Abstract
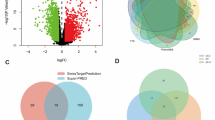
To investigate the molecular mechanism of Astragalus membranaceus (AM) in alleviating silicosis fibrosis using bioinformatics and in vitro experiments. Active ingredients of AM and their corresponding targets were retrieved from the TCMSP and SwissTargetPrediction databases. These ingredients were screened based on oral bioavailability (OB ≥ 30%), drug-likeness (DL ≥ 0.18), and Lipinski’s Rule of Five, which serves as a standard for identifying drug-like molecules with favorable pharmacokinetic properties in network pharmacology. Silicosis-related targets were collected from DisGENET, GeneCards, and the Comparative Toxicogenomics Database (CTD). Common targets were identified for protein-protein interaction (PPI) network analysis, Gene Ontology (GO), and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analyses. Core ingredients and targets were selected through topological analysis in Cytoscape and subsequently validated using molecular docking (AutoDock), immunofluorescence and RT-qPCR assays. Eight main active ingredients of AM were identified, along with 236 AM targets and 7,706 silicosis targets, resulting in 113 shared targets. Further screening revealed three key ingredients: Quercetin, isorhamnetin, and kaempferol, as well as seven core targets: AKT1, CASP3, PTGS2, MMP9, ESR1, JUN and EGFR. The primary pathways involved included PI3K-AKT, IL-17, and MAPK. Molecular docking analyses indicated that MMP9 and EGFR exhibited higher binding energies with Quercetin. In vitro immunofluorescence results demonstrated that Quercetin downregulated the expression of MMP9, EGFR, and α-SMA in silicotic fibroblasts. Additionally, qRT-PCR assays confirmed that the Quercetin alleviated SiO2-induced fibrotic process by suppressing PI3K-AKT pathway in the anti-fibrotic process. Quercetin, the key active ingredient of AM, exerts its anti-silicosis effect by inhibiting the expression of EGFR, MMP9 and the PI3K/Akt pathway.
Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ECM:
-
Extracellular matrix
- TCM:
-
Traditional Chinese medicine
- AM:
-
Astragalus membranaceus
- NP:
-
Network pharmacology
- GO:
-
Gene ontology
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- PPI:
-
Protein‒protein interaction
- BP:
-
Biological process
- CC:
-
Cytological component
- MF:
-
Molecular function
- AKT1:
-
RAC-alpha serine/threonine-protein kinase
- CASP3:
-
Caspase-3
- EGFR:
-
Epidermal growth factor receptor
- MMP9:
-
Matrix metalloproteinase 9
- ESR1:
-
Estrogen receptor
- JUN:
-
Jun proto-oncogene, AP-1 transcription factor subunit
References
Leung, C. C., Yu, I. T. & Chen, W. Silicosis. Lancet 379, 2008–2018 (2012).
Jamshidi, P. et al. Silicosis and tuberculosis: A systematic review and meta-analysis. Pulmonology 31, 2416791 (2025).
Liu, X., Jiang, Q., Wu, P., Han, L. & Zhou, P. Global incidence, prevalence and disease burden of silicosis: 30 years’ overview and forecasted trends. BMC Public Health 23, 1366 (2023).
Fernández Álvarez, R. et al. Guidelines for the diagnosis and monitoring of silicosis. Arch. Bronconeumol. 51, 86–93 (2015).
McEwen, K. & Brodie, L. Lung transplantation for silicosis and recovery: An Australian case study. Br. J. Nurs. 30, 178–183 (2021).
Dong, N. et al. Astragalus polysaccharides alleviates LPS‐induced inflammation via the NF‐κB/MAPK signaling pathway. J. Cell. Physiol. 235, 5525–5540 (2020).
Dong, M., Li, J., Yang, D., Li, M. & Wei, J. Biosynthesis and pharmacological activities of flavonoids, triterpene saponins and polysaccharides derived from *Astragalus membranaceus*. Molecules https://doi.org/10.3390/molecules28135018 (2023).
Zhang, X., Qu, X. & Zou, Y. The effect of astragalus on humoral and cellular immune response: A systematic review and meta-analysis of human studies. Complement. Med. Res. 30, 535–543 (2023).
Dan, L. et al. Efficacy and potential mechanisms of the main active ingredients of Astragalus mongholicus in animal models of liver fibrosis: A systematic review and meta-analysis. J. Ethnopharmacol. 319, 117198 (2024).
Li, X. et al. Network pharmacology approaches for research of Traditional Chinese Medicines. Chin. J. Nat. Med. 21, 323–332 (2023).
Sahu, D. et al. A review on molecular docking as an interpretative tool for molecular targets in disease management. Assay Drug Dev. Technol. 22, 40–50 (2024).
Dasgupta, S. Identification and molecular modelling of potential drugs targeting the genes involved in the progression of lung cancer in patients with idiopathic pulmonary fibrosis. Gene Rep. 37, 102067 (2024).
Li, N. et al. Bioinformatic analysis of differentially expressed genes and pathways in idiopathic pulmonary fibrosis. Annals Trans. Med. 9, 1459 (2021).
Wu, Z. et al. Identifying potential biomarkers of idiopathic pulmonary fibrosis through machine learning analysis. Sci. Rep. 13, 16559 (2023).
Sun, W. et al. ALKBH5 promotes lung fibroblast activation and silica-induced pulmonary fibrosis through miR-320a-3p and FOXM1. Cell. Mol. Biol. Lett. 27, 26 (2022).
Du, Y., Huang, F., Guan, L. & Zeng, M. Role of PI3K/Akt/mTOR pathway-mediated macrophage autophagy in affecting the phenotype transformation of lung fibroblasts induced by silica dust exposure. Zhongnan Daxue Xuebao (Yixue Ban) 48, 1152–1162 (2023).
Kasai, H., Allen, J. T., Mason, R. M., Kamimura, T. & Zhang, Z. TGF-beta1 induces human alveolar epithelial to mesenchymal cell transition (EMT). Respir. Res. 6, 56 (2005).
Cheng, W. H. et al. Amphiregulin induces CCN2 and fibronectin expression by TGF-β through EGFR-dependent pathway in lung epithelial cells. Respir. Res. 23, 381 (2022).
Zhang, Y. et al. Astragali radix (Huangqi): A time-honored nourishing herbal medicine. Chin. Med. 19, 119 (2024).
Ru, J. et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J. Cheminform. 6, 13 (2014).
Cao, Z. et al. A novel pathophysiological classification of silicosis models provides some new insights into the progression of the disease. Ecotoxicol. Environ. Saf. 202, 110834 (2020).
Barabási, A. L. & Oltvai, Z. N. Network biology: Understanding the cell’s functional organization. Nat. Rev. Genet. 5, 101–113 (2004).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28, 1947–1951 (2019).
Kanehisa, M., Furumichi, M., Sato, Y., Matsuura, Y. & Ishiguro-Watanabe, M. KEGG: Biological systems database as a model of the real world. Nucleic Acids Res. 53, D672-d677 (2025).
Kanehisa, M. & Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Mondal, J., Friesner, R. A. & Berne, B. J. Role of desolvation in thermodynamics and kinetics of ligand binding to a kinase. J. Chem. Theory Comput. 10, 5696–5705 (2014).
Wabik, J., Kurcinski, M. & Kolinski, A. Coarse-grained modeling of peptide docking associated with large conformation transitions of the binding protein: Troponin I fragment-Troponin C system. Molecules 20, 10763–10780 (2015).
Ivanov, K. I. et al. Phosphorylation regulates FOXC2-mediated transcription in lymphatic endothelial cells. Mol. Cell. Biol. 33, 3749–3761 (2013).
Liu, Z. et al. MicroRNA-146a negatively regulates PTGS2 expression induced by Helicobacter pylori in human gastric epithelial cells. J. Gastroenterol. 48, 86–92 (2013).
Huang, M. et al. Quercetin: A flavonoid with potential for treating acute lung injury. Drug Des. Devel. Ther. 18, 5709–5728 (2024).
Kim, M. et al. Quercetin and isorhamnetin attenuate benzo[a]pyrene-induced toxicity by modulating detoxification enzymes through the AhR and NRF2 signaling pathways. Antioxidants (Basel) 10, 787 (2021).
Sharma, A. & Wairkar, S. Flavonoids for treating pulmonary fibrosis: Present status and future prospects. Phytother. Res. 38, 4406–4423 (2024).
Yang, D., Wang, T., Long, M. & Li, P. Quercetin: Its main pharmacological activity and potential application in clinical medicine. Oxid. Med. Cell Longev. 2020, 8825387 (2020).
Espindola, M. S. et al. Differential responses to targeting matrix metalloproteinase 9 in idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 203, 458–470 (2021).
Dülger, S. U., Mutlu, N., Ceylan, İ & Özhan, E. The relationship between lung fibrosis, the epidermal growth factor receptor, and disease outcomes in COVID-19 pneumonia: A postmortem evaluation. Clin. Exp. Med. 23, 1181–1188 (2023).
Li, T., Yang, X., Xu, H., Liu, H. & Moitra, S. Early identification, accurate diagnosis, and treatment of silicosis. Can. Respir. J. 2022, 1–6 (2022).
You, Y., Yuan, H., Min, H., Li, C. & Chen, J. Fibroblast-derived CXCL14 aggravates crystalline silica–induced pulmonary fibrosis by mediating polarization and recruitment of interstitial macrophages. J. Hazard. Mater. 460, 132489 (2023).
Du, Q. et al. Therapeutic effect of Astragali Radix extract injection combined with bone marrow mesenchymal stem cells in Bleomycin-induced pulmonary fibrotic rats. Evid. Based Complement. Altern. Med. 2022, 1–12 (2022).
Liu, N. et al. Isorhamnetin inhibits liver fibrosis by reducing autophagy and inhibiting extracellular matrix formation via the TGF-β1/Smad3 and TGF-β1/p38 MAPK pathways. Mediators Inflamm 2019, 6175091 (2019).
Chen, J. et al. Kaempferol attenuates macrophage M1 polarization and liver fibrosis by inhibiting mitogen-activated protein kinase/nuclear factor κB signaling pathway. J. Pharmacol. Exp. Ther. 392, 103533 (2025).
Cuijpers, I. et al. Quercetin, kaempferol and capsaicin counteract the TGF-β1-induced upregulation of αSMA and collagen in myoblasts. Int. J. Mol. Sci. https://doi.org/10.3390/ijms26115151 (2025).
Wang, Q., Wang, F., Li, X., Ma, Z. & Jiang, D. Quercetin inhibits the amphiregulin/EGFR signaling-mediated renal tubular epithelial-mesenchymal transition and renal fibrosis in obstructive nephropathy. Phytother. Res. 37, 111–123 (2023).
Li, X. et al. The flavonoid Quercetin ameliorates liver inflammation and fibrosis by regulating hepatic macrophages activation and polarization in mice. Front. Pharmacol. 9, 72 (2018).
Wu, X. et al. Use quercetin for pulmonary fibrosis: A preclinical systematic review and meta-analysis. Inflammopharmacology https://doi.org/10.1007/s10787-025-01678-1 (2025).
Liu, X., Liang, Q., Qin, Y., Chen, Z. & Yue, R. Advances and perspectives on the anti-fibrotic mechanisms of the Quercetin. Am. J. Chin. Med. 53, 1411–1440 (2025).
Alharbi, H. O. A., Alshebremi, M., Babiker, A. Y. & Rahmani, A. H. The role of quercetin, a flavonoid in the management of pathogenesis through regulation of oxidative stress, inflammation, and biological activities. Biomolecules https://doi.org/10.3390/biom15010151 (2025).
Talbott, H. E., Mascharak, S., Griffin, M., Wan, D. C. & Longaker, M. T. Wound healing, fibroblast heterogeneity, and fibrosis. Cell Stem Cell 29, 1161–1180 (2022).
Seki, E. et al. TLR4 enhances TGF-β signaling and hepatic fibrosis. Nat. Med. 13, 1324–1332 (2007).
Szabo, G., Mandrekar, P. & Dolganiuc, A. Innate immune response and hepatic inflammation. Semin. Liver Dis. 27, 339–350 (2007).
Wang, M. et al. Gefitinib and fostamatinib target EGFR and SYK to attenuate silicosis: A multi-omics study with drug exploration. Signal Transduct. Target. Ther. 7, 157 (2022).
Peng, L. et al. Scutellarin ameliorates pulmonary fibrosis through inhibiting NF-κB/NLRP3-mediated epithelial–mesenchymal transition and inflammation. Cell Death Dis. 11, 978 (2020).
Li, G. et al. Macrophage-secreted TSLP and MMP9 promote bleomycin-induced pulmonary fibrosis. Toxicol. Appl. Pharmacol. 366, 10–16 (2019).
Geng, F. et al. Quercetin alleviates pulmonary fibrosis in mice exposed to silica by inhibiting macrophage senescence. Front. Pharmacol. 13, 912029 (2022).
Hao, X. et al. Inhibition of oncogenic Src ameliorates silica-induced pulmonary fibrosis via PI3K/AKT pathway. Int. J. Mol. Sci. https://doi.org/10.3390/ijms24010774 (2023).
Lv, W. et al. Circular RNA CircCOL5A1 sponges the MiR-7-5p/Epac1 axis to promote the progression of keloids through regulating PI3K/Akt signaling pathway. Front. Cell Dev. Biol. 9, 626027 (2021).
Yong, S., Guo, Y., Chen, X., Xu, Y. & Hu, Y. [Mechanism of IL-17 signaling pathway in spleen inflammatory response induced by altitude hypoxia in mice]. Sichuan Da Xue Xue Bao Yi Xue Ban 55, 118–124 (2024).
Park, H. B. & Baek, K. H. E3 ligases and deubiquitinating enzymes regulating the MAPK signaling pathway in cancers. Biochim. Biophys. Acta Rev. Cancer 1877, 188736 (2022).
Long, L. et al. Mefunidone alleviates silica-induced inflammation and fibrosis by inhibiting the TLR4-NF-κB/MAPK pathway and attenuating pyroptosis in murine macrophages. Biomed. Pharmacother. 178, 117216 (2024).
Schramm, F., Schaefer, L. & Wygrecka, M. EGFR signaling in lung fibrosis. Cells https://doi.org/10.3390/cells11060986 (2022).
Siddika, T. et al. Delivery of active AKT1 to human cells. Cells https://doi.org/10.3390/cells11233834 (2022).
Nie, Y. et al. Akt1 regulates pulmonary fibrosis via modulating IL-13 expression in macrophages. Innate Immun. 25, 451–461 (2019).
Leppä, S. & Bohmann, D. Diverse functions of JNK signaling and c-Jun in stress response and apoptosis. Oncogene 18, 6158–6162 (1999).
Li, Y. et al. GSDME-mediated pyroptosis promotes inflammation and fibrosis in obstructive nephropathy. Cell Death Differ. 28, 2333–2350 (2021).
Jiang, H. et al. Exploring the role and mechanism of Astragalus membranaceus and radix paeoniae rubra in idiopathic pulmonary fibrosis through network pharmacology and experimental validation. Sci. Rep. 13, 10110 (2023).
Acknowledgements
We express our gratitude to all the authors for their assistance and support.
Funding
This work was supported by the National Natural Science Foundation of China (82271626, 82160088, 82560099), Ningxia Hui Autonomous Region Youth Top Talent Training Project. Chinese Academy of Sciences “Western Young Scholars” project. China Postdoctoral Science Foundation (2023MD734192), Ningxia Natural Science Foundation (2025AAC050075, 2022AAC03128, 2022AAC05025), Ningxia Key Research and Development Projects (2020BFH02003, 2021BEG02030), Open competition mechanism to select the best candidates for key research projects of Ningxia Medical University (XJKF230106).
Author information
Authors and Affiliations
Contributions
Y(Y.A.N.) and L.were responsible for most of the data collection and conducted most of the experiments. G and C have made contributions to some data collection and data integration. W and Z(Z.H.N.)made contributions to some experimental work. Y and Z(Z.W.Y.) participated in the drafting of the manuscript. All authors have reviewed the subsequent versions of the manuscript and have read and approved the final version submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This project was approved by the Medical Ethics Committee of Ningxia Medical University. All experiments and strategies follow ethical principles.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, A., Luo, X., Guo, Y. et al. The network pharmacology prediction and experiment validation of Astragalus membranaceus for alleviating silicosis fibrosis via decreasing MMP9 and EGFR expression. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42544-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42544-6