Abstract
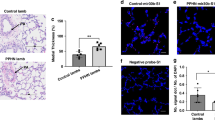
Pulmonary Arterial Hypertension (PAH) is a rare, progressive disorder characterized by pathological vascular remodeling of the pulmonary arteries, ultimately leading to right heart failure. Early diagnosis remains challenging, as clinical manifestations are often nonspecific, and definitive confirmation still requires invasive right heart catheterization. Circulating microRNAs (miRNAs) have emerged as promising biomarkers for cardiovascular diseases due to their plasma stability and direct involvement in disease pathophysiology. We performed small RNA sequencing on plasma samples from 25 IPAH patients and 10 healthy controls to identify differentially expressed miRNAs. Seven candidate miRNAs were selected for further validation by quantitative PCR in a multicenter cohort of 110 IPAH patients and controls. Logistic regression models were built to evaluate diagnostic performance. Functional studies for miR-3168 were performed using western blot and flow cytometry in Pulmonary Artery Endothelial Cells (PAECs), and tube formation assay in human umbilical vein endothelial cells (HUVECs). Our initial screen identified 29 differentially expressed miRNAs. Seven of these candidates, including members of the let-7 family and miR-3168, were successfully validated in the larger cohort. A diagnostic panel incorporating three miRNAs (let-7a-5p, miR-9-5p, and miR-31-5p) was developed, which achieved an area under the curve (AUC) of 0.862 (95% CI = 0.7481–1) for discriminating PAH patients from controls. Separately, we investigated the functional role of miR-3168, which was upregulated in PAH patients. In PAECs, overexpression of miR-3168 led to a reduction of BMPR2 protein levels. Moreover, miR-3168 overexpression impaired tube formation in HUVECs. Our study identifies a plasma-derived three-miRNA signature with strong potential for the non-invasive diagnosis of PAH. Furthermore, we implicate miR-3168 as a potential contributor to endothelial dysfunction through its regulation of BMPR2 and its anti-angiogenic effects in vitro. These findings reinforce the dual utility of circulating miRNAs as both clinically relevant non-invasive biomarkers and as tools to discover novel disease biology.
Similar content being viewed by others
Data availability
All data generated in this article is available upon request to the corresponding author. RNA-seq derived data is freely available at GSE222022, DESeq2 analysis table can be accessed in the supplementary dataset.
Abbreviations
- 3′UTR:
-
3′ Untranslated region
- 6MWD:
-
6-Minute walking distance
- AUC:
-
Area under the curve
- BMPR1A:
-
Bone morphogenetic protein receptor type 1A
- BMPR2:
-
Bone morphogenetic protein receptor type 2
- BNP:
-
Brain natriuretic peptide
- DCs:
-
Dendritic cells
- DE:
-
Differentially expressed
- EDN1/ET-1:
-
Endothelin 1
- FC-NYHA:
-
Functional class in the New York Heart Association
- FDR:
-
False discovery rate
- HDAC2:
-
Histone deacetylase 2
- HPAH:
-
Heritable pulmonary arterial hypertension
- HUVECs:
-
Human umbilical vein endothelial cells
- iNO:
-
Inhaled nitric oxide
- IPAH:
-
Idiopathic pulmonary arterial hypertension
- miRNAs:
-
MicroRNAs
- NT-proBNP:
-
N-terminal pro b-type natriuretic peptide
- PAECs:
-
Pulmonary arterial endothelial cells
- PAH:
-
Pulmonary arterial hypertension
- PCA:
-
Principal component analysis
- PVR:
-
Pulmonary vascular resistance
- qPCR:
-
Quantitative polymerase chain reaction
- RHC:
-
Right heart catheterization
- RNA:
-
Ribonucleic acid
- ROC:
-
Receiver operating characteristic
- SMC:
-
Smooth Muscle Cell
References
Humbert M, Kovacs G, Hoeper MM, Badagliacca R, Berger RMF, Brida M, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: Developed by the task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). Endorsed by the International Society for Heart and Lung Transplantation (ISHLT) and the European Reference Network on rare respiratory diseases (ERN-LUNG). European Heart Journal. 2022 ehac237.
Southgate L, Machado RD, Gräf S, Morrell NW. Molecular genetic framework underlying pulmonary arterial hypertension. Nature Reviews Cardiology. 2019 Agosto;
Galiè, N. et al. ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Respir. J. 46(4), 903–975 (2015).
Hoeper, M. M. et al. Definitions and Diagnosis of Pulmonary Hypertension. J. Am. Coll. Cardiol. 62(25), D42-50 (2014).
Lau, E. M. T., Giannoulatou, E., Celermajer, D. S. & Humbert, M. Epidemiology and treatment of pulmonary arterial hypertension. Nat. Rev. Cardiol. 14(10), 603–614 (2017).
Schoen, S. P. et al. NT-proBNP correlates with right heart haemodynamic parameters and volumes in patients with atrial septal defects. Eur J Heart Fail. 9(6–7), 660–666 (2007).
Rhodes, C. J. et al. Plasma metabolomics implicates modified transfer RNAs and altered bioenergetics in the outcomes of pulmonary arterial hypertension. Circulation 135(5), 460–475 (2017).
Sweatt, A. J. et al. Discovery of distinct immune phenotypes using machine learning in pulmonary arterial hypertension. Circ Res. 124(6), 904–919 (2019).
Bauer, Y. et al. Identifying early pulmonary arterial hypertension biomarkers in systemic sclerosis: machine learning on proteomics from the DETECT cohort. Eur Respir J. 57(6), 2002591 (2021).
Rhodes, C. J. et al. Whole-blood RNA profiles associated with pulmonary arterial hypertension and clinical outcome. Am J Respir Crit Care Med. 202(4), 586–594 (2020).
Farh, K. K. H. et al. The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science 310(5755), 1817–1821 (2005).
Chen, W. & Li, S. Circulating microRNA as a novel biomarker for pulmonary arterial hypertension due to congenital heart disease. Pediatr. Cardiol. 38(1), 86–94 (2017).
Kheyfets, V. O. et al. Circulating miRNAs in pediatric pulmonary hypertension show promise as biomarkers of vascular function. Oxid. Med. Cell. Longev. 2017, 1–11 (2017).
Chouvarine, P. et al. Trans-right-ventricle and transpulmonary microrna gradients in human pulmonary arterial hypertension. Pediatr Crit Care Med. 21(4), 340–349 (2020).
Errington, N. et al. A diagnostic miRNA signature for pulmonary arterial hypertension using a consensus machine learning approach. EBioMedicine 69, 103444 (2021).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10(3), R25–R25 (2009).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17(1), 10–12 (2011).
Liao, Y., Smyth, G. K. & Shi, W. The R package Rsubread is easier, faster, cheaper and better for alignment and quantification of RNA sequencing reads. Nucleic Acids Res. 47(8), e47–e47 (2019).
Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol [Internet]. 2014 [cited 2021 June 7];15(12). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4302049/
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1), 139–140 (2010).
Wickham H. ggplot2: Elegant Graphics for Data Analysis. 2nd ed. 2016. Cham: Springer International Publishing : Imprint: Springer; 2016. 1 p. (Use R!).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3(7), RESEARCH0034 (2002).
Vigneron, N. et al. Towards a new standardized method for circulating miRNAs profiling in clinical studies: Interest of the exogenous normalization to improve miRNA signature accuracy. Mol. Oncol. 10(7), 981–992 (2016).
Matias-Garcia, P. R. et al. Impact of long-term storage and freeze-thawing on eight circulating microRNAs in plasma samples. PLoS ONE 15(1), e0227648 (2020).
Robin, X. et al. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinf. 17(12), 77–77 (2011).
Hoeper MM, Pausch C, Olsson KM, Huscher D, Pittrow D, Grünig E, et al. COMPERA 2.0: a refined four-stratum risk assessment model for pulmonary arterial hypertension. European Respiratory Journal [Internet]. 2022 July 1 [cited 2024 Mar 11];60(1). Available from: https://erj.ersjournals.com/content/60/1/2102311
Kozomara, A. & Griffiths-Jones, S. miRBase: Integrating microRNA annotation and deep-sequencing data. Nucleic Acids Res. 39(Database issue), D152-157 (2011).
Pousada, G. et al. Functional assessment of the \textlessi\textgreaterBMPR2\textless/i\textgreater gene in lymphoblastoid cell lines from Graves’ disease patients. J. Cell Mol. Med. 22(3), 1538–1547 (2018).
Welinder, C. & Ekblad, L. Coomassie Staining as loading control in western blot analysis. J Proteome Res. 10(3), 1416–1419 (2011).
Boizeau, M. L. et al. Automated image analysis of in vitro angiogenesis assay. J Lab Autom. 18(5), 411–415 (2013).
Liu B, Azfar M, Legchenko E, West JA, Martin S, Van den Haute C, et al. ATP13A3 variants promote pulmonary arterial hypertension by disrupting polyamine transport. Cardiovasc Res. 2024 cvae068.
Machado, R. D. et al. Biallelic variants of ATP13A3 cause dose-dependent childhood-onset pulmonary arterial hypertension characterised by extreme morbidity and mortality. J. Med. Genet. 59(9), 906–911 (2022).
Caruso, P. et al. Dynamic changes in lung microRNA profiles during the development of pulmonary hypertension due to chronic hypoxia and monocrotaline. Arterioscler. Thromb. Vasc. Biol. 30(4), 716–723 (2010).
Fabro, A. T. et al. Circulating plasma miRNA and clinical/hemodynamic characteristics provide additional predictive information about acute pulmonary thromboembolism, chronic thromboembolic pulmonary hypertension and idiopathic pulmonary hypertension. Front. Pharmacol. 12, 1287 (2021).
Johnson, C. D. et al. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res. 67(16), 7713–7722 (2007).
Calatayud, D. et al. Tissue MicroRNA profiles as diagnostic and prognostic biomarkers in patients with resectable pancreatic ductal adenocarcinoma and periampullary cancers. Biomark. Res. 5, 8 (2017).
Shell, S. et al. Let-7 expression defines two differentiation stages of cancer. Proc. Natl. Acad. Sci. U S A. 104(27), 11400–11405 (2007).
Yang, N. et al. MicroRNA microarray identifies Let-7i as a novel biomarker and therapeutic target in human epithelial ovarian cancer. Cancer Res. 68(24), 10307–10314 (2008).
Caldeira-Dias, M. et al. Preeclamptic plasma stimulates the expression of miRNAs, leading to a decrease in endothelin-1 production in endothelial cells. Pregnancy. Hypertens. 12, 75–81 (2018).
Tasena, H. et al. microRNA-mRNA regulatory networks underlying chronic mucus hypersecretion in COPD. Eur. Respir. J. 52(3), 1701556 (2018).
Chen, X. et al. MiR-9 promotes tumorigenesis and angiogenesis and is activated by MYC and OCT4 in human glioma. J. Exp. Clin. Cancer Res. 38(1), 99 (2019).
Yao, X., Xie, L. & Zeng, Y. MiR-9 promotes angiogenesis via targeting on Sphingosine-1- Phosphate Receptor 1. Front. Cell Dev. Biol. 8, 755 (2020).
Gwak, J. M. et al. MicroRNA-9 is associated with epithelial-mesenchymal transition, breast cancer stem cell phenotype, and tumor progression in breast cancer. Breast Cancer. Res. Treat. 147(1), 39–49 (2014).
Baroni, S. et al. Exosome-mediated delivery of miR-9 induces cancer-associated fibroblast-like properties in human breast fibroblasts. Cell Death Dis. 7(7), e2312 (2016).
Lv, C. et al. MiR-31 promotes mammary stem cell expansion and breast tumorigenesis by suppressing Wnt signaling antagonists. Nat Commun. 19(8), 1036 (2017).
Mi, B., Li, Q., Li, T., Liu, G. & Sai, J. High miR-31-5p expression promotes colon adenocarcinoma progression by targeting TNS1. Aging (Albany NY). 12(8), 7480–7490 (2020).
Lu, Z. et al. miR-31-5p is a potential circulating biomarker and therapeutic target for oral cancer. Mol. Ther. Nucleic Acids. 7(16), 471–480 (2019).
Peng, Q. S. et al. circRNA_0000140 suppresses oral squamous cell carcinoma growth and metastasis by targeting miR-31 to inhibit Hippo signaling pathway. Cell Death Dis. 11(2), 112 (2020).
Bi, J. et al. Circ-BPTF promotes bladder cancer progression and recurrence through the miR-31-5p/RAB27A axis. Aging (Albany NY). 10(8), 1964–1976 (2018).
Yang, K. et al. Prevention of aortic dissection and aneurysm via an ALDH2-mediated switch in vascular smooth muscle cell phenotype. Eur Heart J. 41(26), 2442–2453 (2020).
Körner, C. et al. MicroRNA-31 sensitizes human breast cells to apoptosis by direct targeting of protein kinase C epsilon (PKCepsilon). J. Biol. Chem. 288(12), 8750–8761 (2013).
Chouvarine, P. et al. RNA expression profiles and regulatory networks in human right ventricular hypertrophy due to high pressure load. iScience. 24(3), 102232 (2021).
Hopper, R. K. et al. In pulmonary arterial hypertension, reduced BMPR2 promotes endothelial-to-mesenchymal transition via HMGA1 and its target slug. Circulation 133(18), 1783–1794 (2016).
Sánchez-Duffhues, G. et al. Inflammation induces endothelial-to-mesenchymal transition and promotes vascular calcification through downregulation of BMPR2. J. Pathol. 247(3), 333–346 (2019).
Spiekerkoetter, E. et al. FK506 activates BMPR2, rescues endothelial dysfunction, and reverses pulmonary hypertension. J. Clin. Invest. 123(8), 3600–3613 (2013).
Diebold, I. et al. BMPR2 preserves mitochondrial function and DNA during reoxygenation to promote endothelial cell survival and reverse pulmonary hypertension. Cell Metab. 21(4), 596–608 (2015).
Yang, X., Long, L., Reynolds, P. N. & Morrell, N. W. Expression of mutant BMPR-II in pulmonary endothelial cells promotes apoptosis and a release of factors that stimulate proliferation of pulmonary arterial smooth muscle cells. Pulm. Circ. 1(1), 103–110 (2012).
Chelladurai, P. et al. Isoform-specific characterization of class I histone deacetylases and their therapeutic modulation in pulmonary hypertension. Sci. Rep. 10(1), 12864 (2020).
Schupp, J. C. et al. Integrated single cell atlas of endothelial cells of the human lung. Circul. Internet https://doi.org/10.1161/CIRCULATIONAHA.120.052318 (2021).
Ferrian, S. et al. Single-cell imaging maps inflammatory cell subsets to pulmonary arterial hypertension vasculopathy. Am. J. Respir. Crit. Care Med. 209(2), 206–218 (2024).
Arroyo, J. D. et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. U S A. 108(12), 5003–5008 (2011).
Acknowledgements
We thank all the patients that participated in this study and all the clinical personnel implied in sample retrieval and the IDIBAPS Biobank. We thank the REHAP investigators for their effort to recruit and manage the patients. We thank Fundación Contra la Hipertensión Pulmonar for their continues support and effort. We thank Laura Muinelo Romay for providing us with the controls for the discovery cohort. We thank Merce Peleteiro from the Cytometry core facility of CINBIO for her technical help. In loving memory of Guillermo Pousada.
Funding
This study was partially funded by grants from Fundación Contra la Hipertensión Pulmonar, Jansen Pharmaceuticals, Ministerio de Ciencia e Innovación (PI18/01233, PI24/01182, Xunta de Galicia Axudas para consolidación e estructuración de unidades de investigación competitivas e outras accións de fomento (GRC-ED431C 2022/26) to DV. MLD was supported by a Xunta de Galicia predoctoral fellowship (ED481A-2018/304). AIG was supported by a collaboration grant from Ministerio de Educación y Formación Profesional 2020–21. This research was funded by the AGAUR Catalan Agency, grant number 2021_SGR_01493.
Author information
Authors and Affiliations
Contributions
MLD and DV designed and conceptualized the study. MLD performed all the experimental and analysis work. AIG assisted in the validation cohort analyses. MLD wrote the manuscript. AB, CV, JAB and IB collected samples and retrieved clinical history. DV supervised the study. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
The study was conducted with the approval of the Comité de ética da Investigación de Galicia (CEIC Galicia). Written informed consent was obtained from all patients and control participants included in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lago-Docampo, M., Iglesias-López, A., Vilariño, C. et al. A circulating MicroRNA signature for the diagnosis of pulmonary arterial hypertension and functional characterization of candidate miR-3168. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42550-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42550-8