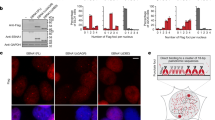
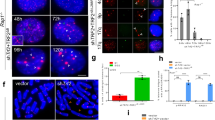
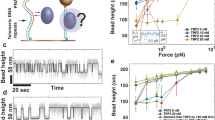
Abstract
Epstein-Barr Nuclear Antigen 1 (EBNA1) is essential for the episomal maintenance and DNA replication of Epstein-Barr virus (EBV) in latently infected cells and acts through binding to oriP. The minimal replicative unit of oriP (½DS) contains four EBNA1 binding sites flanked by single telomeric nonamers that recruit shelterin proteins TRF2 and Rap1, but the structural basis for host-factor engagement is not known. Here, we integrate cryo-electron microscopy, zero-length cross-linking mass spectrometry, Alphafold3 modeling, and biochemical binding assays to define the complex formed by EBNA1-TRF2-Rap1 assembly on the ½DS. We find that a highly dynamic complex is formed, with the TRF2 homodimerization domain (TRFH) flexibly interacting with EBNA1 on the surface opposite the DNA-binding region, where there is a large acidic patch in EBNA1 that is unique amongst the herpesvirus episome maintenance proteins. Mutagenesis of this acidic patch abolishes TRFH binding and oriP-dependent plasmid replication. These findings identify a previously uncharacterized acidic patch docking surface on EBNA1 essential for coordinating TRF2-Rap1 at oriP and provide new insights into both EBV and telomere DNA replication.
Similar content being viewed by others
Data availability
The datasets generated during the current study are available in the Supplementary Materials and at https://www.ebi.ac.uk/emdb/EMD-73305 for Cryo-EM data and at https://proteomecentral.proteomexchange.org/cgi/GetDataset? ID=PXD068321.
References
Damania, B., Kenney, S. C. & Raab-Traub, N. Epstein-Barr virus: Biology and clinical disease. Cell 185, 3652–3670. https://doi.org/10.1016/j.cell.2022.08.026 (2022).
Young, L. S., Yap, L. F. & Murray, P. G. Epstein-Barr virus: more than 50 years old and still providing surprises. Nat. Rev. Cancer. 16, 789–802. https://doi.org/10.1038/nrc.2016.92 (2016).
Wilson, J. B. et al. EBNA1: Oncogenic activity, immune evasion and biochemical functions provide targets for novel therapeutic strategies against Epstein-Barr virus-associated cancers. Cancers (Basel) https://doi.org/10.3390/cancers10040109 (2018).
Frappier, L. EBNA1. Curr. Top. Microbiol. Immunol. https://doi.org/10.1007/82_2025_299 (2025).
Chaudhuri, B., Xu, H., Todorov, I., Dutta, A. & Yates, J. L. Human DNA replication initiation factors, ORC and MCM, associate with oriP of Epstein-Barr virus. Proc. Natl. Acad. Sci. U. S. A. 98, 10085–10089. https://doi.org/10.1073/pnas.181347998 (2001).
Dhar, S. K. et al. Replication from oriP of Epstein-Barr virus requires human ORC and is inhibited by geminin. Cell 106, 287–296. https://doi.org/10.1016/s0092-8674(01)00458-5 (2001).
Hung, S. C., Kang, M. S. & Kieff, E. Maintenance of Epstein-Barr virus (EBV) oriP-based episomes requires EBV-encoded nuclear antigen-1 chromosome-binding domains, which can be replaced by high-mobility group-I or histone H1. Proc. Natl. Acad. Sci. U. S. A. 98, 1865–1870. https://doi.org/10.1073/pnas.98.4.1865 (2001).
Marechal, V. et al. Mapping EBNA-1 domains involved in binding to metaphase chromosomes. J. Virol. 73, 4385–4392. https://doi.org/10.1128/JVI.73.5.4385-4392.1999 (1999).
Kanda, T. et al. Interaction between basic residues of Epstein-Barr virus EBNA1 protein and cellular chromatin mediates viral plasmid maintenance. J. Biol. Chem. 288, 24189–24199. https://doi.org/10.1074/jbc.M113.491167 (2013).
Tempera, I. et al. Identification of MEF2B, EBF1, and IL6R as direct gene targets of Epstein-Barr Virus (EBV) nuclear antigen 1 critical for EBV-infected B-Lymphocyte survival. J. Virol. 90, 345–355. https://doi.org/10.1128/jvi.02318-15 (2016).
Lu, F., Tempera, I., Lee, H. T., Dewispelaere, K. & Lieberman, P. M. EBNA1 binding and epigenetic regulation of Gastrokine tumor suppressor genes in gastric carcinoma cells. Virol. J. 11, 12. https://doi.org/10.1186/1743-422x-11-12 (2014).
Nanbo, A., Sugden, A. & Sugden, B. The coupling of synthesis and partitioning of EBV’s plasmid replicon is revealed in live cells. EMBO J. 26, 4252–4262. https://doi.org/10.1038/sj.emboj.7601853 (2007).
Ritzi, M. et al. Complex protein-DNA dynamics at the latent origin of DNA replication of Epstein-Barr virus. J. Cell Sci. 116, 3971–3984. https://doi.org/10.1242/jcs.00708 (2003).
Hirai, K. & Shirakata, M. Replication licensing of the EBV oriP minichromosome. Curr. Top. Microbiol. Immunol. 258, 13–33. https://doi.org/10.1007/978-3-642-56515-1_2 (2001).
Shirakata, M., Imadome, K. I. & Hirai, K. Requirement of replication licensing for the dyad symmetry element-dependent replication of the Epstein-Barr virus oriP minichromosome. Virology 263, 42–54. https://doi.org/10.1006/viro.1999.9965 (1999).
Deng, Z., Atanasiu, C., Burg, J. S., Broccoli, D. & Lieberman, P. M. Telomere repeat binding factors TRF1, TRF2, and hRAP1 modulate replication of Epstein-Barr virus OriP. J. Virol. 77, 11992–12001. https://doi.org/10.1128/jvi.77.22.11992-12001.2003 (2003).
Deng, Z. et al. Telomeric proteins regulate episomal maintenance of Epstein-Barr virus origin of plasmid replication. Mol. Cell 9, 493–503. https://doi.org/10.1016/s1097-2765(02)00476-8 (2002).
Zhou, J. et al. Cell cycle regulation of chromatin at an origin of DNA replication. Embo j. 24, 1406–1417. https://doi.org/10.1038/sj.emboj.7600609 (2005).
Atanasiu, C., Deng, Z., Wiedmer, A., Norseen, J. & Lieberman, P. M. ORC binding to TRF2 stimulates OriP replication. EMBO Rep. 7, 716–721. https://doi.org/10.1038/sj.embor.7400730 (2006).
Deng, Z., Dheekollu, J., Broccoli, D., Dutta, A. & Lieberman, P. M. The origin recognition complex localizes to telomere repeats and prevents telomere-circle formation. Curr. Biol. 17, 1989–1995. https://doi.org/10.1016/j.cub.2007.10.054 (2007).
Tatsumi, Y. et al. Involvement of human ORC and TRF2 in pre-replication complex assembly at telomeres. Genes Cells. 13, 1045–1059. https://doi.org/10.1111/j.1365-2443.2008.01224.x (2008).
Deng, Z., Norseen, J., Wiedmer, A., Riethman, H. & Lieberman, P. M. TERRA RNA binding to TRF2 facilitates heterochromatin formation and ORC recruitment at telomeres. Mol. Cell. 35, 403–413. https://doi.org/10.1016/j.molcel.2009.06.025 (2009).
Higa, M. et al. TRF2-mediated ORC recruitment underlies telomere stability upon DNA replication stress. Nucleic Acids Res. 49, 12234–12251. https://doi.org/10.1093/nar/gkab1004 (2021).
Drosopoulos, W. C. et al. TRF2 mediates replication initiation within human telomeres to prevent telomere dysfunction. Cell. Rep. 33, 108379. https://doi.org/10.1016/j.celrep.2020.108379 (2020).
Bochkarev, A., Bochkareva, E., Frappier, L. & Edwards, A. M. The 2.2 A structure of a permanganate-sensitive DNA site bound by the Epstein-Barr virus origin binding protein, EBNA1. J. Mol. Biol. 284, 1273–1278. https://doi.org/10.1006/jmbi.1998.2247 (1998).
Malecka, K. A. et al. Structural basis for cooperative binding of EBNA1 to the Epstein-Barr virus dyad symmetry minimal origin of replication. J. Virol. https://doi.org/10.1128/jvi.00487-19 (2019).
Mei, Y. et al. Cryo-EM structure and functional studies of EBNA1 binding to the family of repeats and dyad symmetry elements of Epstein-Barr virus oriP. J. Virol. 96, e0094922. https://doi.org/10.1128/jvi.00949-22 (2022).
Janoušková, E. et al. Human Rap1 modulates TRF2 attraction to telomeric DNA. Nucleic Acids Res. 43, 2691–2700. https://doi.org/10.1093/nar/gkv097 (2015).
Kastner, B. et al. GraFix: sample preparation for single-particle electron cryomicroscopy. Nat. Methods. 5, 53–55. https://doi.org/10.1038/nmeth1139 (2008).
Young, G. et al. Quantitative mass imaging of single biological macromolecules. Science 360, 423–427. https://doi.org/10.1126/science.aar5839 (2018).
Hellert, J. et al. The 3D structure of Kaposi sarcoma herpesvirus LANA C-terminal domain bound to DNA. Proc. Natl. Acad. Sci. U. S. A. 112, 6694–6699. https://doi.org/10.1073/pnas.1421804112 (2015).
Kim, S. S., Tam, J. K., Wang, A. F. & Hegde, R. S. The structural basis of DNA target discrimination by papillomavirus E2 proteins. J. Biol. Chem. 275, 31245–31254. https://doi.org/10.1074/jbc.M004541200 (2000).
Hellert, J. et al. A structural basis for BRD2/4-mediated host chromatin interaction and oligomer assembly of Kaposi sarcoma-associated herpesvirus and murine gammaherpesvirus LANA proteins. PLoS Pathog. 9, e1003640. https://doi.org/10.1371/journal.ppat.1003640 (2013).
Domsic, J. F., Chen, H. S., Lu, F., Marmorstein, R. & Lieberman, P. M. Molecular basis for oligomeric-DNA binding and episome maintenance by KSHV LANA. PLoS Pathog. 9, e1003672. https://doi.org/10.1371/journal.ppat.1003672 (2013).
Lin, A., Wang, S., Nguyen, T., Shire, K. & Frappier, L. The EBNA1 protein of Epstein-Barr virus functionally interacts with Brd4. J. Virol. 82, 12009–12019. https://doi.org/10.1128/jvi.01680-08 (2008).
Baxter, M. K., McPhillips, M. G., Ozato, K. & McBride, A. A. The mitotic chromosome binding activity of the papillomavirus E2 protein correlates with interaction with the cellular chromosomal protein, Brd4. J. Virol. 79, 4806–4818. https://doi.org/10.1128/JVI.79.8.4806-4818.2005 (2005).
You, J., Croyle, J. L., Nishimura, A., Ozato, K. & Howley, P. M. Interaction of the bovine papillomavirus E2 protein with Brd4 tethers the viral DNA to host mitotic chromosomes. Cell 117, 349–360. https://doi.org/10.1016/s0092-8674(04)00402-7 (2004).
Abbate, E. A., Voitenleitner, C. & Botchan, M. R. Structure of the papillomavirus DNA-tethering complex E2:Brd4 and a peptide that ablates HPV chromosomal association. Mol. Cell 24, 877–889. https://doi.org/10.1016/j.molcel.2006.11.002 (2006).
Saridakis, V. et al. Structure of the p53 binding domain of HAUSP/USP7 bound to Epstein-Barr nuclear antigen 1 implications for EBV-mediated immortalization. Mol. Cell 18, 25–36. https://doi.org/10.1016/j.molcel.2005.02.029 (2005).
Cao, J. Y. et al. Identification of a novel protein interaction motif in the regulatory subunit of casein kinase 2. Mol. Cell. Biol. 34, 246–258. https://doi.org/10.1128/mcb.00968-13 (2014).
Sivachandran, N., Cao, J. Y. & Frappier, L. Epstein-Barr virus nuclear antigen 1 hijacks the host kinase CK2 to disrupt PML nuclear bodies. J. Virol. 84, 11113–11123. https://doi.org/10.1128/jvi.01183-10 (2010).
Nakada, R., Hirano, H. & Matsuura, Y. Structural basis for the regulation of nuclear import of Epstein-Barr virus nuclear antigen 1 (EBNA1) by phosphorylation of the nuclear localization signal. Biochem. Biophys. Res. Commun. 484, 113–117. https://doi.org/10.1016/j.bbrc.2017.01.063 (2017).
Kim, A. L. et al. An imperfect correlation between DNA replication activity of Epstein-Barr virus nuclear antigen 1 (EBNA1) and binding to the nuclear import receptor, Rch1/importin alpha. Virology 239, 340–351. https://doi.org/10.1006/viro.1997.8874 (1997).
Zhou, J., Deng, Z., Norseen, J. & Lieberman, P. M. Regulation of Epstein-Barr virus origin of plasmid replication (OriP) by the s-phase checkpoint kinase Chk2. J. Virol. 84, 4979–4987. https://doi.org/10.1128/jvi.01300-09 (2010).
Deng, Z. et al. Inhibition of Epstein-Barr virus OriP function by tankyrase, a telomere-associated poly-ADP ribose polymerase that binds and modifies EBNA1. J. Virol. 79, 4640–4650. https://doi.org/10.1128/jvi.79.8.4640-4650.2005 (2005).
Janovič, T., Perez, G. I. & Schmidt, J. C. TRF1 and TRF2 form distinct shelterin subcomplexes at telomeres. bioRxiv https://doi.org/10.1101/2024.12.23.630076 (2024).
Zinder, J. C. et al. Shelterin is a dimeric complex with extensive structural heterogeneity. Proc. Natl. Acad. Sci. U. S. A. 119, e2201662119. https://doi.org/10.1073/pnas.2201662119 (2022).
Chambers, M. C. et al. A cross-platform toolkit for mass spectrometry and proteomics. Nat. Biotechnol. 30, 918–920. https://doi.org/10.1038/nbt.2377 (2012).
Lenz, S. et al. Reliable identification of protein-protein interactions by crosslinking mass spectrometry. Nat. Commun. 12, 3564. https://doi.org/10.1038/s41467-021-23666-z (2021).
Mendes, M. L. et al. An integrated workflow for crosslinking mass spectrometry. Mol. Syst. Biol. 15, e8994. https://doi.org/10.15252/msb.20198994 (2019).
Combe, C. W., Graham, M., Kolbowski, L., Fischer, L. & Rappsilber, J. xiVIEW: Visualisation of crosslinking mass spectrometry data. J. Mol. Biol. 436, 168656. https://doi.org/10.1016/j.jmb.2024.168656 (2024).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: Algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296. https://doi.org/10.1038/nmeth.4169 (2017).
Chen, C. et al. USP7 inhibitors destabilize EBNA1 and suppress Epstein-Barr Virus tumorigenesis. J. Med. Virol. 97, e70168. https://doi.org/10.1002/jmv.70168 (2025).
Dheekollu, J., Ayyanathan, W. A., Deakyne, K., Messick, J. S. & Lieberman, T. E. Cell-cycle-dependent EBNA1-DNA crosslinking promotes replication termination at oriP and viral episome maintenance. Cell 184, 643–654. https://doi.org/10.1016/j.cell.2020.12.022 (2021).
Acknowledgements
We thank members of the Electron Microscopy Resource Lab (EMRL) and the Beckman Center for Cryo-EM at the University of Pennsylvania for cryo-EM screening and grid clipping. We thank the University of Pennsylvania institute for Structural Biology (ISB) and Jesper Pallesen at the Wistar Institute for advice on cryo-EM methodology and analyses. This research was, in part, supported by the National Cancer Institute’s National Cryo-EM Facility at the Frederick National Laboratory for Cancer Research under contract 75N91019D00024. This work was supported by grants from the NIH R01 CA140652 and R01 CA093606 to PML, R01 CA259171 to TEM, and NIH R01GM123233 to KM. This work was also supported by an Institutional Core grant to the Wistar Institute Cancer Center P30 CA010815.
Funding
This research was, in part, supported by the National Cancer Institute’s National Cryo-EM Facility at the Frederick National Laboratory for Cancer Research under contract 75N91019D00024. This work was supported by grants from the NIH R01 CA140652 and R01 CA093606 to PML, R01 CA259171 to TEM, and NIH R01GM123233 to KM. This work was also supported by an Institutional Core grant to the Wistar Institute Cancer Center P30 CA010815.
Author information
Authors and Affiliations
Contributions
SS wrote the main manuscript text, prepared figures, and performed all EMSA experiments, mass photometry, cryo-EM sample preparation and data analysis, and Alphafold3 modeling. TM expressed and purified proteins, assisted with all structural work, and assisted with manuscript edits. JD and CA performed replication assay experiments. CC performed cyclohexamide chase experiments. AF and HYT performed crosslinking mass spectrometry experiments. HJK and KM assisted with cryo-EM data analysis and provided resources. PML supervised the project and assisted with manuscript edits and provided resources. All authors discussed the results and contributed to the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
PML is a founder of Vironika LLC and is listed on patents relating to EBNA1 inhibitors. PML has served on advisory panels for Pfizer, GSK, Sanofi, Merck, and Moderna.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sustek, S., Messick, T.E., Dheekollu, J. et al. Structural basis for TRF2-RAP1 recruitment by EBNA1 at the EBV origin of replication. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43067-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43067-w