Abstract
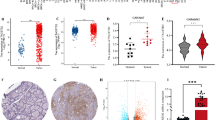
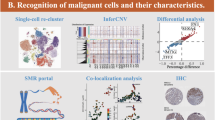
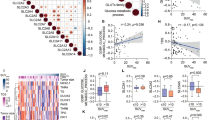
This study aims to investigate the role of DUSP6 in thyroid cancer metastasis and elucidate its underlying molecular mechanisms. Immunohistochemistry were performed to explore the expression of DUSP6, IL-8 and PAD4 in papillary thyroid carcinoma (PTC) tissues and adjacent non-cancerous thyroid tissues. Effects of DUSP6 on the proliferation, apoptosis, migration, and invasion of thyroid cancer cell lines TPC-1 and IHH4 were performed through in vitro experiments. A rescue experiment was performed to verified DUSP6 regulated the migration and invasion of thyroid cancer cells TPC-1 and IHH4 through IL-8 and neutrophil extracellular traps (NETs) formation. In addition, in vitro cell experiments were conducted to analyze the regulation of NETs formation by DUSP6 through IL-8. Finally, the effect of sh-DUSP6 on the growth of thyroid cancer tumors in mice were conducted through in vivo experiments. DUSP6 expression was significantly upregulated in PTC tissues. Moreover, the expression of DUSP6 in tumor tissues of TPC patients is positively correlated with the expression of IL-8 and PAD4. Overexpression of DUSP6 promotes the proliferation, migration, and invasion of thyroid cancer cells (TPC-1 and IHH4) while inhibiting apoptosis. Conversely, silencing DUSP6 exerts opposing effects. Knockdown of IL-8 reverses the enhanced migratory and invasive capabilities induced by DUSP6 overexpression in these cell lines. NETs treatment enhances the migration and invasion of TPC-1 and IHH4 cells, whereas the application of sh-DUSP6 or sh-IL-8 counteracts this NETs-mediated promotion. Furthermore, DUSP6 overexpression facilitates NETs formation by upregulating IL-8 expression. In vivo experiments demonstrate that sh-DUSP6 suppresses thyroid cancer tumor growth in mouse models. Conclusion: DUSP6 promotes the metastasis of thyroid cancer by facilitating the formation of neutrophil extracellular traps via IL-8.
Similar content being viewed by others
Data availability
All data presented in the study were included in the article. Further inquiries can be directed to the first author.
References
Forma, A. et al. Thyroid cancer: Epidemiology, classification, risk factors, diagnostic and prognostic markers, and current treatment strategies. Int. J. Mol. Sci. 26(11), 5173. https://doi.org/10.3390/ijms26115173 (2025).
Franchini, F. et al. Obesity and thyroid cancer risk: An update. Int. J. Environ. Res. Public Health 19(3), 1116. https://doi.org/10.3390/ijerph19031116 (2022).
Ye, H. et al. The emerging roles of circRNAs in papillary thyroid carcinoma: Molecular mechanisms and biomarker potential. Protein & Peptide Letters 30(9), 709–718. https://doi.org/10.2174/0929866530666230804104057 (2023).
Feng, Y. et al. A bibliometric analysis of inflammation and thyroid carcinoma: Research trends and future perspectives. Front. Oncol. 15, 1593006. https://doi.org/10.3389/fonc.2025.1593006 (2025).
Wang, H. et al. Knockdown of programmed death 1 inhibited progression of papillary thyroid carcinoma in mice. Protein & Peptide Letters 30(5), 396–400. https://doi.org/10.2174/0929866530666230306112912 (2023).
Volpe, F. et al. Current advances in radioactive iodine-refractory differentiated thyroid cancer. Curr Oncol 31(7), 3870–3884. https://doi.org/10.3390/curroncol31070286 (2024).
Wan, Y. et al. Reprogramming of thyroid cancer metabolism: From mechanism to therapeutic strategy. Mol Cancer 24(1), 74. https://doi.org/10.1186/s12943-025-02263-4 (2025).
Chen, C. & Shen, Z. FN1 promotes thyroid carcinoma cell proliferation and metastasis by activating the NF-Κb pathway. Protein Pept. Lett. 30(1), 54–64. https://doi.org/10.2174/0929866530666221019162943 (2023).
Kong, T. et al. DUSP6 mediates resistance to JAK2 inhibition and drives leukemic progression. Nat. Cancer. 4 (1), 108–127. https://doi.org/10.1038/s43018-022-00486-8 (2023).
Laragione, T. et al. DUSP6 deletion protects mice and reduces disease severity in autoimmune arthritis. iScience 27(6), 110158. https://doi.org/10.1016/j.isci.2024.110158 (2024).
Png, C. W. et al. DUSP6 regulates Notch1 signalling in colorectal cancer. Nat. Commun. 15(1), 10087. https://doi.org/10.1038/s41467-024-54383-y (2024).
Degl’Innocenti, D. et al. DUSP6/MKP3 is overexpressed in papillary and poorly differentiated thyroid carcinoma and contributes to neoplastic properties of thyroid cancer cells. Endocr. Relat. Cancer. 20(1), 23–37. https://doi.org/10.1530/ERC-12-0078 (2013).
Lee, J. U. et al. Dual specificity phosphatase 6 as a predictor of invasiveness in papillary thyroid cancer. Eur. J. Endocrinol. 167(1), 93–101. https://doi.org/10.1530/EJE-12-0010 (2012).
Gu, Y. et al. MicroRNA-145 inhibits human papillary cancer TPC1 cell proliferation by targeting DUSP6. Int. J. Clin. Exp. Med. 8 (6), 8590–8598 (2015).
Zhai, X. et al. Dual specificity phosphatase 6 suppresses the growth and metastasis of prostate cancer cells. Mol. Med. Rep. 10(6), 3052–8. https://doi.org/10.3892/mmr.2014.2575 (2014).
Zhang, J. et al. Targeting neutrophil extracellular traps in cancer progression and metastasis. Theranostics 15(12), 5846–5869. https://doi.org/10.7150/thno.111096 (2025).
An, Z. et al. Neutrophil extracellular traps induced by IL-8 aggravate atherosclerosis via activation NF-κB signaling in macrophages. Cell. Cycle. 18 (21), 2928–2938. https://doi.org/10.1080/15384101.2019.1662678 (2019).
Shu, Q. et al. IL-8 triggers neutrophil extracellular trap formation through an nicotinamide adenine dinucleotide phosphate oxidase- and mitogen-activated protein kinase pathway-dependent mechanism in uveitis. Invest. Ophthalmol. Vis. Sci. 64(13), 19. https://doi.org/10.1167/iovs.64.13.19 (2023).
Wang, Y. et al. Neutrophil extracellular traps (NETs) promote non-small cell lung cancer metastasis by suppressing lncRNA MIR503HG to activate the NF-κB/NLRP3 inflammasome pathway. Front Immunol 13, 867516. https://doi.org/10.3389/fimmu.2022.867516 (2022).
Chu, C. et al. Neutrophil extracellular traps drive intestinal microvascular endothelial ferroptosis by impairing Fundc1-dependent mitophagy. Redox Biol. 67, 102906. https://doi.org/10.1016/j.redox.2023.102906 (2023).
Tang, J. et al. IL33-induced neutrophil extracellular traps (NETs) mediate a positive feedback loop for synovial inflammation and NET amplification in rheumatoid arthritis. Exp. Mol. Med. 56(12), 2602–2616. https://doi.org/10.1038/s12276-024-01351-7 (2024).
Tai, C. H. et al. Functional Role of DUSP6 in the EGFR-mediated Progression of Head and Neck Squamous Cell Carcinoma. Anticancer Res. 44 (6), 2545–2554. https://doi.org/10.21873/anticanres.17060 (2024).
Kim, Y. H. et al. TSH signaling overcomes B-RafV600E-induced senescence in papillary thyroid carcinogenesis through regulation of DUSP6. Neoplasia 16(12), 1107–20. https://doi.org/10.1016/j.neo.2014.10.005 (2014).
Shen, Y. et al. CircPDIA4 induces gastric cancer progression by promoting ERK1/2 activation and enhancing biogenesis of oncogenic circRNAs. Cancer Res. 83(4), 538–552. https://doi.org/10.1158/0008-5472.CAN-22-1923 (2023).
Gao, Y. et al. Overexpression of DUSP6 enhances chemotherapy-resistance of ovarian epithelial cancer by regulating the ERK signaling pathway. J. Cancer 11(11), 3151–3164. https://doi.org/10.7150/jca.37267 (2020).
Momeny, M. et al. DUSP6 inhibition overcomes neuregulin/HER3-driven therapy tolerance in HER2 + breast cancer. EMBO Mol. Med. 16(7), 1603–1629. https://doi.org/10.1038/s44321-024-00088-0 (2024).
Chen, D. et al. Unveiling the functional roles of patient-derived tumour organoids in assessing the tumour microenvironment and immunotherapy. Clin. Transl. Med. 14(9), e1802. https://doi.org/10.1002/ctm2.1802 (2024).
Li, X. et al. Tumor-derived IL-8 facilitates lymph node metastasis of gastric cancer via PD-1 up-regulation in CD8 + T cells. Cancer Immunol. Immunother. 71(12), 3057–3070. https://doi.org/10.1007/s00262-022-03223-3 (2022).
Lin, L. et al. Overexpression of IL-8 and Wnt2 is associated with prognosis of gastric cancer. Folia Histochem. Cytobiol. 60(1), 66–73. https://doi.org/10.5603/FHC.a2022.0002 (2022).
Matsushima, K., Yang, D. & Oppenheim, J. J. Interleukin-8: An evolving chemokine. Cytokine 153, 155828. https://doi.org/10.1016/j.cyto.2022.155828 (2022).
Vilotić, A. et al. IL-6 and IL-8: An overview of their roles in healthy and pathological pregnancies. Int. J. Mol. Sci. 23(23), 14574. https://doi.org/10.3390/ijms232314574 (2022).
Peng, S. et al. Hsa-microRNA-370-3p targeting Snail and Twist1 suppresses IL-8/STAT3-driven hepatocellular carcinoma metastasis. Cancer Sci. 113(12), 4120–4134. https://doi.org/10.1111/cas.15571 (2022).
Principe, S. et al. Salivary IL-8 as a putative predictive biomarker of radiotherapy response in head and neck cancer patients. Clin. Oral Investig. 26(1), 437–448. https://doi.org/10.1007/s00784-021-04017-0 (2022).
Fang, W. et al. Tumor-associated macrophages promote the metastatic potential of thyroid papillary cancer by releasing CXCL8. Carcinogenesis 35(8), 1780–7. https://doi.org/10.1093/carcin/bgu060 (2014).
Ostadkarampour, M. & Putnins, E. E. A monoamine oxidase B inhibitor altered gene expression of catalytically active dual-specificity phosphatases in human oral gingival keratinocytes. Eur. Rev. Med. Pharmacol. Sci. 28 (24), 4681–4690. https://doi.org/10.26355/eurrev_202412_37002 (2024).
Leng, Y. P. et al. l-Homocysteine-induced cathepsin V mediates the vascular endothelial inflammation in hyperhomocysteinaemia. Br. J. Pharmacol. 175(8), 1157–1172. https://doi.org/10.1111/bph.13920 (2018).
Masucci, M. T. et al. The emerging role of neutrophil extracellular traps (NETs) in tumor progression and metastasis. Front. Immunol. 11, 1749. https://doi.org/10.3389/fimmu.2020.01749 (2020).
Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 18(2), 134–147. https://doi.org/10.1038/nri.2017.105 (2018).
Zhang, H. et al. Neutrophil, neutrophil extracellular traps and endothelial cell dysfunction in sepsis. Clin Transl Med 13(1), e1170. https://doi.org/10.1002/ctm2.1170 (2023).
Zeng, J. et al. Kaempferol blocks neutrophil extracellular traps formation and reduces tumour metastasis by inhibiting ROS-PAD4 pathway. J Cell Mol Med 24(13), 7590–7599. https://doi.org/10.1111/jcmm.15394 (2020).
Modestino, L. et al. Neutrophil extracellular traps and neutrophil-related mediators in human thyroid cancer. Front. Immunol. 14, 1167404. https://doi.org/10.3389/fimmu.2023.1167404 (2023).
Author information
Authors and Affiliations
Contributions
WX drafted the manuscript and conducted the experiments. LL, YY and WC conducted the experiments and performed data analysis. CY designed the experiments and revised the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This article was approved by the Ethics Committee of Jiangxi Provincial People’s Hospital (Clinical approval number: KEKUAI2025(62)). The patients/participants provided their written informed consent to participate in this study. Besides, animals were housed and operated in strict compliance with the ethical principles of animal experimentation, and the operations were ratified by the animal ethics committee of Jiangxi Provincial People’s Hospital (approval number: NYLLSC20250922). All animal experiments were in accordance with ARRIVE guidelines and complied with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xiao, W., Luo, L., Yang, Y. et al. DUSP6 promotes motility, invasion, and tumorigenicity of thyroid cancer cells via IL8-induced neutrophil extracellular traps. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43233-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43233-0