Abstract
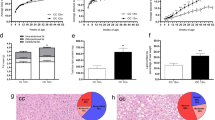
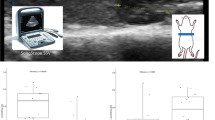
Lipid metabolism disorders and hepatic lipid deposition are major contributors to the development of metabolic diseases such as non-alcoholic fatty liver disease. Omega-3 fatty acids have been shown to exert beneficial effects on lipid homeostasis and liver function, but their molecular regulatory mechanisms remain incompletely understood. Here, we investigated the effects of omega-3 fatty acids on lipid metabolism and hepatic lipid deposition in addition to the modulatory role of miR-34a using an in vivo high-fat diet (HFD) model and an in vitro palmitic acid (PA)-induced HepG2 cell model. We assessed the effects of omega-3 fatty acid intervention and measured lipid levels in mouse serum, triglyceride (TG) and total cholesterol (TC) levels in the mouse liver, and TG levels in HepG2 cells. Further, we evaluated the mRNA and protein levels of miR-34a, SIRT1, SREBP-1c, CPT-1 A and PGC-1α. We observed that miR-34a expression was upregulated in the HFD and PA groups. Omega-3 fatty acid intervention reduced the serum levels of TG, TC, and low-density lipoproteins(LDL-C), and decreased the levels of TG and TC in the liver tissue, alleviating hepatocyte steatosis. Further, omega-3 fatty acids intervention reduced miR-34a and SREBP-1c expression, and increased the expression of SIRT1,PGC-1α, CPT-1 A. Moreover, miR-34a overexpression inhibited the effects of omega-3 fatty acids on SIRT1, SREBP-1c, PGC-1α and CPT-1 A. Thus, omega-3 fatty acids promote lipid metabolism, thereby reducing hepatic lipid deposition by down-regulating miR-34a expression.
Similar content being viewed by others
Data availability
The datasets used and analysed during the current study available from the corresponding author on reasonable request.
References
Shapiro, H. et al. The therapeutic potential of long-chain omega-3 fatty acids in nonalcoholic fatty liver disease. Clin. Nutr. 30(1), 6–19. https://doi.org/10.1016/j.clnu.2010.06.001 (2011).
Hong, L., Zahradka, P., Cordero-Monroy, L., Wright, B. & Taylor, C. G. Dietary docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) operate by different mechanisms to modulate hepatic steatosis and hyperinsulemia in fa/fa Zucker rats. Nutrients 11(4), 917. https://doi.org/10.3390/nu11040917 (2019).
Klop, B., Elte, J. W. & Cabezas, M. C. Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 5(4), 1218–40. https://doi.org/10.3390/nu5041218 (2013).
Kimura, M., Kothari, S., Gohir, W., Camargo, J. F. & Husain, S. MicroRNAs in infectious diseases: Potential diagnostic biomarkers and therapeutic targets. Clin. Microbiol. Rev. 36(4), e0001523. https://doi.org/10.1128/cmr.00015-23 (2023).
Zhang, X., Price, N. L. & Fernández-Hernando, C. Non-coding RNAs in lipid metabolism. Vascul. Pharmacol. 114, 93–102. https://doi.org/10.1016/j.vph.2018.06.011 (2019).
Zhang, Z., Moon, R., Thorne, J. L. & Moore, J. B. NAFLD and vitamin D: Evidence for intersection of microRNA-regulated pathways. Nutr. Res. Rev. 36(1), 120–139. https://doi.org/10.1017/S095442242100038X (2023).
Guo, Y. et al. Total flavonoids from the Carya cathayensis Sarg. leaves inhibit HUVEC senescence through the miR‐34a/SIRT1 pathway. J. Cell. Biochem. 120(10), 17240–17249. https://doi.org/10.1002/jcb.28986 (2019).
Yang, J. et al. Buckwheat protein-derived peptide ameliorates insulin resistance by directing O-linked N-acetylglucosamine transferase to regulate the SIRT1/PGC1α pathway. Int. J. Biol. Macromol. 304(Pt 2), 140925. https://doi.org/10.1016/j.ijbiomac.2025.140925 (2025).
Xu, C. et al. Neuregulin1 ameliorates metabolic dysfunction-associated fatty liver disease via the ERK/SIRT1 signaling pathways. BMC Gastroenterol. 25(1), 47. https://doi.org/10.1186/s12876-025-03632-5 (2025).
Liang, M. et al. Artemisia capillaris Thunb. water extract alleviates metabolic dysfunction-associated steatotic liver disease disease by inhibiting miR-34aa-5p to activate Sirt1-mediated hepatic lipid metabolism. J. Ethnopharmacol. 338(Pt 2), 119030. https://doi.org/10.1016/j.jep.2024.119030 (2025).
Gatsiou, A. et al. Additive contribution of microRNA-34a/b/c to human arterial ageing and atherosclerosis. Atherosclerosis 327, 49–58. https://doi.org/10.1016/j.atherosclerosis.2021.05.005 (2021).
Tian, C., Huang, R. & Xiang, M. SIRT1: Harnessing multiple pathways to hinder NAFLD. Pharmacol. Res. 203, 107155. https://doi.org/10.1016/j.phrs.2024.107155 (2024).
Jin, M. et al. Dietary betaine mitigates hepatic steatosis and inflammation induced by a high-fat-diet by modulating the Sirt1/Srebp-1/Pparɑ pathway in juvenile black seabream (Acanthopagrus schlegelii). Front. Immunol. 12, 694720. https://doi.org/10.3389/fimmu.2021.694720 (2021).
Alshehri, A. S., El-Kott, A. F., El-Kenawy, A. E., Khalifa, H. S. & AlRamlawy, A. M. Cadmium chloride induces non-alcoholic fatty liver disease in rats by stimulating miR-34aa/SIRT1/FXR/p53 axis. Sci. Total Environ. 784, 147182. https://doi.org/10.1016/j.scitotenv.2021.147182 (2021).
Yang, Z. H., Miyahara, H., Takeo, J., Hatanaka, A. & Katayama, M. Pollock oil supplementation modulates hyperlipidemia and ameliorates hepatic steatosis in mice fed a high-fat diet. Lipids Health Dis. 10, 189. https://doi.org/10.1186/1476-511X-10-189 (2011).
Hao, L., Chen, C. Y., Nie, Y. H., Kaliannan, K. & Kang, J. X. Differential interventional effects of Omega-6 and Omega-3 polyunsaturated fatty acids on high fat diet-induced obesity and hepatic pathology. Int. J. Mol. Sci. 24(24), 17261. https://doi.org/10.3390/ijms242417261 (2023).
Wong, C. Y. et al. Fish-oil supplement has neutral effects on vascular and metabolic function but improves renal function in patients with type 2 diabetes mellitus. Diabet. Med. 27(1), 54–60. https://doi.org/10.1111/j.1464-5491.2009.02869.x (2010).
Lu, L. et al. Associations between omega-3 fatty acids and insulin resistance and body composition in women with polycystic ovary syndrome. Front Nutr. ;9:1016943. (2022). https://doi.org/10.3389/fnut.2022.1016943. Erratum in: Front Nutr. 2024;11:1383808. doi: 10.3389/fnut.2024.1383808.
Thota, R. N., Acharya, S. H. & Garg, M. L. Curcumin and/or omega-3 polyunsaturated fatty acids supplementation reduces insulin resistance and blood lipids in individuals with high risk of type 2 diabetes: A randomised controlled trial. Lipids Health Dis. 18(1), 31. https://doi.org/10.1186/s12944-019-0967-x (2019).
Capanni, M. et al. Prolonged n-3 polyunsaturated fatty acid supplementation ameliorates hepatic steatosis in patients with non-alcoholic fatty liver disease: A pilot study. Aliment. Pharmacol. Ther. 23(8), 1143–51. https://doi.org/10.1111/j.1365-2036.2006.02885.x (2006).
Hartweg, J., Farmer, A. J., Holman, R. R. & Neil, A. Potential impact of omega-3 treatment on cardiovascular disease in type 2 diabetes. Curr. Opin. Lipidol. 20 (1), 30–38. https://doi.org/10.1097/mol.0b013e328321b3be (2009).
Gallo Ruelas, M. et al. Effects of seal oil supplementation on lipid profile biomarkers: A systematic review and meta-analysis of randomized controlled trials. Prostaglandins Leukot. Essent. Fatty Acids 204, 102666. https://doi.org/10.1016/j.plefa.2025.102666 (2025).
Katare, P. B. et al. Krill oil supplementation in vivo promotes increased fuel metabolism and protein synthesis in cultured human skeletal muscle cells. Front. Nutr. 11, 1452768. https://doi.org/10.3389/fnut.2024.1452768 (2024).
Corral-Jara, K. F. et al. An integrated analysis of miRNA and gene expression changes in response to an obesogenic diet to explore the impact of transgenerational supplementation with omega 3 fatty acids. Nutrients 12(12), 3864. https://doi.org/10.3390/nu12123864 (2020).
Visioli, F., Giordano, E., Nicod, N. M. & Dávalos, A. Molecular targets of omega 3 and conjugated linoleic fatty acids - “Micromanaging” cellular response. Front. Physiol. 3, 42. https://doi.org/10.3389/fphys.2012.00042 (2012).
Mohamed, A. A. et al. NAFLD mark: An accurate model based on microRNA-34 for diagnosis of non-alcoholic fatty liver disease patients. J. Genet. Eng. Biotechnol. 19(1), 157. https://doi.org/10.1186/s43141-021-00257-5 (2021).
Dongiovanni, P., Meroni, M., Longo, M., Fargion, S. & Fracanzani, A. L. miRNA signature in NAFLD: A turning point for a non-invasive diagnosis. Int. J. Mol. Sci. 19(12), 3966. https://doi.org/10.3390/ijms19123966 (2018).
Zia, A. et al. A review study on the modulation of SIRT1 expression by miRNAs in aging and age-associated diseases. Int. J. Biol. Macromol. 188, 52–61. https://doi.org/10.1016/j.ijbiomac.2021.08.013 (2021).
Rezk, N. A., Lashin, M. B. & Sabbah, N. A. MiRNA 34-a regulate SIRT-1 and Foxo-1 expression in endometriosis. Non-coding RNA Res. 6(1), 35–41. https://doi.org/10.1016/j.ncrna.2021.02.002 (2021).
Peyman, M. et al. SIRT1 regulates hepatic vldlr levels. Cell Commun. Signal. 22(1), 297. https://doi.org/10.1186/s12964-024-01666-y (2024).
Wang, L. et al. miR-34aa regulates lipid metabolism by targeting SIRT1 in non-alcoholic fatty liver disease with iron overload. Arch. Biochem. Biophys. 695, 108642. https://doi.org/10.1016/j.abb.2020.108642 (2020).
Dong, Z., Xie, X., Sun, Y., Wu, H. & Dai, M. Paeonol prevents lipid metabolism dysfunction in palmitic acid-induced HepG2 injury through promoting SIRT1-FoxO1-ATG14-dependent autophagy. Eur. J. Pharmacol. 880, 173145. https://doi.org/10.1016/j.ejphar.2020.173145 (2020).
de Collin l’Hortet, A. et al. Generation of human fatty livers using custom-engineered induced pluripotent stem cells with modifiable SIRT1 metabolism. Cell Metab. 30(2), 385-401.e9. https://doi.org/10.1016/j.cmet.2019.06.017 (2019).
Yin, X., Liu, Z. & Wang, J. Tetrahydropalmatine ameliorates hepatic steatosis in nonalcoholic fatty liver disease by switching lipid metabolism via AMPK-SREBP-1c-Sirt1 signaling axis. Phytomedicine 119, 155005. https://doi.org/10.1016/j.phymed.2023.155005 (2023).
MacLeod, B. et al. Fads2 knockout mice reveal that ALA prevention of hepatic steatosis is dependent on delta-6 desaturase activity. J. Lipid Res. 65(10), 100642. https://doi.org/10.1016/j.jlr.2024.100642 (2024).
Kim, J. K. et al. Omega-3 polyunsaturated fatty acid and ursodeoxycholic acid have an additive effect in attenuating diet-induced nonalcoholic steatohepatitis in mice. Exp. Mol. Med. 46(12), e127. https://doi.org/10.1038/emm.2014.90 (2014).
Wang, P. et al. Berberine alleviates non-alcoholic hepatic steatosis partially by promoting SIRT1 deacetylation of CPT1A in mice. Gastroenterol. Rep. 11, goad032. https://doi.org/10.1093/gastro/goad032 (2023).
Acknowledgements
We deeply appreciate the instructors at the Clinical Medical Research Centre of Hebei General Hospital for their help with this study. We appreciate Editage (www.editage.cn) for English language editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This work was supported by the National Natural Science Foundation of China (82170878).
Author information
Authors and Affiliations
Contributions
Luxuan Li: Conceptualization, Methodology, Investigation, Data curation, Writing-original draft, Visualization. Yale Tang: Investigation, Data curation. Xiaoyu Wang: Methodology, Data curation. Chao Wang: Investigation, Methodology, Validation. Guangyao Song: Conceptualization, Resources, Writing-review & editing. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest exits in the submission of this manuscript, and manuscript is approved by all authors for publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, L., Tang, Y., Wang, X. et al. Omega-3 fatty acids improve lipid metabolism by regulating miR-34a. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43353-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43353-7