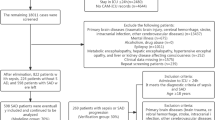
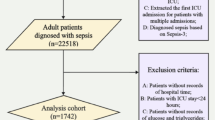
Abstract
Sepsis-associated delirium (SAD) is a common and severe complication in critically ill patients and is associated with increased mortality. Thiamine is an essential coenzyme in mitochondrial energy metabolism, and deficiency is frequent in critical illness. However, the association between thiamine supplementation and survival in ICU patients with SAD remains unclear. We conducted a retrospective cohort study using the MIMIC-IV (v3.1) database. Adult ICU patients meeting Sepsis-3 criteria and diagnosed with delirium during ICU stay were included. Patients were categorized according to thiamine supplementation during ICU admission. Propensity score–based methods, including matching, adjustment, and multiple weighting approaches, were applied to balance baseline covariates. The primary outcome was 30-day all-cause mortality. We performed survival, subgroup, and sensitivity analyses. We also conducted duration–response and dose–response analyses to evaluate whether thiamine treatment duration and average daily dose were associated with prognosis. In the MIMIC-IV cohort, 332 ICU patients with sepsis-associated delirium received thiamine supplementation, while 956 did not. Thirty-day mortality was significantly lower in the thiamine group compared with the non-thiamine group (P < 0.001). Thiamine use was associated with improved survival in crude analysis and remained significantly associated with lower 30-day mortality after multivariable adjustment (HR, 0.51; 95% CI, 0.33–0.79; P = 0.002). Consistent associations were observed across propensity score analyses, including propensity score–adjusted analysis (HR, 0.59; 95% CI, 0.40–0.85; P = 0.005), propensity score matching (HR, 0.59; 95% CI, 0.39–0.90; P = 0.015), inverse probability of treatment weighting (HR, 0.54; 95% CI, 0.38–0.77; P = 0.002), standardized mortality ratio weighting (HR, 0.68; 95% CI, 0.47–0.98; P = 0.052), pairwise algorithmic weighting (HR, 0.63; 95% CI, 0.40–0.97; P = 0.016), and overlap weight (HR, 0.60; 95% CI, 0.36–1.02; P = 0.008). Weighted subgroup analyses demonstrated consistent associations across clinical strata, with a significant interaction by illness severity indicating a survival benefit of thiamine among patients with SOFA scores < 4. Duration-response and dose-response analyses suggested greater benefit with longer treatment courses and lower daily dosing. Thiamine supplementation was associated with reduced 30-day mortality in ICU patients with sepsis-associated delirium, with an observed interaction in the SOFA score subgroup, where a SOFA score below 4 was associated with survival benefit. Duration–response and dose–response analyses suggested greater benefit with longer treatment courses and lower daily dosing.
Similar content being viewed by others
Data availability
The datasets analyzed during this investigation are publicly accessible in the MIMIC-IV repository (v3.1) at: https://physionet.org/content/mimiciv/3.1/.
References
Wilson, J. E. et al. Delirium. Nat Rev. Dis. Primers. 6, 90. https://doi.org/10.1038/s41572-020-00223-4 (2020).
Siddiqi, N., House, A. O. & Holmes, J. D. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 35, 350–364. https://doi.org/10.1093/ageing/afl005 (2006).
Van Den Boogaard, M. et al. The impact of delirium on the prediction of in-hospital mortality in intensive care patients. Crit. Care. 14, R146. https://doi.org/10.1186/cc9214 (2010).
Turon, M. et al. Mechanisms involved in brain dysfunction in mechanically ventilated critically ill patients: implications and therapeutics. Ann. Transl Med. 6, 30–30. https://doi.org/10.21037/atm.2017.12.10 (2018).
Sasannejad, C., Ely, E. W. & Lahiri, S. Long-term cognitive impairment after acute respiratory distress syndrome: a review of clinical impact and pathophysiological mechanisms. Crit. Care. 23, 352. https://doi.org/10.1186/s13054-019-2626-z (2019).
Balas, M. C. et al. Effectiveness and safety of the awakening and breathing coordination, delirium monitoring/management, and early exercise/mobility bundle*. Crit. Care Med. 42, 1024–1036. https://doi.org/10.1097/CCM.0000000000000129 (2014).
Lee, H. W., Park, Y., Jang, E. J. & Lee, Y. J. Intensive care unit length of stay is reduced by protocolized family support intervention: a systematic review and meta-analysis. Intensive Care Med. 45, 1072–1081. https://doi.org/10.1007/s00134-019-05681-3 (2019).
Page, V. J. et al. Effect of intravenous haloperidol on the duration of delirium and coma in critically ill patients (hope-ICU): a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 1, 515–523. https://doi.org/10.1016/S2213-2600(13)70166-8 (2013).
Schrijver, E. J. M. et al. Haloperidol versus placebo for delirium prevention in acutely hospitalised older at risk patients: a multi-centre double-blind randomised controlled clinical trial. Age Ageing. 47, 48–55. https://doi.org/10.1093/ageing/afx124 (2018).
Agar, M. R. et al. Efficacy of oral risperidone, haloperidol, or placebo for symptoms of delirium among patients in palliative care: a randomized clinical trial. JAMA Intern. Med. 177, 34. https://doi.org/10.1001/jamainternmed.2016.7491 (2017).
Brück, E., Schandl, A., Bottai, M. & Sackey, P. The impact of sepsis, delirium, and psychological distress on self-rated cognitive function in ICU survivors-a prospective cohort study. J. Intensive Care. 6, 2. https://doi.org/10.1186/s40560-017-0272-6 (2018).
Sonneville, R. et al. Potentially modifiable factors contributing to sepsis-associated encephalopathy. Intensive Care Med. 43, 1075–1084. https://doi.org/10.1007/s00134-017-4807-z (2017).
Tokuda, R. et al. Sepsis-associated delirium: a narrative review. J. Clin. Med. 12, 1273. https://doi.org/10.3390/jcm12041273 (2023).
Pendlebury, S. et al. Observational, longitudinal study of delirium in consecutive unselected acute medical admissions: age-specific rates and associated factors, mortality and re-admission. BMJ Open. 5, e007808. https://doi.org/10.1136/bmjopen-2015-007808 (2015).
Mazeraud, A. et al. Septic-associated encephalopathy: a comprehensive review. Neurotherapeutics 17, 392–403. https://doi.org/10.1007/s13311-020-00862-1 (2020).
Sekino, N., Selim, M. & Shehadah, A. Sepsis-associated brain injury: underlying mechanisms and potential therapeutic strategies for acute and long-term cognitive impairments. J. Neuroinflammation. 19, 101. https://doi.org/10.1186/s12974-022-02464-4 (2022).
Gao, Q. & Hernandes, M. S. Sepsis-associated encephalopathy and blood-brain barrier dysfunction. Inflammation 44, 2143–2150. https://doi.org/10.1007/s10753-021-01501-3 (2021).
Sonneville, R. et al. The spectrum of sepsis-associated encephalopathy: a clinical perspective. Crit. Care. 27, 386. https://doi.org/10.1186/s13054-023-04655-8 (2023).
Girard, T. D. et al. Clinical phenotypes of delirium during critical illness and severity of subsequent long-term cognitive impairment: a prospective cohort study. Lancet Respir Med. 6, 213–222. https://doi.org/10.1016/S2213-2600(18)30062-6 (2018).
Devlin, J. W. et al. Clinical practice guidelines for the prevention and management of pain, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit. Care Med. 46, e825–e873. https://doi.org/10.1097/CCM.0000000000003299 (2018).
Park, S. Y. & Lee, H. B. Prevention and management of delirium in critically ill adult patients in the intensive care unit: a review based on the 2018 PADIS guidelines. Acute Crit. Care. 34, 117–125. https://doi.org/10.4266/acc.2019.00451 (2019).
Kerns, J. C. & Gutierrez, J. L. Thiamin. Adv Nutr (bethesda Md,) 8:395–397. (2017). https://doi.org/10.3945/an.116.013979
Zhang, L. et al. Thiamine may Be beneficial for patients with ventilator-associated pneumonia in the intensive care unit: a retrospective study based on the MIMIC-IV database. Front. Pharmacol. 13, 898566. https://doi.org/10.3389/fphar.2022.898566 (2022).
Aleshin, V. A., Mkrtchyan, G. V. & Bunik, V. I. Mechanisms of non-coenzyme action of thiamine: protein targets and medical significance. Biochem. Biokhimiia. 84, 829–850. https://doi.org/10.1134/S0006297919080017 (2019).
Smith, T. J. et al. Thiamine deficiency disorders: a clinical perspective. Ann. N Y Acad. Sci. 1498, 9–28. https://doi.org/10.1111/nyas.14536 (2021).
Manzanares, W. & Hardy, G. Thiamine supplementation in the critically ill. Curr. Opin. Clin. Nutr. Metab. Care. 14, 610–617. https://doi.org/10.1097/MCO.0b013e32834b8911 (2011).
Donnino, M. W. et al. Randomized, double-blind, placebo-controlled trial of thiamine as a metabolic resuscitator in septic shock: a pilot study. Crit. Care Med. 44, 360–367. https://doi.org/10.1097/CCM.0000000000001572 (2016).
Woolum, J. A. et al. Effect of thiamine administration on lactate clearance and mortality in patients with septic shock. Crit. Care Med. 46, 1747–1752. https://doi.org/10.1097/CCM.0000000000003311 (2018).
Iglesias, J. et al. Outcomes of metabolic resuscitation using ascorbic acid, thiamine, and glucocorticoids in the early treatment of sepsis: the ORANGES trial. CHEST 158, 164–173. https://doi.org/10.1016/j.chest.2020.02.049 (2020).
Attaluri, P., Castillo, A., Edriss, H. & Nugent, K. Thiamine deficiency: an important consideration in critically ill patients. Am. J. Med. Sci. 356, 382–390. https://doi.org/10.1016/j.amjms.2018.06.015 (2018).
Johnson, A. E. W. et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data. 10, 1. https://doi.org/10.1038/s41597-022-01899-x (2023).
Erik von Elm et al. The Strengthening the Reporting of Observational Studies in Epidemiology [STROBE] statement: guidelines for reporting observational studies. PLoS Med. 12, 1495–1499. https://doi.org/10.1371/journal.pmed.0040296 (2007).
Mervyn Singer, Singer, M. et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315, 801–810. https://doi.org/10.1001/jama.2016.0287 (2016).
Guenther, U. et al. Validity and Reliability of the CAM-ICU Flowsheet to diagnose delirium in surgical ICU patients. J. Crit. Care. 25, 144–151. https://doi.org/10.1016/j.jcrc.2009.08.005 (2010).
Butterworth, R. F. Thiamine deficiency-related brain dysfunction in chronic liver failure. Metab. Brain Dis. 24, 189–196. https://doi.org/10.1007/s11011-008-9129-y (2009).
Desai, A. P. et al. Confusion assessment method accurately screens for hepatic encephalopathy and predicts short-term mortality in hospitalized patients with cirrhosis. Metab. Brain Dis. 38, 1749–1758. https://doi.org/10.1007/s11011-022-01149-4 (2023).
Pisani, M. A. et al. A research algorithm to improve detection of delirium in the intensive care unit. Crit. Care. 10, R121. https://doi.org/10.1186/cc5027 (2006).
Shi, Q., Warren, L., Saposnik, G. & MacDermid, J. C. Confusion assessment method: a systematic review and meta-analysis of diagnostic accuracy. Neuropsychiatr Dis. Treat. 9, 1359–1370. https://doi.org/10.2147/NDT.S49520 (2013).
Shen, Y., Zhang, W. & Shen, Y. Early diuretic use and mortality in critically ill patients with vasopressor support: a propensity score-matching analysis. Crit. Care (lond Engl). 23, 9. https://doi.org/10.1186/s13054-019-2309-9 (2019).
van den Boogaard, M. & Slooter, A. J. C. Delirium in critically ill patients: current knowledge and future perspectives. BJA Educ. 19, 398–404. https://doi.org/10.1016/j.bjae.2019.09.004 (2019).
van den Boogaard, M. et al. Development and validation of PRE-DELIRIC (PREdiction of DELIRium in ICu patients) delirium prediction model for intensive care patients: observational multicentre study. BMJ 344 https://doi.org/10.1136/bmj.e420 (2012).
Swart, L. M. et al. The comparative risk of delirium with different opioids: a systematic review. Drugs Aging. 34, 437–443. https://doi.org/10.1007/s40266-017-0455-9 (2017).
Austin, P. C. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivar. Behav. Res. 46, 399–424. https://doi.org/10.1080/00273171.2011.568786 (2011).
Austin, P. C. & Stuart, E. A. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat. Med. 34, 3661–3679. https://doi.org/10.1002/sim.6607 (2015).
Sato, T. & Matsuyama, Y. Marginal structural models as a tool for standardization. Epidemiology 14, 680–686. https://doi.org/10.1097/01.EDE.0000081989.82616.7d (2003).
Li, L. & Greene, T. A weighting analogue to pair matching in propensity score analysis. ijb 9, 215–234. https://doi.org/10.1515/ijb-2012-0030 (2013).
Li, F., Morgan, K. L. & Zaslavsky, A. M. Balancing covariates via propensity score weighting. J. Am. Stat. Assoc. 113, 390–400. https://doi.org/10.1080/01621459.2016.1260466 (2018).
Rosenbaum, P. R. & Rubin, D. B. The central role of the propensity score in observational studies for causal effects. 0–15 (1983).
Brookhart, M. A., Wyss, R., Layton, J. B. & Stürmer, T. Propensity score methods for confounding control in nonexperimental research. Circ: Cardiovasc. Qual. Outcomes. 6, 604–611. https://doi.org/10.1161/CIRCOUTCOMES.113.000359 (2013).
Hashimoto, Y. & Yasunaga, H. Theory and practice of propensity score analysis. Ann. Clin. Epidemiol. 4, 101–109. https://doi.org/10.37737/ace.22013 (2022).
Yang, Q. et al. Association between metformin use on admission and outcomes in intensive care unit patients with acute kidney injury and type 2 diabetes: A retrospective cohort study. J. Crit. Care. 62, 206–211. https://doi.org/10.1016/j.jcrc.2020.12.007 (2021).
Yang, Q. et al. Association between wait time of central venous pressure measurement and outcomes in critical patients with acute kidney injury: a retrospective cohort study. Front. Public. Health. 10, 893683. https://doi.org/10.3389/fpubh.2022.893683 (2022).
Trancă, S., Petrișor, C., Hagău, N. & Ciuce, C. Can APACHE II, SOFA, ISS, and RTS severity scores be used to predict septic complications in multiple trauma patients? J. Crit. Care Med. 2, 124–130. https://doi.org/10.1515/jccm-2016-0019 (2016).
Zhang, T. et al. Thiamine use and short-term prognosis in critical cerebrovascular disease: a propensity score matched retrospective analysis from the MIMIC-IV database. BMC Neurol. 25, 450. https://doi.org/10.1186/s12883-025-04478-w (2025).
Dingwall, K. M. et al. What is the optimum thiamine dose to treat or prevent wernicke’s encephalopathy or wernicke–korsakoff syndrome? Results of a randomized controlled trial. Alcohol: Clin. Exp. Res. 46, 1133–1147. https://doi.org/10.1111/acer.14843 (2022).
De Man, A. M. E. et al. LLL 44 – 4: micronutrients in acute disease and critical illness. Clin. Nutr. ESPEN. 61, 437–446. https://doi.org/10.1016/j.clnesp.2024.04.011 (2024).
Berger, M. M. et al. ESPEN practical short micronutrient guideline. Clin. Nutr. 43, 825–857. https://doi.org/10.1016/j.clnu.2024.01.030 (2024).
Rosewarne, R. E. & Farina, N. Thiamine use in hospitalized patients: a clinical review. J. Nutr. Metab. 2026, 7143046. https://doi.org/10.1155/jnme/7143046 (2026).
Donnino, M. W. et al. Randomized, double-blind, placebo-controlled trial of thiamine as a metabolic resuscitator in septic shock: a pilot study. Crit. Care Med. 44, 361–367. https://doi.org/10.1097/CCM.0000000000001572 (2016).
Kaźmierczak-Barańska, J., Halczuk, K. & Karwowski, B. T. Thiamine (vitamin B1)—an essential health regulator. Nutrients 17, 2206. https://doi.org/10.3390/nu17132206 (2025).
Depeint, F. et al. Mitochondrial function and toxicity: role of the B vitamin family on mitochondrial energy metabolism. Chem-Biol Interact. 163, 94–112. https://doi.org/10.1016/j.cbi.2006.04.014 (2006).
Donnino, M. W. et al. Thiamine deficiency in critically ill patients with sepsis. J. Crit. Care. 25, 576–581. https://doi.org/10.1016/j.jcrc.2010.03.003 (2010).
Marik, P. E. Hydrocortisone, ascorbic acid and thiamine (HAT therapy) for the treatment of sepsis. Focus on ascorbic acid. Nutrients 10, 1762. https://doi.org/10.3390/nu10111762 (2018).
Manzetti, S., Zhang, J. & Van Der Spoel, D. Thiamin function, metabolism, uptake, and transport. Biochemistry 53, 821–835. https://doi.org/10.1021/bi401618y (2014).
Jhala, S. S. & Hazell, A. S. Modeling neurodegenerative disease pathophysiology in thiamine deficiency: consequences of impaired oxidative metabolism. Neurochem. Int. 58, 248–260. https://doi.org/10.1016/j.neuint.2010.11.019 (2011).
Huang, H-M., Chen, H-L. & Gibson, G. E. Thiamine and Oxidants Interact to Modify Cellular Calcium Stores. Neurochem Res. 35, 2107–2116. https://doi.org/10.1007/s11064-010-0242-z (2010).
Navarro, D., Zwingmann, C., Hazell, A. S. & Butterworth, R. F. Brain lactate synthesis in thiamine deficiency: a re-evaluation using1 H‐13 C nuclear magnetic resonance spectroscopy. J. Neurosci. Res. 79, 33–41. https://doi.org/10.1002/jnr.20290 (2005).
Desjardins, P. & Butterworth, R. F. Role of mitochondrial dysfunction and oxidative stress in the pathogenesis of selective neuronal loss in wernicke’s encephalopathy. Mol. Neurobiol. 31, 17–25. https://doi.org/10.1385/MN:31:1-3:017 (2005).
Chan, H., Butterworth, R. F. & Hazell, A. S. Primary cultures of rat astrocytes respond to thiamine deficiency-induced swelling by downregulating aquaporin-4 levels. Neurosci. Lett. 366, 231–234. https://doi.org/10.1016/j.neulet.2004.04.095 (2004).
Hazell, A. S. & Butterworth, R. F. Update of cell damage mechanisms in thiamine deficiency: focus on oxidative stress, excitotoxicity and inflammation. Alcohol Alcohol (oxf Oxfs). 44, 141–147. https://doi.org/10.1093/alcalc/agn120 (2009).
Schenker, S., Henderson, G. I., Hoyumpa, A. M. & McCandless, D. W. Hepatic and wernicke’s encephalopathies: current concepts of pathogenesis. Am. J. Clin. Nutr. 33, 2719–2726. https://doi.org/10.1093/ajcn/33.12.2719 (1980).
Moskowitz, A. & Donnino, M. W. Thiamine (vitamin B1) in septic shock: a targeted therapy. J. Thorac. Dis. 12, S78–S83. https://doi.org/10.21037/jtd.2019.12.82 (2020).
Sahakian, Y. & Dyakov, D. The effects of thiamine on patients with sepsis and septic shock. J. Emerg. Crit. Care Med. 6, 1–1. https://doi.org/10.21037/jeccm-21-74 (2022).
Nisar, S. et al. Thiamine deficiency-related neuropathy: A reversible entity from an endemic area. Eur. J. Neurol. 31, e16155. https://doi.org/10.1111/ene.16155 (2024).
Serra, M. et al. A systematic review of thiamine supplementation in improving diabetes and its related cardiovascular dysfunction. Int. J. Mol. Sci. 26, 3932. https://doi.org/10.3390/ijms26093932 (2025).
Huang, P., Chen, J., Deng, Z. & Yang, W. Thiamine deficiency secondary to intestinal surgery leading to shoshin beriberi and Wernicke encephalopathy: a case report. Front. Pediatr. 13 https://doi.org/10.3389/fped.2025.1576805 (2025).
Li, S. & Xing, C. Wernicke encephalopathy: a mini review of the clinical spectrum, atypical manifestations, and diagnostic challenges. Front. Neurol. 16, 1566366. https://doi.org/10.3389/fneur.2025.1566366 (2025).
Osiezagha K, Ali S, Freeman C, et al. Thiamine deficiency and delirium. Innov ClinNeurosci10 ,(4):26–32.https://pmc.ncbi.nlm.nih.gov/articles/PMC3659035/ (2013).
Berg, R. M. G., Møller, K. & Bailey, D. M. Neuro-oxidative-nitrosative stress in sepsis. J. Cereb. Blood Flow. Metab: Off J. Int. Soc. Cereb. Blood Flow. Metab. 31, 1532–1544. https://doi.org/10.1038/jcbfm.2011.48 (2011).
Heming, N. et al. Neuroanatomy of sepsis-associated encephalopathy. Crit. Care. 21, 65. https://doi.org/10.1186/s13054-017-1643-z (2017).
Moraes, C. et al. Neuroinflammation in sepsis: molecular pathways of microglia activation. Pharmaceuticals 14, 416. https://doi.org/10.3390/ph14050416 (2021).
Kann, O. Lactate as a supplemental fuel for synaptic transmission and neuronal network oscillations: potentials and limitations. J. Neurochem. 168, 608–631. https://doi.org/10.1111/jnc.15867 (2024).
Xiang, Z-M. & Bergold, P. J. Synaptic depression and neuronal loss in transiently acidic hippocampal slice cultures. Brain Res. 881, 77–87. https://doi.org/10.1016/S0006-8993(00)02795-5 (2000).
Sedhai, Y. R. et al. Effect of thiamine supplementation in critically ill patients: a systematic review and meta-analysis. J. Crit. Care. 65, 104–115. https://doi.org/10.1016/j.jcrc.2021.05.016 (2021).
Park, J. E. et al. Impact of vitamin C and thiamine administration on delirium-free days in patients with septic shock. J. Clin. Med. 9, 193. https://doi.org/10.3390/jcm9010193 (2020).
Stuart, E. A. Matching methods for causal inference: a review and a look forward. Stat. Sci. 25 https://doi.org/10.1214/09-STS313 (2010).
Zhao, Q-Y. et al. Propensity score matching with R: conventional methods and new features. Ann. Transl Med. 9, 812–812. https://doi.org/10.21037/atm-20-3998 (2021).
Li, F., Zaslavsky, A. M. & Landrum, M. B. Propensity score weighting with multilevel data. Stat. Med. 32, 3373–3387. https://doi.org/10.1002/sim.5786 (2013).
Innocenti, F., Candel, M. J. J. M., Tan, F. E. S. & van Breukelen, G. J. P. Relative efficiencies of two-stage sampling schemes for mean estimation in multilevel populations when cluster size is informative. Stat. Med. 38, 1817–1834. https://doi.org/10.1002/sim.8070 (2019).
Acknowledgements
We would like to thank the Massachusetts Institute of Technology and the Beth Israel Deaconess Medical Center for the MIMIC-IV database. We are grateful for the guidance on database extraction provided by Dr. Yang Qilin from the Second Affiliated Hospital of Guangzhou Medical University.
Funding
This study was not supported by any targeted funding from public sector agencies, commercial entities, or nonprofit organizations.
Author information
Authors and Affiliations
Contributions
Haibo Wang 1† , Yaotang Wang 2† , Caixia Li 3* , and Ruimin Hu 4Co-First Authors (†):Haibo Wang and Yaotang Wang contributed equally to core research execution (study design, data acquisition, analysis, and manuscript drafting).Corresponding Author (*):Caixia Li oversaw project coordination, critical revision of the manuscript, and acts as the primary contact for communication.Ruimin Hu finished technical assistance, resources provision and data curation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
MIMIC-IV is a de-identified, publicly available database approved by the institutional review boards of Beth Israel Deaconess Medical Center and the Massachusetts Institute of Technology. Given the de-identified nature of the data, the requirement for informed consent was waived. Co-first author Yaotang Wang obtained access to the MIMIC-IV database after completing the required training on the National Institutes of Health (NIH) platform, including the courses “Study data or Specimens only” and “Conflict of interest” (certification numbers: 59979404 and 59979406).
Consent for publication
All authors have approved the manuscript and have provided consent for submission to the journal.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, H., Wang, Y., Li, C. et al. Association of thiamine supplementation with 30-day mortality among ICU patients with sepsis-associated delirium. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44384-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44384-w