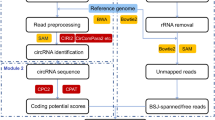
Abstract
CircRNAs have attracted more and more attentions in recent years as they play important roles in many biological processes. It is essential for determining the functions of circRNAs. The subcellular localizations of circRNAs are deemed to be related to their functions. Thus, it is necessary to determine the subcellular localizations of circRNAs. The traditional biochemical experiments are expensive and time-consuming in determining subcellular localizations of circRNAs. It is an alternative way to design computation models. In this study, a new computational model, namely CircLoc, was designed to predict subcellular localizations of circRNAs. This model employed circRNA sequences and networks, from which circRNA features were extracted through both traditional methods (e.g. k-mer), large language model (RNAErnie), and network representation learning algorithms (e.g. node2vec, graph attention auto-encoder). All features were processed by a self-attention layer and fed into a fully connected layer to make predictions. The model was evaluated by ten-fold cross-validation, yielding average AUC and AUPR of 0.7856 and 0.4055, respectively. Such performance was better than that of the models using traditional multi-label classification algorithms and the miRNA subcellular localization prediction models. The reasonableness of CircLoc was also elaborated using ablation tests. The CircLoc was effective in predicting circRNA subcellular localizations and can be a latent useful tool in circRNA study.
Similar content being viewed by others
Data availability
All codes and data are available at https:/github.com/CNHanFei/CircLoc.
References
Oddo, J. C., Saxena, T., McConnell, O. L., Berglund, J. A. & Wang, E. T. Conservation of context-dependent splicing activity in distant Muscleblind homologs. Nucleic Acids Res. 44, 8352–8362. https://doi.org/10.1093/nar/gkw735 (2016).
Cheng, L. & Leung, K. S. Quantification of non-coding RNA target localization diversity and its application in cancers. J. Mol. Cell. Biol. 10, 130–138. https://doi.org/10.1093/jmcb/mjy006 (2018).
Frias-Lasserre, D. & Villagra, C. A. The importance of ncRNAs as epigenetic mechanisms in phenotypic variation and organic evolution. Front. Microbiol. 8, 2483. https://doi.org/10.3389/fmicb.2017.02483 (2017).
Patop, I. L., Wüst, S. & Kadener, S. Past, present, and future of circRNAs. EMBO J. 38, e100836 (2019).
Sanger, H. L., Klotz, G., Riesner, D., Gross, H. J. & Kleinschmidt, A. K. Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc. Natl. Acad. Sci. U. S. A. 73, 3852–3856. https://doi.org/10.1073/pnas.73.11.3852 (1976).
Cocquerelle, C., Mascrez, B., Hetuin, D. & Bailleul, B. Mis-splicing yields circular RNA molecules. FASEB J. 7, 155–160. https://doi.org/10.1096/fasebj.7.1.7678559 (1993).
Li, Z. et al. Exon-intron circular RNAs regulate transcription in the nucleus. Nat. Struct. Mol. Biol. 22, 256–264. https://doi.org/10.1038/nsmb.2959 (2015).
Salzman, J., Gawad, C., Wang, P. L., Lacayo, N. & Brown, P. O. Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types. PLoS One 7, e30733. https://doi.org/10.1371/journal.pone.0030733 (2012).
Geng, X. et al. Circular RNA: Biogenesis, degradation, functions and potential roles in mediating resistance to anticarcinogens. Epigenomics 12, 267–283. https://doi.org/10.2217/epi-2019-0295 (2020).
Liu, J., Yang, L., Fu, Q. & Liu, S. Emerging roles and potential biological value of CircRNA in osteosarcoma. Front. Oncol. 10, 552236. https://doi.org/10.3389/fonc.2020.552236 (2020).
Holdt, L. M., Kohlmaier, A. & Teupser, D. Molecular roles and function of circular RNAs in eukaryotic cells. Cell. Mol. Life Sci. 75, 1071–1098. https://doi.org/10.1007/s00018-017-2688-5 (2018).
Bachmayr-Heyda, A. et al. Correlation of circular RNA abundance with proliferation–Exemplified with colorectal and ovarian cancer, idiopathic lung fibrosis, and normal human tissues. Sci. Rep. 5, 8057. https://doi.org/10.1038/srep08057 (2015).
Li, F. et al. Circular RNA ITCH has inhibitory effect on ESCC by suppressing the Wnt/β-catenin pathway. Oncotarget 6, 6001–6013. https://doi.org/10.18632/oncotarget.3469 (2015).
Li, P. et al. Using circular RNA as a novel type of biomarker in the screening of gastric cancer. Clin. Chim. Acta 444, 132–136. https://doi.org/10.1016/j.cca.2015.02.018 (2015).
Lei, B., Tian, Z., Fan, W. & Ni, B. Circular RNA: A novel biomarker and therapeutic target for human cancers. Int. J. Med. Sci. 16, 292 (2019).
Zhang, Z., Yang, T. & Xiao, J. Circular RNAs: Promising biomarkers for human diseases. EBioMedicine 34, 267–274 (2018).
Chen, L. & Shan, G. CircRNA in cancer: Fundamental mechanism and clinical potential. Cancer Lett. 505, 49–57 (2021).
Zhang, T. et al. Protein subcellular localization prediction model based on graph convolutional network. Interdiscip. Sci. Comput. Life Sci. 14, 937–946. https://doi.org/10.1007/s12539-022-00529-9 (2022).
Ullah, M. et al. PScL-HDeep: Image-based prediction of protein subcellular location in human tissue using ensemble learning of handcrafted and deep learned features with two-layer feature selection. Brief. Bioinform. 22, bbab278. https://doi.org/10.1093/bib/bbab278 (2021).
Cheng, X., Zhao, S. G., Lin, W. Z., Xiao, X. & Chou, K. C. pLoc-mAnimal: Predict subcellular localization of animal proteins with both single and multiple sites. Bioinformatics 33, 3524–3531. https://doi.org/10.1093/bioinformatics/btx476 (2017).
Chen, L., Qu, R. & Liu, X. Improved multi-label classifiers for predicting protein subcellular localization. Math. Biosci. Eng. 21, 214–236. https://doi.org/10.3934/mbe.2024010 (2024).
Pan, X. et al. Identifying protein subcellular locations with embeddings-based node2loc. IEEE/ACM Trans. Comput. Biol. Bioinform. 19, 666–675. https://doi.org/10.1109/tcbb.2021.3080386 (2021).
Wang, R. & Chen, L. Identification of human protein subcellular location with multiple networks. Curr. Proteomics. 19, 344–356 (2022).
Musleh, S., Islam, M. T., Qureshi, R., Alajez, N. M. & Alam, T. MSLP: mRNA subcellular localization predictor based on machine learning techniques. BMC Bioinformatics 24, 109 (2023).
Yan, Z., Lécuyer, E. & Blanchette, M. Prediction of mRNA subcellular localization using deep recurrent neural networks. Bioinformatics 35, i333–i342 (2019).
Wang, D. et al. DM3Loc: Multi-label mRNA subcellular localization prediction and analysis based on multi-head self-attention mechanism. Nucleic Acids Res. 49, e46–e46 (2021).
Xiao, Y., Cai, J., Yang, Y., Zhao, H. & Shen, H. in IEEE International Conference on Data Mining (ICDM). 1332–1337 (IEEE). 1332–1337 (IEEE). (2018).
Asim, M. N. et al. MirLocPredictor: A ConvNet-based multi-label microRNA subcellular localization predictor by incorporating k-Mer positional information. Genes (Basel) 11, 1475. https://doi.org/10.3390/genes11121475 (2020).
Asim, M. N. et al. in 2021 International Joint Conference on Neural Networks (IJCNN). 1–8 (IEEE).
Meher, P. K., Satpathy, S. & Rao, A. R. miRNALoc: Predicting miRNA subcellular localizations based on principal component scores of physico-chemical properties and pseudo compositions of di-nucleotides. Sci. Rep. 10, 14557. https://doi.org/10.1038/s41598-020-71381-4 (2020).
Yang, Y., Fu, X., Qu, W., Xiao, Y. & Shen, H. B. MiRGOFS: A GO-based functional similarity measurement for miRNAs, with applications to the prediction of miRNA subcellular localization and miRNA-disease association. Bioinformatics 34, 3547–3556. https://doi.org/10.1093/bioinformatics/bty343 (2018).
Xu, M. et al. MiRLoc: Predicting miRNA subcellular localization by incorporating miRNA-mRNA interactions and mRNA subcellular localization. Brief. Bioinform. 23, bbac044. https://doi.org/10.1093/bib/bbac044 (2022).
Bai, T., Yan, K. & Liu, B. DAmiRLocGNet: miRNA subcellular localization prediction by combining miRNA-disease associations and graph convolutional networks. Brief. Bioinform. 24, bbad212. https://doi.org/10.1093/bib/bbad212 (2023).
Chen, L., Gu, J. & Zhou, B. PMiSLocMF: Predicting miRNA subcellular localizations by incorporating multi-source features of miRNAs. Brief. Bioinform. 25, bbae386 (2024).
Liang, Y. et al. MGFmiRNAloc: Predicting miRNA subcellular localization using molecular graph feature and convolutional block attention module. IEEE/ACM Trans. Comput. Biol. Bioinform. 21, 1348–1357. https://doi.org/10.1109/tcbb.2024.3383438 (2024).
Jiang, J. & Yan, C. PMLocMSCAM: Predicting miRNA subcellular localisations by miRNA similarities and cross-attention mechanism. IET Syst. Biol. 19, e70023. https://doi.org/10.1049/syb2.70023 (2025).
Asim, M. N., Ibrahim, M. A., Imran Malik, M., Dengel, A. & Ahmed, S. Circ-LocNet: A computational framework for circular RNA sub-cellular localization prediction. Int. J. Mol. Sci. 23, 8221. https://doi.org/10.3390/ijms23158221 (2022).
Zeng, M. et al. CellCircLoc: Deep neural network for predicting and explaining cell line-specific CircRNA subcellular localization. IEEE J. Biomed. Health Inform. 29, 1494–1503. https://doi.org/10.1109/jbhi.2024.3491732 (2025).
Wei, M.-M. et al. Integrating transformer and graph attention network for circRNA-miRNA interaction prediction. IEEE J. Biomed. Health Inform. https://doi.org/10.1109/JBHI.2025.3561197 (2025).
Yuan, S. et al. FDENet: Frequency-guided dual-encoder network for building footprint extraction from remote sensing images. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. https://doi.org/10.1109/JSTARS.2025.3601023 (2025).
Li, D. et al. DeepHIV: A sequence-based deep learning model for predicting HIV-1 protease cleavage sites. IEEE Trans. Comput. Biol. Bioinform. https://doi.org/10.1109/TCBBIO.2025.3610881 (2025).
Lan, W. et al. The large language models on biomedical data analysis: A survey. IEEE J. Biomed. Health Inform. 29, 4486–4497. https://doi.org/10.1109/JBHI.2025.3530794 (2025).
Noor, S. et al. Deep-m5U: A deep learning-based approach for RNA 5-methyluridine modification prediction using optimized feature integration. BMC Bioinformatics 25, 360. https://doi.org/10.1186/s12859-024-05978-1 (2024).
Khan, S., AlQahtani, S. A., Noor, S. & Ahmad, N. PSSM-Sumo: Deep learning based intelligent model for prediction of sumoylation sites using discriminative features. BMC Bioinformatics 25, 284. https://doi.org/10.1186/s12859-024-05917-0 (2024).
Khan, S. et al. XGBoost-enhanced ensemble model using discriminative hybrid features for the prediction of sumoylation sites. Biodata Min. 18, 12. https://doi.org/10.1186/s13040-024-00415-8 (2025).
Khan, S., Dilshad, N., Ahmad, N., Noor, S. & AlQahtani, S. A. Integrating AI in security information and event management for real time cyber defense. Sci. Rep. 15, 35872 (2025).
Salehi, A. & Davulcu, H. Graph attention auto-encoders. arXiv preprint, doi:arXiv: 10715 (2019). (1905).
Wu, L. et al. RNALocate v3.0: Advancing the repository of RNA subcellular localization with dynamic analysis and prediction. Nucleic Acids Res. 53, D284-d292. https://doi.org/10.1093/nar/gkae872 (2025).
Cui, T. et al. RNALocate v2.0: An updated resource for RNA subcellular localization with increased coverage and annotation. Nucleic Acids Res. 50, D333–D339. https://doi.org/10.1093/nar/gkab825 (2022).
Feng, J. et al. CSCD2: An integrated interactional database of cancer-specific circular RNAs. Nucleic Acids Res. 50, D1179-d1183. https://doi.org/10.1093/nar/gkab830 (2022).
Liu, B., Liu, F., Fang, L., Wang, X. & Chou, K. C. repDNA: A Python package to generate various modes of feature vectors for DNA sequences by incorporating user-defined physicochemical properties and sequence-order effects. Bioinformatics 31, 1307–1309. https://doi.org/10.1093/bioinformatics/btu820 (2015).
Liu, B. et al. Pse-in-One: A web server for generating various modes of pseudo components of DNA, RNA, and protein sequences. Nucleic Acids Res. 43, W65-71. https://doi.org/10.1093/nar/gkv458 (2015).
Liu, B., Liu, F., Fang, L., Wang, X. & Chou, K. C. repRNA: A web server for generating various feature vectors of RNA sequences. Mol. Genet. Genomics. 291, 473–481. https://doi.org/10.1007/s00438-015-1078-7 (2016).
Liu, B. BioSeq-Analysis: A platform for DNA, RNA and protein sequence analysis based on machine learning approaches. Brief. Bioinform. 20, 1280–1294. https://doi.org/10.1093/bib/bbx165 (2019).
Glazar, P., Papavasileiou, P. & Rajewsky, N. circBase: A database for circular RNAs. RNA 20, 1666–1670. https://doi.org/10.1261/rna.043687.113 (2014).
Lv, H. et al. Evaluation of different computational methods on 5-methylcytosine sites identification. Brief. Bioinform. 21, 982–995. https://doi.org/10.1093/bib/bbz048 (2020).
Tang, G., Shi, J., Wu, W., Yue, X. & Zhang, W. Sequence-based bacterial small RNAs prediction using ensemble learning strategies. BMC Bioinform. 19, 503. https://doi.org/10.1186/s12859-018-2535-1 (2018).
Gupta, S. et al. Predicting human nucleosome occupancy from primary sequence. PLoS Comput. Biol. 4, e1000134. https://doi.org/10.1371/journal.pcbi.1000134 (2008).
Akiyama, M. & Sakakibara, Y. Informative RNA base embedding for RNA structural alignment and clustering by deep representation learning. NAR genomics Bioinf. 4, lqac012. https://doi.org/10.1093/nargab/lqac012 (2022).
Wang, N. et al. Multi-purpose RNA language modelling with motif-aware pretraining and type-guided fine-tuning. Nat. Mach. Intell. 6, 548–557 (2024).
Chen, J. et al. Interpretable RNA foundation model from unannotated data for highly accurate RNA structure and function predictions. arXiv preprint arXiv:2204.00300 (2022).
Wei, M., Wang, L., Su, X., Zhao, B. & You, Z. Multi-hop graph structural modeling for cancer-related circRNA-miRNA interaction prediction. Pattern Recogn. 170, 112078 (2026).
Wang, S., Lee, H. C. & Lee, S. Predicting herb-disease associations using network-based measures in human protein interactome. BMC Complement. Med. Ther. 24, 218. https://doi.org/10.1186/s12906-024-04503-4 (2024).
Yin, A., Chen, L., Zhou, B. & Cai, Y. D. CMAGN: circRNA–miRNA association prediction based on graph attention auto-encoder and network consistency projection. BMC Bioinform. 25, 336. https://doi.org/10.1186/s12859-024-05959-4 (2024).
Smith, T. F. & Waterman, M. S. Identification of common molecular subsequences. J. Mol. Biol. 147, 195–197 (1981).
Meng, S. et al. CircRNA: Functions and properties of a novel potential biomarker for cancer. Mol. Cancer 16, 1–8 (2017).
Gao, J.-L., Chen, G., He, H.-Q. & Wang, J. CircRNA as a new field in human disease research. Zhongguo Zhong Yao Za Zhi 43, 457–462 (2018).
Guarnerio, J. et al. Oncogenic role of fusion-circRNAs derived from cancer-associated chromosomal translocations. Cell 165, 289–302 (2016).
Shang, Q., Yang, Z., Jia, R. & Ge, S. The novel roles of circRNAs in human cancer. Mol. Cancer. 18, 1–10 (2019).
Lan, W. et al. LGCDA: Predicting circRNA-disease association based on fusion of local and global features. IEEE/ACM Trans. Comput. Biol. Bioinform. 21, 1413–1422. https://doi.org/10.1109/TCBB.2024.3387913 (2024).
Lan, W. et al. IGNSCDA: Predicting circRNA-disease associations based on improved graph convolutional network and negative sampling. IEEE/ACM Trans. Comput. Biol. Bioinform. 19, 3530–3538. https://doi.org/10.1109/TCBB.2021.3111607 (2022).
Lan, W. et al. KGANCDA: Predicting circRNA-disease associations based on knowledge graph attention network. Brief Bioinform. 23, bbab494. https://doi.org/10.1093/bib/bbab494 (2022).
Lan, W. et al. Predicting CircRNA-disease associations based on heterogeneous graph neural network and knowledge graph attribute mining attention. Interdiscip. Sci. Comput. Life Sci. 17, 586–597. https://doi.org/10.1007/s12539-025-00706-6 (2025).
Lan, W. et al. Benchmarking of computational methods for predicting circRNA-disease associations. Brief. Bioinform. 24, bbac613. https://doi.org/10.1093/bib/bbac613 (2023).
Chen, L. & Zhao, X. PCDA-HNMP: Predicting circRNA-disease association using heterogeneous network and meta-path. Math. Biosci. Eng. 20, 20553–20575 (2023).
Fan, C. et al. CircR2Disease v2.0: An updated web server for experimentally validated circRNA-disease associations and its application. Genomics. Proteomics. Bioinform. 20, 435–445. https://doi.org/10.1016/j.gpb.2021.10.002 (2022).
Wei, L. et al. Noncoding RNAs in gastric cancer: Implications for drug resistance. Mol. Cancer. 19, 62. https://doi.org/10.1186/s12943-020-01185-7 (2020).
Cui, C. et al. Functions and mechanisms of circular RNAs in cancer radiotherapy and chemotherapy resistance. Mol. Cancer. 19, 58. https://doi.org/10.1186/s12943-020-01180-y (2020).
Deng, L., Liu, Z., Qian, Y. & Zhang, J. Predicting circRNA-drug sensitivity associations via graph attention auto-encoder. BMC. Bioinform. 23, 160. https://doi.org/10.1186/s12859-022-04694-y (2022).
Chen, Y., Wang, Y., Ding, Y., Su, X. & Wang, C. RGCNCDA: Relational graph convolutional network improves circRNA-disease association prediction by incorporating microRNAs. Comput. Biol. Med. 143, 105322. https://doi.org/10.1016/j.compbiomed.2022.105322 (2022).
Liu, M., Wang, Q., Shen, J., Yang, B. B. & Ding, X. Circbank: A comprehensive database for circRNA with standard nomenclature. RNA. Biol. 16, 899–905. https://doi.org/10.1080/15476286.2019.1600395 (2019).
Dudekula, D. B. et al. CircInteractome: A web tool for exploring circular RNAs and their interacting proteins and microRNAs. RNA Biol. 13, 34–42. https://doi.org/10.1080/15476286.2015.1128065 (2016).
Grover, A. & Leskovec, J. in Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining 855–864ACM, San Francisco, California, USA, (2016).
Kingma, D. P. & Ba, J. in 3rd International Conference on Learning RepresentationsLouisiana, USA, (2019).
Kohavi, R. in International joint Conference on artificial intelligence. 1137–1145 (Lawrence Erlbaum Associates Ltd).
Yuan, F. et al. Integrative multi-omics machine learning reveals novel driver genes associations in lung adenocarcinoma. Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics 1874, 141113. https://doi.org/10.1016/j.bbapap.2025.141113 (2026).
Ma, Q. et al. Identifying transcriptional signatures of leukocytes in tissue and blood for multicancer diagnosis by using machine learning methods. Cancer Genet. 302–303, 13–26. https://doi.org/10.1016/j.cancergen.2026.01.003 (2026).
Ren, J. et al. Identification of Gene Signatures Associated with Multisystem Inflammatory Syndrome in Children after SARS-CoV-2 Infection. Current Bioinformatics (2026).
Chen, L., Yang, J., Zhou, B. & Cai, Y.-D. PLysPTM-HGNN: Predicting lysine PTM sites of proteins using hybrid graph neural networks. BMC. Bioinformatics 27, 32 (2026).
Powers, D. Evaluation: From precision, recall and f-measure to roc., informedness, markedness & correlation. Journal of Machine Learning Technologies 2, 37–63 (2011).
Chen, L., Xu, L., Zhou, B., Chen, Y. & AntiCanNet A Graph Convolution and Chemical LLM Framework for Predicting Anti-Cancer Small Molecules. Current Bioinformatics (2026).
Chen, L., Lu, Y., Xu, J. & Zhou, B. Prediction of drug’s anatomical therapeutic chemical (ATC) code by constructing biological profiles of ATC codes. BMC. Bioinformatics 26, 86 (2025).
Chen, L., Zhang, S. & Zhou, B. Herb-disease association prediction model based on network consistency projection. Sci. Rep. 15, 3328 (2025).
Chen, L., Xun, X. & Zhou, B. Root-associated protein prediction using a protein large language model and hypergraph convolutional networks. Sci. Rep. 16, 4876 (2026).
Pedregosa, F. et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Yang, B. & Chen, H. Predicting circRNA-drug sensitivity associations by learning multimodal networks using graph auto-encoders and attention mechanism. Brief. Bioinform. 24, bbac596. https://doi.org/10.1093/bib/bbac596 (2023).
Chen, L. et al. Predicting human protein subcellular locations by using a combination of network and function features. Front. Genet. 12, 783128. https://doi.org/10.3389/fgene.2021.783128 (2021).
Pan, X. et al. Identifying protein subcellular locations with embeddings-based node2loc. IEEE/ACM Trans. Comput. Biol. Bioinform. 19, 666–675. https://doi.org/10.1109/tcbb.2021.3080386 (2022).
Garapati, H. S., Male, G. & Mishra, K. Predicting subcellular localization of proteins using protein-protein interaction data. Genomics 112, 2361–2368. https://doi.org/10.1016/j.ygeno.2020.01.007 (2020).
Zhang, M.-L., Li, Y.-K., Liu, X.-Y. & Geng, X. Binary relevance for multi-label learning: An overview. Front. Comput. Sci. 12, 191–202. https://doi.org/10.1007/s11704-017-7031-7 (2018).
Tsoumakas, G., Katakis, I. & Vlahavas, I. Random k-Labelsets for multilabel classification. IEEE. Trans. Knowl. Data Eng. 23, 1079–1089. https://doi.org/10.1109/TKDE.2010.164 (2011).
Read, J., Reutemann, P., Pfahringer, B. & Holmes, G. MEKA: A multi-label/multi-target extension to WEKA. J. Mach. Learn. Res. 17, 1–5 (2016).
Author information
Authors and Affiliations
Contributions
L.C. designed the research; L.C., J.H. and B.Z. conducted the experiments; J.H. and B.Z. analyzed the results; L.C. and J.H. wrote the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, L., Hu, J. & Zhou, B. Predicting circRNA subcellular localization by fusing circRNA sequence and network information. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43808-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43808-x