Abstract
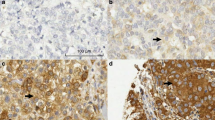
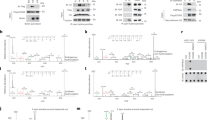
This study aims to explore the mechanism by which the Translationally controlled tumor protein (TCTP) promotes hyperplasia of the mammary glands (HMG). In this study, the TCTP (Tpt1flox/flox) and TCTP (Tpt1KI/KI) gene mice were respectively mated with MMTV-Cre to obtain mice withmammary glands-specific TCTP knockout (TCTP cKO) and overexpression (TCTP KI). Prepare the HMG animal model by using estrogen combined with progesterone. After the establishment of the TCTP cKO and TCTP KI mouse models, they are uniformly referred to as HMG cKO and HMG KI. Hematoxylin and Eosin (H&E) staining and hormone and receptor expression levels were detected. Detect the levels of TCTP, P53, p-AKT, as well as the indicators related to cell apoptosis and cell cycle in the breast tissues of each group. The H&E results showed that compared with the HMG group, the ductal cavity dilation and the number of milk glands in the breast tissue of the HMG cKO group were significantly reduced, while the HMG KI group exhibited obvious mammary gland hyperplasia. The results showed that compared with the HMG group, the expression levels of Ki67, E2, FSH, LH, ERα, PR, TCTP, p-AKT, p-BAD, Bcl-2, Cyclin D1, CDK4 and CDK6 in the mammary tissues of the HMG cKO group were significantly decreased, while the expression levels of PROG, ERβ, P53, Bax and P27 were significantly increased. In the HMG KI group, the opposite results were observed. In our research, it was confirmed that TCTP inhibits cell apoptosis and promotes cell cycle progression by regulating the AKT/P53 signaling pathway, leading to abnormal hyperplasia of the mammary glands. In HMG, by regulating the expression level of TCTP, the effect of treating HMG can be achieved.
Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study available from the corresponding author (Jun-Fei Zhang E-mail: zhangjunfei007@126.com) on reasonable request.
Abbreviations
- TCTP:
-
Translationally controlled tumor protein
- HMG:
-
Hyperplasia of the mammary glands
- H&E:
-
Directory of open access journals
- E2:
-
Estradiol
- LH:
-
Luteinizing hormone
- FSH:
-
Follicle-stimulating hormone
- PROG:
-
Progesterone
- IHC:
-
Immunohistochemistry
- mIHC:
-
Multiplex Immunofluorescence
References
Ma, W. et al. Clinical practice guideline for diagnosis and treatment of hyperplasia of the mammary glands: Chinese Society of Breast Surgery (CSBrS) practice guideline 2021. Chin. Med. J. (Engl). 134, 1891–1893. https://doi.org/10.1097/CM9.0000000000001521 (2021).
Xujie, Y. & Xiaohua, P. Review of Mammary Gland Hyperplasia in Classical Chinese Medical Literature. Altern. Ther. Health Med. 29, 97–105 (2023).
Dyrstad, S. W., Yan, Y., Fowler, A. M. & Colditz, G. A. Breast cancer risk associated with benign breast disease: systematic review and meta-analysis. Breast Cancer Res. Treat. 149, 569–575. https://doi.org/10.1007/s10549-014-3254-6 (2015).
Mullooly, M. et al. Temporal changes in mammographic breast density and breast cancer risk among women with benign breast disease. Breast Cancer Res. 26 https://doi.org/10.1186/s13058-024-01764-2 (2024).
Kim, S. et al. Mammographic Breast Density, Benign Breast Disease, and Subsequent Breast Cancer Risk in 3.9 Million Korean Women. Radiology 304, 534–541. https://doi.org/10.1148/radiol.212727 (2022).
Hurson, A. N. et al. Risk factors for breast cancer subtypes by race and ethnicity: A scoping review of the literature. medRxiv https://doi.org/10.1101/2024.03.18.24304210 (2024).
Zhang, J. F., Liu, J., Gong, G. H., Zhang, B. & Wei, C. X. Mongolian Medicine RuXian-I Treatment of Estrogen-Induced Mammary Gland Hyperplasia in Rats Related to TCTP Regulating Apoptosis. Evid. Based Complement. Alternat Med. 2019 (1907263). https://doi.org/10.1155/2019/1907263 (2019).
Liu, A. B., Liu, J., Wang, S., Ma, L. & Zhang, J. F. Biological role and expression of translationally controlled tumor protein (TCTP) in tumorigenesis and development and its potential for targeted tumor therapy. Cancer Cell. Int. 24, 198. https://doi.org/10.1186/s12935-024-03355-9 (2024).
Amson, R. & Telerman, A. in Results and Problems in Cell Differentiation, 1 online resource (X, 309 pages 351 illustrations, 336 illustrations in color (Springer International Publishing, 2017).
Li, F., Zhang, D. & Fujise, K. Characterization of fortilin, a novel antiapoptotic protein. J. Biol. Chem. 276, 47542–47549. https://doi.org/10.1074/jbc.M108954200 (2001).
Amson, R. et al. Reciprocal repression between P53 and TCTP. Nat. Med. 18, 91–99. https://doi.org/10.1038/nm.2546 (2011).
Zhao, H. et al. The anti-hyperplasia effect of polysaccharide from Prunella vulgaris L. on mammary gland hyperplasia in rats through regulation of the AKT-FOXO3a signaling pathway and intestinal flora. J. Sci. Food Agric. 104, 8190–8200. https://doi.org/10.1002/jsfa.13652 (2024).
Liang, C. et al. Exploring the inhibitory impact of Mongolian medicinal He-Zi-3 soup on mammary gland hyperplasia in rats induced by estrogen and progestogen. J. Ethnopharmacol. 329, 117854. https://doi.org/10.1016/j.jep.2024.117854 (2024).
Liu, J., Zhang, J. F., Gong, G. H., Zhang, B. & Wei, C. X. Therapeutic effect of mongolian medicine RuXian-I on hyperplasia of mammary gland induced by estrogen/progesterone through CRYAB-promoted apoptosis. Evid. Based Complement. Altern. Med. 5707106, (2020). https://doi.org/10.1155/2020/5707106 (2020).
Liu, Y. et al. Dose-dependent effects of royal jelly on estrogen- and progesterone-induced mammary gland hyperplasia in rats. Mol. Nutr. Food Res. 66, e2100355. https://doi.org/10.1002/mnfr.202100355 (2022).
Das Gupta, S. et al. Tocopherols inhibit oxidative and nitrosative stress in estrogen-induced early mammary hyperplasia in ACI rats. Mol. Carcinog. 54, 916–925. https://doi.org/10.1002/mc.22164 (2015).
Silva, M. S. B. et al. Activation of arcuate nucleus GABA neurons promotes luteinizing hormone secretion and reproductive dysfunction: Implications for polycystic ovary syndrome. EBioMedicine 44, 582–596. https://doi.org/10.1016/j.ebiom.2019.05.065 (2019).
Dewailly, D. et al. Interactions between androgens, FSH, anti-Mullerian hormone and estradiol during folliculogenesis in the human normal and polycystic ovary. Hum. Reprod. Update. 22, 709–724. https://doi.org/10.1093/humupd/dmw027 (2016).
Munoz, J., Wheler, J. & Kurzrock, R. Expression of estrogen and progesterone receptors across human malignancies: new therapeutic opportunities. Cancer Metastasis Rev. 34, 547–561. https://doi.org/10.1007/s10555-014-9543-z (2015).
Le Romancer, M. et al. Cracking the estrogen receptor’s posttranslational code in breast tumors. Endocr. Rev. 32, 597–622. https://doi.org/10.1210/er.2010-0016 (2011).
Shen, K. et al. Anticancer or carcinogenic? The role of estrogen receptor beta in breast cancer progression. Pharmacol. Ther. 242, 108350. https://doi.org/10.1016/j.pharmthera.2023.108350 (2023).
Tanos, T., Rojo, L., Echeverria, P. & Brisken, C. ER and PR signaling nodes during mammary gland development. Breast Cancer Res. 14, 210. https://doi.org/10.1186/bcr3166 (2012).
Intrieri, T., Manneschi, G. & Caldarella, A. 10-year survival in female breast cancer patients according to ER, PR and HER2 expression: a cancer registry population-based analysis. J. Cancer Res. Clin. Oncol. 149, 4489–4496. https://doi.org/10.1007/s00432-022-04245-1 (2023).
Bommer, U. A. & Telerman, A. Dysregulation of TCTP in Biological Processes and Diseases. Cells 9 https://doi.org/10.3390/cells9071632 (2020).
Shen, J. H. et al. siRNA targeting TCTP suppresses osteosarcoma cell growth and induces apoptosis in vitro and in vivo. Biotechnol. Appl. Biochem. 63, 5–14. https://doi.org/10.1002/bab.1335 (2016).
Amson, R. et al. TCTP regulates genotoxic stress and tumorigenicity via intercellular vesicular signaling. EMBO Rep. 25, 1962–1986. https://doi.org/10.1038/s44319-024-00108-7 (2024).
Xia, D. et al. Targeting inhibition of TCTP could inhibit proliferation and induce apoptosis in AML cells. Cell. Signal. 117, 111074. https://doi.org/10.1016/j.cellsig.2024.111074 (2024).
Sun, R., Lu, X., Gong, L. & Jin, F. TCTP promotes epithelial-mesenchymal transition in lung adenocarcinoma. Onco Targets Ther. 12, 1641–1653. https://doi.org/10.2147/OTT.S184555 (2019).
Zhang, J. F. et al. 18beta-Glycyrrhetinic acid inhibits the proliferation and metastasis of gastric cancer by inhibiting the TCTP/AKT/P53 signaling pathway. Sci. Rep. 15, 33429. https://doi.org/10.1038/s41598-025-18912-z (2025).
Du, J., Yang, P., Kong, F. & Liu, H. Aberrant expression of translationally controlled tumor protein (TCTP) can lead to radioactive susceptibility and chemosensitivity in lung cancer cells. Oncotarget 8, 101922–101935. https://doi.org/10.18632/oncotarget.21747 (2017).
Zhang, X., Niu, Y. & Huang, Y. Melatonin inhibits cell proliferation in a rat model of breast hyperplasia by mediating the PTEN/AKT pathway. Oncol. Rep. 45, 66. https://doi.org/10.3892/or.2021.8017 (2021).
Bergmann, A. Survival signaling goes BAD. Dev. Cell. 3, 607–608. https://doi.org/10.1016/s1534-5807(02)00328-3 (2002).
Fischer, N. et al. A novel ligand of the translationally controlled tumor protein (TCTP) identified by virtual drug screening for cancer differentiation therapy. Invest. New. Drugs. 39, 914–927. https://doi.org/10.1007/s10637-020-01042-w (2021).
Jeffreys, S. A. et al. Prognostic and Predictive Value of CCND1/Cyclin D1 Amplification in Breast Cancer With a Focus on Postmenopausal Patients: A Systematic Review and Meta-Analysis. Front. Endocrinol. (Lausanne). 13, 895729. https://doi.org/10.3389/fendo.2022.895729 (2022).
Acknowledgements
We thank Ningxia Keji Biological Co., Ltd., for providing technical support in the histopathological experiments.
Funding
This study was supported by the National Natural Science Foundation of China (82205116; 82160958), the Natural Science Fund of Inner Mongolia (2021MS08077; 2021BS08022).
Author information
Authors and Affiliations
Contributions
ZJF, ML, MMY, LJ, WS and JXM designed the study. SY, CYM, YB and DHH performed multiple experiments and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All animal procedures were carried out in accordance with the regulations of the Animal Protection Committee of Ningxia Medical University, and all experimental procedures were approved by the Ethics Committee of the General Hospital of Ningxia Medical University (KYLL-2022-0094).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ma, MY., Wang, S., Jin, XM. et al. Regulation of the AKT/P53 signaling pathway by translated control tumor protein inhibits apoptosis and promotes hyperplasia of the mammary glands. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43565-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43565-x