain activity in the frontal-parietal network getting more decreased. In addition, decreased brain activity in the frontal-parietal network might be valuable neuroimaging markers for predicting the severity of depression in DOR patients.
A key consideration raised by our findings is the extent to which the neural alterations in DOR patients with depression align with or diverge from the well-established pathophysiology of major depressive disorder (MDD). (1) Consistency with General MDD Pathophysiology: Our observation of widespread decreased spontaneous activity in the prefrontal cortex (including the inferior and middle frontal gyri), anterior cingulate gyrus, and precuneus in depressive DOR patients demonstrates significant overlap with canonical models of MDD61. For instance, the dorsolateral and ventrolateral prefrontal cortex (encompassing the middle and inferior frontal gyri) are consistently implicated in MDD, with hypoactivity linked to impaired cognitive control and emotion regulation62,63,64. Similarly, dysfunction in the anterior cingulate cortex, particularly its subgenual region, is a hallmark of MDD and is central to models of affective dysregulation65. The precuneus, as a key node of the DMN, frequently shows altered activity in MDD, often associated with ruminative self-referential thought66. Therefore, the overarching pattern in depressive DOR patients strongly resonates with the fronto-limbic and DMN dysfunction characteristic of MDD. This suggests that the comorbid depression in DOR may, in part, be mediated by these common, central depressive pathways. (2) Potential Distinctiveness and Interaction with DOR: However, several aspects of our data suggest potential interactions with the DOR condition itself. The most compelling evidence for a DOR-related effect comes from the non-depressed DOR patients. Compared to HC, this group exhibited decreased fALFF in the precuneus and increased fALFF in the anterior cingulate gyrus. The increased ACC activity in the absence of depression could be interpreted as a compensatory or adaptive mechanism, where enhanced cognitive control is recruited to maintain emotional stability despite the underlying endocrine challenge of DOR. The loss of this compensatory activation in the depressed DOR group might represent the neural correlate of the transition to clinical depression. The confluence of this MDD-like neural pattern with the specific endocrine profile of DOR is highly suggestive. Sex hormones, particularly estradiol, exert significant neuromodulatory effects on mood-regulating brain regions67,68. It is plausible that the hormonal milieu of DOR acts as a disease modifier, either potentiating the typical depressive circuits or lowering the threshold for their breakdown in vulnerable individuals. (3) In conclusion, while the neural alterations in DOR patients with depression largely mirror those found in general MDD, the unique baseline alterations in non-depressed DOR patients point to a distinct neural substrate associated with DOR itself. The co-occurrence of depression in DOR may thus be best understood as an interaction between general MDD pathophysiology and the specific endocrine and neural context of DOR. Future studies directly comparing DOR with and without depression to MDD patients without endocrine disorders are needed to definitively disentangle these effects.
Therefore, there were several limitations in the present study. Firstly, this study involved a small number of DOR patients and healthy controls, therefore, these findings should be validated in future studies with larger sample sizes. Our study involved a modest number of participants from a single center, which may limit the generalizability of our findings. The sample size, while sufficient for initial exploratory analyses using rigorous statistical thresholds, restricts the statistical power for more complex subgroup analyses and increases the risk of type II errors. Future large-scale, multi-center studies with larger cohorts are essential to validate and extend our findings, and to better account for potential confounding variables. Secondly, this was a cross-sectional study, the changes of brain activity during the process of disease progression were not observed. In addition, the exact central neural mechanisms underlying the effects of drugs on DOR-related depression remained to be elucidated. First and foremost, it is critical to emphasize the principal limitation of our cross-sectional design. Although we identified specific neuroimaging alterations in depressed women with DOR and their correlations with hormonal indices, this design cannot elucidate the direction of causality between DOR and depression. That is, we cannot determine whether ovarian decline leads to changes in brain function and mood, or whether the pathophysiological burden of chronic depression impacts ovarian reserve, or if a bidirectional relationship exists. Indeed, existing longitudinal and cohort studies provide evidence for both directions. For instance, some studies suggest that psychological distress and depression can be a risk factor for later adverse reproductive outcomes. A large prospective cohort study found that women with higher depressive symptoms had a significantly greater probability of infertility and a lower probability of live birth69. Conversely, other longitudinal research demonstrates that a diagnosis of infertility or diminished reserve itself acts as a profound stressor, significantly increasing the risk for subsequent depressive episodes70. Our findings of altered brain function in this comorbid population could represent the neural consequence of either or both of these pathways. Therefore, in future studies, we would further longitudinally follow up on changes in brain activity with treatment progression to explore the mechanisms by which psychological interventions affect depression in DOR patients. Consequently, future studies should aim to: (1) Employ prospective longitudinal cohorts to follow women with normal ovarian reserve and depression as well as those with DOR without depression, in order to clarify which factor serves as a risk predictor for the other; (2) Integrate multi-omics data with neuroimaging to deeply explore shared biological pathways (e.g., dysregulation of the hypothalamic-pituitary-adrenal/gonadal axes, inflammatory mechanisms), thereby providing direct evidence for potential causal mechanisms; and (3) Functionally validate the role of specific hormonal levels or neural circuits in the DOR-depression comorbidity using animal model interventions. Finally, this study evaluated the changes of regional brain activity instead of brain networks, therefore, the relationships between changes of both functional and structural brain network and DOR-related depression were still unclear. These limitations need to be addressed by large-scale, multicenter, longitudinal prospective, multimodal MRI studies in the future. Crucially, the absence of a control group comprising depressed patients with normal ovarian reserve limited our ability to definitively conclude that the observed neural alterations were specific to DOR. It remained possible that these patterns were associated with depressive symptomatology more broadly. Therefore, we would recruit three groups in our further studies: (a) DOR patients with depression, (b) DOR patients without depression, and (c) age-matched non-DOR women with depression. This design would allow us to directly disentangle the effects of DOR and depression and identify biomarkers that were truly specific to their co-occurrence.
In this study, the refined results in STable 1 showed more coherent findings involving the default mode network (DMN) and the fronto-cingulate cognitive control network. In non-depressed DOR patients, the left precuneus showed lower fALFF compared to HC. This might suggest a compensatory deactivation or a unique neural adaptation that protected against depression in the face of DOR. In moderately depressed patients, the right precuneus showed lower fALFF compared to mildly depressed. The precuneus is a key DMN hub for self-referential thought. Altered activity here was a well-established biomarker for depression, potentially linked to ruminative thinking. Non-depressed DOR patients showed increased fALFF in the anterior cingulate gyrus compared to HC. This could represent a compensatory mechanism-enhanced activity in this region, which was involved in emotional regulation and conflict monitoring, might help maintain mood stability despite the hormonal challenges of DOR. Depressed DOR patients showed decreased fALFF in the anterior cingulate gyrus compared to their non-depressed counterparts. This loss of compensatory capacity could be the neural “breaking point” that led to the emergence of clinical depression. The dorsolateral prefrontal regions including the right middle frontal gyrus and right superior frontal gyrus were crucial for cognitive control, executive function, and emotion regulation. Their decreased fALFF in depressed DOR patients aligned perfectly with the cognitive deficits (e.g., poor concentration, executive dysfunction) commonly reported in depression. This finding suggested that DOR-related depression shared the top-down cognitive control impairments seen in general depression.
Moreover, the most compelling finding emerging from the robust ReHo data was the dysfunction within a left-lateralized fronto-temporal network. The consistent involvement of the left inferior frontal gyrus (orbital, triangular, opercular parts) and left middle temporal gyrus strongly linked DOR-related depression to established models of depression. The inferior frontal gyrus was critical for inhibiting negative thoughts and regulating emotional responses. Its dyssynchrony could underlie the rumination and impaired cognitive control seen in depression. The co-occurrence of inferior frontal gyrus and middle temporal gyrus abnormalities might point to disturbances in the neural substrates of internal verbal thought, potentially related to depressive rumination or negative self-referential thinking. The consistent finding of reduced ReHo in the left precuneus in non-depressed DOR patients (compared to HC) was highly significant. As a central node of the DMN, the precuneus was involved in self-awareness and autobiographical memory. This alteration could represent a vulnerability factor or a neural compensatory mechanism related to the hormonal changes in DOR. It suggested that the DOR condition itself imparted a specific neural signature, which might interact with other risk factors to determine whether a patient would develop clinical depression. The robust fronto-temporal and precuneus clusters were strong candidates for neurobiological biomarkers to distinguish depressed from non-depressed DOR patients. The results steered the pathological model away from a “whole-brain” dysfunction and towards a more specific circuit-based dysfunction involving cognitive/emotional control networks (fronto-temporal) and self-referential networks (precuneus/DMN). Non-invasive neuromodulation techniques like transcranial magnetic stimulation could target these identified hubs, particularly the left middle frontal gyrus and inferior frontal gyrus, which were already established targets for treating depression.
Our study provides a foundation for understanding local neural correlates of depression in DOR. However, to build a more comprehensive model of the underlying brain dynamics, future research should prioritize the incorporation of advanced neuroimaging methodologies. Based on the emerging literature, we propose several promising directions. (1) Network-Level Analysis Using Graph Theory: Applying graph theory to resting-state fMRI data would allow for the quantification of whole-brain network topology. Key metrics, such as modularity (segregation) and global efficiency (integration), could reveal whether depression in DOR is associated with a breakdown in the optimal balance between specialized neural processing and global communication30,31. (2) Capturing Temporal Dynamics with dFC: The brain’s functional architecture is non-stationary. Employing dFC analyses, such as sliding-window correlation or co-activation pattern analysis, would enable the investigation of how functional connections wax and wane over time. This approach could identify specific, reoccurring brain states and determine if patients spend more time in states characterized by maladaptive network configurations31,32. This temporal instability may be a key biomarker for the condition. (3) Assessing Neural Complexity with Information Theory: Information-theoretic measures, such as entropy and mutual information, can quantify the complexity and predictability of the fMRI BOLD signal. A reduction in neural signal complexity has been implicated in various neuropsychiatric disorders. Applying these measures could determine if depression in DOR is characterized by a more rigid, less adaptable, and less complex neural system32. Integrating these advanced analyses with the local metrics presented here will be crucial for moving from correlative findings to a mechanistic understanding of how altered brain network dynamics contribute to the emergence and severity of depression in the context of DOR.
Conclusion
In summary, decreased intensity and consistency of spontaneous brain activity were detected in DOR patients without depression while compensated increase of activity were revealed in several regions, which might represent the compensatory mechanism to maintain normal emotion. In addition, the conjoint decreased activity in the DMN, attention network and sensory-motor areas might be the central neuropathological mechanism underlying depression in DOR patients. Moreover, more decreased activity in the frontal-parietal network might lead to more serious depression in DOR patients. These findings might contribute to new understanding about the pathological mechanism of DOR and its concomitant depression from the view of neuroimaging.
Data availability
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
References
CEDARS MI. Managing poor ovarian response in the patient with diminished ovarian reserve[J]. Fertil. Steril. 117 (4), 655–656 (2022).
ZHANG, Q. L. et al. Treatment Progress in Diminished Ovarian Reserve: Western and Chinese Medicine[J]. Chin. J. Integr. Med. 29 (4), 361–367 (2023).
ATA, B. & SEYHAN, A. Diminished ovarian reserve versus ovarian aging: overlaps and differences[J]. Curr. Opin. Obstet. Gynecol. 31 (3), 139–147 (2019).
BUNNEWELL, S. J. et al. Diminished ovarian reserve in recurrent pregnancy loss: a systematic review and meta-analysis[J]. Fertil. Steril. 113 (4), 818–827e813 (2020).
LEVI, A. J. et al. Reproductive outcome in patients with diminished ovarian reserve[J]. Fertil. Steril. 76 (4), 666–669 (2001).
SCOTT RT, J. R. Prognostic assessment of ovarian reserve[J]. Fertil. Steril. 63 (1), 1–11 (1995).
ZHOU, Z. et al. Epidemiology of infertility in China: a population-based study[J]. Bjog 125 (4), 432–441 (2018).
MESSERLIAN, C. & MACLAGAN, L. Infertility and the risk of adverse pregnancy outcomes: a systematic review and meta-analysis[J]. Hum. Reprod. 28 (1), 125–137 (2013).
NARKWICHEAN, A. et al. Efficacy of dehydroepiandrosterone to improve ovarian response in women with diminished ovarian reserve: a meta-analysis[J]. Reprod. Biol. Endocrinol. 11, 44 (2013).
Testing interpreting measures of ovarian reserve: a committee opinion[J]. Fertil. Steril. 98 (6), 1407–1415 (2012).
ZHU, Q. et al. Potential factors result in diminished ovarian reserve: a comprehensive review[J]. J. Ovarian Res. 16 (1), 208 (2023).
ARABAMERI, A. & SAMENI, H. The effects of propolis extract on ovarian tissue and oxidative stress in rats with maternal separation stress[J]. Int. J. Reprod. Biomed. 15 (8), 509–520 (2017).
ÖNEL, T. et al. The effect of maternal separation stress-induced depression on ovarian reserve in Sprague Dawley Rats: The possible role of imipramine and agmatine through a mTOR signal pathway[J]. Physiol. Behav. 269, 114270 (2023).
NICOLORO-SANTABARBARA JM, LOBEL, M. Psychological and emotional concomitants of infertility diagnosis in women with diminished ovarian reserve or anatomical cause of infertility[J]. Fertil. Steril. 108 (1), 161–167 (2017).
KIM, J. YOU S. High Housing Density-Induced Chronic Stress Diminishes Ovarian Reserve via Granulosa Cell Apoptosis by Angiotensin II Overexpression in Mice[J]. Int. J. Mol. Sci., 23(15). (2022).
LI, J. et al. Orcinol glucoside improves the depressive-like behaviors of perimenopausal depression mice through modulating activity of hypothalamic-pituitary-adrenal/ovary axis and activating BDNF- TrkB-CREB signaling pathway[J]. Phytother Res. 35 (10), 5795–5807 (2021).
PAL, L., BEVILACQUA, K. & SANTORO, N. F. Chronic psychosocial stressors are detrimental to ovarian reserve: a study of infertile women[J]. J. Psychosom. Obstet. Gynaecol. 31 (3), 130–139 (2010).
ZHOU, H. X. et al. Rumination and the default mode network: Meta-analysis of brain imaging studies and implications for depression[J]. Neuroimage 206, 116287 (2020).
KEREN, H. et al. Reward Processing in Depression: A Conceptual and Meta-Analytic Review Across fMRI and EEG Studies[J]. Am. J. Psychiatry. 175 (11), 1111–1120 (2018).
KOTOULA, V. et al. Review: The use of functional magnetic resonance imaging (fMRI) in clinical trials and experimental research studies for depression[J]. Front. Neuroimaging. 2, 1110258 (2023).
ZOU, Q. H. et al. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF[J]. J. Neurosci. Methods. 172 (1), 137–141 (2008).
ZANG, Y. et al. Regional homogeneity approach to fMRI data analysis[J]. Neuroimage 22 (1), 394–400 (2004).
LI, X. et al. Altered Regional Homogeneity and Amplitude of Low-Frequency Fluctuations Induced by Electroconvulsive Therapy for Adolescents with Depression and Suicidal Ideation[J]. Brain Sci., 12(9). (2022).
GRAY, J. P. et al. Multimodal Abnormalities of Brain Structure and Function in Major Depressive Disorder: A Meta-Analysis of Neuroimaging Studies[J]. Am. J. Psychiatry. 177 (5), 422–434 (2020).
FERRARETTI AP, LA MARCA A, FAUSER, B. C. et al. ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria[J]. Hum. Reprod. 26 (7), 1616–1624 (2011).
DESMOND, J. E. & GLOVER, G. H. Estimating sample size in functional MRI (fMRI) neuroimaging studies: statistical power analyses[J]. J. Neurosci. Methods. 118 (2), 115–128 (2002).
ZIMMERMAN, M. & MARTINEZ, J. H. Severity classification on the Hamilton Depression Rating Scale[J]. J. Affect. Disord. 150 (2), 384–388 (2013).
CHAO-GAN Y & YU-FENG Z. DPARSF: A MATLAB Toolbox for Pipeline Data Analysis of Resting-State fMRI[J]. Front. Syst. Neurosci. 4, 13 (2010).
SONG, X. W. et al. REST: a toolkit for resting-state functional magnetic resonance imaging data processing[J]. PLoS One. 6 (9), e25031 (2011).
ACHARYA, U. V. et al. Functional network connectivity imprint in febrile seizures[J]. Sci. Rep. 12 (1), 3267 (2022).
PANDA, R. et al. Whole-brain analyses indicate the impairment of posterior integration and thalamo-frontotemporal broadcasting in disorders of consciousness[J]. Hum. Brain Mapp. 44 (11), 4352–4371 (2023).
RAY, S. et al. Altered dynamic functional connectivity and reduced higher order information interaction in Parkinson’s patients with hyposmia[J]. NPJ Syst. Biol. Appl. 11 (1), 93 (2025).
BARRY, J. A. & KUCZMIERCZYK, A. R. Anxiety and depression in polycystic ovary syndrome: a systematic review and meta-analysis[J]. Hum. Reprod. 26 (9), 2442–2451 (2011).
COONEY, L. G. et al. High prevalence of moderate and severe depressive and anxiety symptoms in polycystic ovary syndrome: a systematic review and meta-analysis[J]. Hum. Reprod. 32 (5), 1075–1091 (2017).
BRUTOCAO, C. et al. Psychiatric disorders in women with polycystic ovary syndrome: a systematic review and meta-analysis[J]. Endocrine 62 (2), 318–325 (2018).
LI, G. & HU, J. Changes in Resting-State Cerebral Activity in Women With Polycystic Ovary Syndrome: A Functional MR Imaging Study[J]. Front. Endocrinol. (Lausanne). 11, 603279 (2020).
LAI, W. et al. Plasma luteinizing hormone level affects the brain activity of patients with polycystic ovary syndrome[J]. Psychoneuroendocrinology 112, 104535 (2020).
SOLEMAN RS, KREUKELS, B. P. C. et al. Does polycystic ovary syndrome affect cognition? A functional magnetic resonance imaging study exploring working memory[J]. Fertil. Steril. 105 (5), 1314–1321e1311 (2016).
LANSDOWN, A. J. et al. Regional Cerebral Activation Accompanies Sympathoexcitation in Women With Polycystic Ovary Syndrome[J]. J. Clin. Endocrinol. Metab. 104 (9), 3614–3623 (2019).
ROTSHTEIN, P. et al. Distinct and convergent visual processing of high and low spatial frequency information in faces[J]. Cereb. Cortex. 17 (11), 2713–2724 (2007).
PRADO, J. & WEISSMAN, D. H. Spatial attention influences trial-by-trial relationships between response time and functional connectivity in the visual cortex[J]. Neuroimage 54 (1), 465–473 (2011).
TU, S. et al. Category-selective attention modulates unconscious processes in the middle occipital gyrus[J]. Conscious. Cogn. 22 (2), 479–485 (2013).
TOLOMEO, S. et al. A causal role for the anterior mid-cingulate cortex in negative affect and cognitive control[J]. Brain 139 (Pt 6), 1844–1854 (2016).
RAMIREZ-MAHALUF JP, PERRAMON, J. Subgenual anterior cingulate cortex controls sadness-induced modulations of cognitive and emotional network hubs[J]. Sci. Rep. 8 (1), 8566 (2018).
IGBO, E. J. & OKOLIKO, U. Structural Changes in the Medial Prefrontal Cortex and Anterior Cingulate Cortex of Dehydroepiandrosterone-Induced Wistar Rat Model of Polycystic Ovarian Syndrome[J]. Basic. Clin. Neurosci. 13 (5), 695–708 (2022).
YAN, C. G. et al. Reduced default mode network functional connectivity in patients with recurrent major depressive disorder[J]. Proc. Natl. Acad. Sci. U S A. 116 (18), 9078–9083 (2019).
HAMILTON, J. P. et al. Depressive Rumination, the Default-Mode Network, and the Dark Matter of Clinical Neuroscience[J]. Biol. Psychiatry. 78 (4), 224–230 (2015).
EYLER LT, ELMAN, J. A. et al. Resting State Abnormalities of the Default Mode Network in Mild Cognitive Impairment: A Systematic Review and Meta-Analysis[J]. J. Alzheimers Dis. 70 (1), 107–120 (2019).
CRITCHLEY, H. et al. Explicit and implicit neural mechanisms for processing of social information from facial expressions: a functional magnetic resonance imaging study[J]. Hum. Brain Mapp. 9 (2), 93–105 (2000).
SEEHAUSEN, M. et al. Talking about social conflict in the MRI scanner: neural correlates of being empathized with[J]. Neuroimage 84, 951–961 (2014).
BALDAUF, D. & DESIMONE, R. Neural mechanisms of object-based attention[J]. Science 344 (6182), 424–427 (2014).
LI, W. et al. Characterization of brain blood flow and the amplitude of low-frequency fluctuations in major depressive disorder: A multimodal meta-analysis[J]. J. Affect. Disord. 210, 303–311 (2017).
LIANG, S. et al. Altered brain function and clinical features in patients with first-episode, drug naïve major depressive disorder: A resting-state fMRI study[J]. Psychiatry Res. Neuroimaging. 303, 111134 (2020).
KAISER RH, ANDREWS-HANNA JR, WAGER, T. D. et al. Large-Scale Network Dysfunction in Major Depressive Disorder: A Meta-analysis of Resting-State Functional Connectivity[J]. JAMA Psychiatry. 72 (6), 603–611 (2015).
GAO, Y. et al. Decreased dorsal attention network homogeneity as a potential neuroimaging biomarker for major depressive disorder[J]. J. Affect. Disord. 332, 136–142 (2023).
SHIMADA, K. et al. Subclinical maternal depressive symptoms modulate right inferior frontal response to inferring affective mental states of adults but not of infants[J]. J. Affect. Disord. 229, 32–40 (2018).
LIU, C. H. et al. Decreased Resting-State Activity in the Precuneus Is Associated With Depressive Episodes in Recurrent Depression[J]. J. Clin. Psychiatry. 78 (4), e372–e382 (2017).
KONG, Z. & ZHU, X. Somatic symptoms mediate the association between subclinical anxiety and depressive symptoms and its neuroimaging mechanisms[J]. BMC Psychiatry. 22 (1), 835 (2022).
CAO, Y. et al. Common and distinct patterns of gray matter alterations in young adults with borderline personality disorder and major depressive disorder[J]. Eur. Arch. Psychiatry Clin. Neurosci. 272 (8), 1569–1582 (2022).
WEI, J. et al. Disrupted association between structural and functional coupling of the supplementary motor area and neurocognition in major depressive disorder[J]. Chin. Med. J. (Engl). 136 (17), 2131–2133 (2023).
TANG, S. et al. Abnormal amygdala resting-state functional connectivity in adults and adolescents with major depressive disorder: A comparative meta-analysis[J]. EBioMedicine 36, 436–445 (2018).
NEJATI, V. & MAJIDINEZHAD, M. The role of the dorsolateral and ventromedial prefrontal cortex in emotion regulation in females with major depressive disorder (MDD): A tDCS study[J]. J. Psychiatr Res. 148, 149–158 (2022).
ZHANG, X. & ZHANG, R. Correlation between cognitive deficits and dorsolateral prefrontal cortex functional connectivity in first-episode depression[J]. J. Affect. Disord. 312, 152–158 (2022).
LEE, Y. J. & PARK, S. Y. Reduced left ventrolateral prefrontal cortex activation during verbal fluency tasks is associated with suicidal ideation severity in medication-naïve young adults with major depressive disorder: A functional near-infrared spectroscopy study[J]. Psychiatry Res. Neuroimaging. 312, 111288 (2021).
PENG, X. et al. Sub-regional anterior cingulate cortex functional connectivity revealed default network subsystem dysfunction in patients with major depressive disorder[J]. Psychol. Med. 51 (10), 1687–1695 (2021).
ZHANG, L. et al. Shared and distinct patterns of default mode network dysfunction in major depressive disorder and bipolar disorder: A comparative meta-analysis[J]. J. Affect. Disord. 368, 23–32 (2025).
PAN, Q. et al. Estradiol exerts a neuroprotective effect on SH-SY5Y cells through the miR-106b-5p/TXNIP axis[J]. J. Biochem. Mol. Toxicol. 35 (9), e22861 (2021).
SIKES-KEILP C, RUBINOW, D. R. In search of sex-related mediators of affective illness[J]. Biol. Sex. Differ. 12 (1), 55 (2021).
NILLNI, Y. I. et al. Depression, anxiety, and psychotropic medication use and fecundability[J]. Am. J. Obstet. Gynecol. 215 (4), 453e451–453e458 (2016).
GAMEIRO, S. et al. Women’s adjustment trajectories during IVF and impact on mental health 11–17 years later[J]. Hum. Reprod. 31 (8), 1788–1798 (2016).
Funding
The work was supported by the grants of: Special Project for Scientific Research of Traditional Chinese Medicine in Henan Province (Key Project) (No. 2023ZYZD09); Henan Province Key Research and Promotion Special Project (Science and Technology Tackle) (No. 222102310139).
Author information
Authors and Affiliations
Contributions
Miao Guo and Dawei Zhang designed the experiments. Miao Guo, Yihan Li, Jing Feng, Ping Chen, Liya Ma, Shurong Li, Xuan Zhou and Dawei Zhang contributed to clinical data collection and assessment. Miao Guo, Siwen Liu and Dawei Zhang analyzed the results. Miao Guo, Siwen Liu and Dawei Zhang wrote the manuscript. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest Disclosures
All authors declared that they had no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, M., Li, Y., Feng, J. et al. Changes of regional brain activity associated with the occurrence and severity of depression in diminished ovarian reserve patients. Sci Rep 16, 12827 (2026). https://doi.org/10.1038/s41598-026-41986-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-41986-2