Abstract
Bone regeneration of oral and maxillofacial defects in situ remains a significant clinical challenge. This study investigates the synergistic effects of phosphorus (P) and zinc (Zn) co-doping in wollastonite bioceramics to enhance osteogenic activity and antibacterial potential for bone repair. Cylindrical bioceramic granules with varying P/Zn ratios (CSi-P8: CSi-Zn6 = 3:0, 2:1, 1:2 and 0:3) were fabricated via co-precipitation and evaluated for in-vitro bioactivity, cell proliferation, osteogenic differentiation, and antibacterial performance. The experimental results demonstrated that the co-doped groups (8P/6Zn-2:1 and 8P/6Zn-1:2) exhibited superior hydroxyapatite formation in simulated body fluid (SBF), sustained Ca²⁺/Si⁴⁺ ion release, and synergistic osteogenic effects, as evidenced by elevated RUNX2/β-catenin expression and BMSC proliferation. Zn doping conferred dose-dependent antibacterial activity against Staphylococcus aureus (93% inhibition for 8P/6Zn-0:3). In vivo cranial defect experiments in rabbits revealed that the 8P/6Zn-2:1 granules significantly promoted new bone trabeculae formation and defect closure within 16 weeks, outperforming single-doped and control groups. Histological and micro-CT analyses confirmed enhanced bone volume fraction (BV/TV%) and trabecular density (Tb.N) in the co-doped groups. These findings highlight the dual functionality of P/Zn co-doped wollastonite (e.g. 8P/6Zn-2:1), where P enhances osteogenesis via increasing the expression of RUNX2/β-catenin and apatite deposition, while Zn provides antibacterial protection, offering more promising implants for infected oral bone defects.
Similar content being viewed by others
Introduction
For common conditions in oral diseases, whether it’s bone resorption caused by periodontitis or maxillofacial bone defects resulting from trauma, the key to treatment lies in the repair of bone tissue. Clinically, for large-volume bone defects that are difficult to heal spontaneously, there are two primary treatment approaches. The first is autologous/xenogeneic bone grafting. For example, bone defects formed after oral and maxillofacial surgical resection are challenging to repair spontaneously. Clinically, vascularized bone flap grafting is considered the clinical gold standard for reconstructing maxillofacial bone defects. However, its application is limited by drawbacks such as limited graft volume, the necessity of creating a secondary surgical site, donor site morbidity (including pain and bleeding), and difficulties in contouring. The second approach involves 3D-printed bioactive scaffolds and titanium alloy implants. For instance, the use of dental implants in alveolar ridge rehabilitation is a common practice1,2. Achieving optimal dental implant outcomes requires a sufficient quantity and quality of newly formed bone to provide adequate mechanical support for the implant fixture. However, augmenting alveolar ridge volume and height within a narrow therapeutic window to enable successful implant placement presents a significant challenge. Bio-ceramic grafts and tissue regeneration techniques play a critical role in addressing this challenge3,4.
In fact, the bioactive stimulation of bone implants depends not only on the material properties but also on the material’s biodegradation (ion release) behavior and its spatiotemporal evolution. The former is influenced by compositional properties, while the latter’s morphological properties depend on the material’s macro/microstructure. Among the primary structural parameters, pore size, pore interconnectivity, and surface microstructure are directly correlated with osteogenic activity and bone mineralization5,6. From a biological perspective, the composition of bioceramics has been demonstrated to significantly influence adequate tissue integration and bone regeneration7,8,9. Concurrently, doping bioceramics with different ionic constituents can modulate their osteogenic activity and antibacterial activity. Research on bioceramics is increasingly focused on their ability to release bioactive ions (e.g., Ca²⁺, Si⁴⁺, Mg²⁺, Sr²⁺) to trigger corresponding biological effects10. These controlled-release bioactive ions possess the potential to act as functional substitutes for certain biomolecules, including growth factors11.
Calcium silicate-based bioceramics are regarded as highly promising bone repair materials due to their excellent bioactivity12. Calcium silicate compounds universally possess the ability to form a hydroxyapatite-like layer in SBF13. Compared to calcium phosphate-based ceramics, wollastonite (a key calcium silicate phase) exhibits superior fracture toughness and flexural strength. Furthermore, its broader range of chemical compositions enables the enhancement of its mechanical properties through more diverse approaches, such as ionic doping or the introduction of a second phase. The potential of calcium silicate-based bioceramics as promising bone substitutes is closely linked to their ability to release Ca²⁺ and Si⁴⁺ ions. Calcium constitutes approximately 2% of the human body, with the vast majority (98%) residing within the skeleton. It maintains a homeostatic equilibrium with calcium present in the blood and tissues, playing a crucial role in vascular development and bone growth14.
Silicon, an essential trace element in the human body, plays a crucial role in the normal development of connective tissues and bone15,16. Benefiting from the compositional diversity of calcium silicate-based bioceramics, the mechanical properties of wollastonite can be enhanced by introducing trace dopant elements. Relevant experiments indicate that incorporating elements such as Mg, Zn, and Sr into the CaO-SiO₂ structure can significantly improve the material’s mechanical properties17. For instance, introducing zinc into the wollastonite system generates new phase compositions, with hardystonite (Ca₂ZnSi₂O₇) emerging as a representative mineral phase within this newly formed system. Related research demonstrates that, compared to wollastonite, hardystonite exhibits a more pronounced stimulatory effect on cell proliferation and differentiation18. Hardystonite also possesses excellent antibacterial properties. Li et al. introduced a hardystonite coating onto a Ti-6Al-4 V substrate using plasma spray coating technology; this coating achieved a 93% antibacterial rate against Staphylococcus aureus19. In the treatment of burns, it has been found that hardystonite promotes proliferation of endothelial cells and wound healing through the synergistic effect of the released Zn²⁺ and Si⁴⁺ ions20.
Phosphorus is the fundamental component of bone minerals such as hydroxyapatite. The latest research has revealed that phosphorus is not only a key biological signaling molecule but also plays a crucial role in regulating cellular behavior21.The extracellular phosphorus level can activate specific cellular signaling pathways (such as FGFR/Raf/MEK/ERK), regulating the gene expression, differentiation and mineralization of osteoblasts. Moreover, phosphorus in the form of polyphosphate can promote cells to produce energy for bone repair. The phosphorus doped wollastonite nanomaterials prepared through biomimetic mineralization have highly biomimetic components and structures, providing an ideal growth environment for bone cells22. Recently, some researchers have utilized polyurethane-urea based electrospun nanofibrous scaffold, combined with functional nano-hydroxyapatite, to promote the regeneration of bone tissue, and achieved excellent repair results23.
Building upon the aforementioned research status and progress, we designed wollastonite doped with varying ratios of zinc and phosphorus. This design aimed to investigate the compositional distribution during the in vitro biodegradation of the materials and the activity of ion release, and to explore the osteogenic activity and antibacterial properties of each material group at the cellular level. Furthermore, the study sought to evaluate the potential of phosphorus- and zinc-doped wollastonite with different doping ratios as bone repair implants for the complex bacterial microenvironment of the oral cavity. This research demonstrates that doping bioceramic composites with diverse ions facilitates the modulation of new bone growth while concurrently maintaining a certain level of antibacterial performance. It provides novel insights for the design of innovative biomaterials conducive to oral bone repair.
Experiments and method
Materials and reagents
The inorganic salt reagents, carboxymethyl cellulose (CMC) and carboxylate chitosan (CC; degree of deacetylation > 90%) were brought from Sinopharm Chemical Reagent Co., Shanghai China. Besides, tris(hydroxymethyl) aminomethane (Tris) and phosphate buffer solution (PBS) were purchased from Sigma–Aldrich (St. Louis, MO, USA). The all reagents were all used directly without further purification. Bone Mesenchymal Stem Cells (BMSCs) used in this experiment were isolated from the femoral bone marrow of 4-week-old Sprague Dawley rats. The rats were purchased from Slack Laboratory Animal Co., Ltd (Shanghai, China). High-glucose Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco, USA) was used for the initial culture and expansion of BMSCs. Cell Counting Kit-8 (CCK-8) reagent was purchased from Biosharp Life Sciences (Hefei, China). The following primary antibodies were used for Western blot analysis: anti-LRP5 (clone D80F2; Cell Signaling Technology, #5731), anti-RUNX2 (clone D1H7; Cell Signaling Technology, #8486), anti-β-catenin (Cell Signaling Technology, #8480), anti-SP7 (Abcam, ab209484), and anti-β-Actin (Sigma-Aldrich, #A1978). The β-Actin antibody served as a loading control. Antibody dilutions were prepared according to the manufacturers’ protocols and optimized for specific experimental conditions. Staphylococcus aureus (S. aureus, ATCC 25923) used in this experiment was purchased from a relevant strain preservation center. LB medium was used for the routine culture and expansion of S. aureus. Its formula is: 10 g/L tryptone, 5 g/L yeast extract, and 10 g/L sodium chloride, with the pH adjusted to 7.0-7.2. It was used after high-pressure sterilization. Twenty-four 4-month-old New Zealand rabbits (male, 3.2–3.5 kg) were purchased from Slack Laboratory Animal Co., Ltd (Shanghai, China) and used for in vivo cranial defect experiments.
Preparation of wollastonite ceramic granules
The 8 mol% P and 6 mol% Zn doping into CSi (signed as CSi-P8 and CSi-Zn6) powders were firstly obtained by the conventional co-precipitation method24. After that, these powders were mixed with anhydrous ethanol and ground with zirconia ball in grounding media for 6 h. The average particle size of the superfine powder was below 5 μm after dried and these particles were respectively added into the mixture solutions composed of 1.5 wt% carboxymethyl cellulose (CMC) and 7.0 wt% carboxylate chitosan (CC) with the ratio of 2:2:1 in mass.
The two hybrid slurries were mixed at the corresponding proportions (CSi-P8:CSi-Zn6 = 3:0, 2:1, 1:2 and 0:3) respectively and then transferred into four 10 mL syringes and fixed to the nozzle system. The same diameter ratio was prepared by extruding the slurries through the tunnels of nozzles (Ø1.69 mm) and then followed by calcium-ion-containing solution collection, as shown by Fig. 1A. These fibers were immersed in the collection for 6 h and then cut into cylindrical granules with the length of 2.0 mm approximately. The four groups of granules were dried in a 120 ℃ oven and eventually sintered at 1100℃ for 3 h. The four groups of fibrous granules were signed as 8P/6Zn-3:0, 8P/6Zn-2:1, 8P/6Zn-1:2, 8P/6Zn-0:3, respectively.
Primary characterizations of the granules
The phases of granules were characterized from 10° to 60°/2θ in steps of 0.026°/2θ using a PANalytical X’Pert PRO X-ray diffractometer (Holland) with a CuKα anode (40 kV ,40 mA). The fibrous granules after sintering were observed by using a field emission scanning electron microscopy (FE-SEM, Gemini Sigma 300, Zeiss, Germany) with an accelerating voltage of 3 kV. The face-scanning energy dispersive spectroscopy (EDS) analysis were used to evaluate the elemental composition of the nCSi ceramic Granules.
In vitro bio-degradation, ion release and mechanical analysis
The bioceramic granules (1 g; n = 4) was submerged with the 20mL Tris buffer and an initial pH ~ 7.40 at 37℃. The weight loss during bio-degradation test were obtained for determining of the the stability of the granule at 1, 3, 5, and 7 days. To investigate the bioactive inorganic ion release of Zn 2+, Ca2+, PO43− and SiO32− from the bioceramic fibrous granules at the pre-set time intervals, the Tris supernatant (1.0 mL) was centrifuged and followed by diluting with 5% HCl solution for Ca, Zn, Si, and P determination using inductive coupled plasma atomic emission spectroscopy (ICP-AES; Thermo), and then the same amount of fresh buffer was added. The uniaxial compression tests of the granules were evaluated by a universal testing machine (Instron, 5848 Micro Tester) with a 10 KN load cell and a crosshead-loading rate of 1.0 mm·min− 1.
In vitro biomimetic re-mineralization experiment
The in vitro bioactivity (biomimetic re-mineralization) of the granules was immersed in a SBF prepared on the basis of the procedure shown by Kokubo25. The granules (1.0 g; n = 4) were fully immersed in 10 mL SBF for 7 and 14 days in 37℃ incubator, respectively. The SBF was replaced every 2 days in the immersion period. At the 0, 1 and 2 weeks, the granules were rinsed with diH2O (deionized water) and absolute ethanol for three times separately. The deposited apatite layers of those immersed granules was visualized using SEM and the Ca/P ratios were conducted by quantitative EDS measurement.
Cell and antibacterial tests in vitro
The BMSCs culture preparation
The Bone Mesenchymal Stem Cells (BMSCs) were isolated from the femoral bone marrow of 4-week-old Sprague-Dawley rats. All methods were performed in accordance with the relevant guidelines and regulations. The experiment was approved by the Ethics Committee of Zhejiang University (Ethics Approval Number: ZJU21202), according to the ARRIVE guidelines (PLoS Bio 8(6), e1000412,2010). All rats were euthanized by intraperitoneal injection of an overdose of 3% sodium pentobarbital (30 mg/kg body weight) in accordance with the American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals (2020). After euthanasia, femurs and tibias of these rats were removed and trepanned, then the bone marrow was flushed out with Alpha Minimum Essential Medium (AMEM) through a syringe. After isolated, the BMSCs were cultured in the high-glucose Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with the 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin.The cultivation was carried out in an environment with 5% CO₂ and an incubation condition at 37℃. The culture medium was replaced every two days.
Cell proliferation testing
Each of the four groups of materials was incubated in 37 °C shaker at a concentration of 100 mg/mL for a duration of 48 h, and the extract solution was collected for sterilization and reserve for use. The concentration of the solution would be diluted to 1/4, 1/8, 1/16 and 1/32 using culture medium, respectively. Then, 1 × 104cultured BMSCs were seeded onto each scaffold in a 96-well plate and the culture medium was changed every two days. The Cells incubated in a culture medium were used as the control group. A cell counting kit-8 was used to determine cell proliferation after 1, 3, and 7 days of culture. Then the 10 µL CCK-8 solution was added to each well, followed by incubation at 37℃ for 2 h. Lastly, the 100 µL of the supernatant was transferred to a 96-well plate and the absorbance at 450 nm was measured by the Multi-Mode microplate reader (SpectraMax i3x, Molecular Devices, CA) to observe cell proliferation.
Osteogenic differentiation testing and Alizarin Red S staining
To test the effects of different materials solution on the osteogenic differentiation of BMSCs, the cells were seeded on 1/8 and 1/16 culture medium concentration and cultured for 7 days to extract proteins and total RNA firstly. The expression levels of osteogenesis-related proteins (LRP5, SP7, β-Catenin and Runx-2) and genes (LRP5, Runx-2, OCN and BMP2) in BMSCs were tested by Western blot and real-time polymerase chain reaction (RT-PCR), respectively. The culture medium (Control) was designed as the negative control.
ARS staining study was performed to was performed to visually assess the formation of mineralized nodules during the osteogenic differentiation testing. Firstly, we seeded BMSCs at an appropriate density (5 × 104\(\text{c}\text{e}\text{l}\text{l}\text{s}\)) into a 12-well plate. When the cell confluence reaches 60–70%, replace the medium with osteogenic induction medium (basal medium + 10% FBS + 50 µg/mL ascorbic acid + 10 mM β-glycerophosphate sodium + 10 nM dexamethasone). Change the medium every 2–3 days and continue the induction for 21 days. Remove the medium, rinse the cells with PBS, and fix the cells with 4% paraformaldehyde for 30 min. Then, add 1 mL of Alizarin Red S staining solution per well and incubate at room temperature in the dark for 30 min.
Antibacterial rates
The Staphylococcus aureus (S. aureus, ATCC 25923) was incubated on plates (n = 5) at 37 °C for 12 h and then the plates were rinsed by 2.0 mL sterile PBS by ultrasonic vibration for three times. After being serially diluted \({10}^{6}\) folds, the bacterial solutions were mixed with the extract solutions of 1/16 concentration of the four groups of materials, respectively, then 100 µL of the mix solution were inoculated onto LB plates to culture for 24 h at 37 °C. The results of the LB plates were toke by ordinary camera and the number of the colonies was calculated after incubation. The antibacterial rate was conducted by the following equation:
where A and B represent the CFU counts of control group and experimental group, respectively.
Critical size cranial parietal bone defects model evaluation in vivo
To verify the influence of different doping ions proportion on osteogenic properties in vivo, twenty-four male New Zealand rabbits (3.2 ~ 3.5 kg, 4 months old) were purchased for cranial parietal bone implantation experiments, which were randomly divided into two groups for 8 and 16-week time. All methods were performed in accordance with the relevant guidelines and regulations. The experiment was approved by the Ethics Committee of Zhejiang University (Ethics Approval Number: ZJU20220475), also strictly according to the ARRIVE guidelines (PLoS Bio 8(6), e1000412,2010). Firstly, the rabbits were anesthetized by auricular vein injection of sodium pentobarbital (1 mg/kg, Sigma) in advance and then four critical-sized cylindrical defects (Ø ~5.0 × 5.0 mm) were caused by dental trephine (Ø 5 mm) in cranial parietal bone on each rabbit. The four groups of bioceramic fibrous granules (diameter: ~1.4 mm; height: ~1.3 mm) were filled into the bone defects (23 ~ 25 in amount), respectively. The incision was covered by applying penicillin powder after implantation for anti-infection. In the following 3 days, these animals would receive the intramuscular injection of penicillin daily to prevent from infection. At 6 and 18 weeks, these rabbits were euthanized by injection of an overdose of 3% pentobarbital sodium in accordance with the AVMA Guidelines for the Euthanasia of Animals (2020) and then those cranial parietal bone specimens were collected and fixed in the paraformaldehyde fixation solution.
X-ray radiological examination and micro-CT analysis
The bone regeneration at the bone defect site was observed by X-rays (XPERT; KUBTEC Co., USA). After the cranial parietal bone specimens containing materials were retrieved, the X-ray images were taken at 200 µA and 45 kV to evaluate the mineral deposition, qualitatively. In order to assess the distribution and volume of newly formed bone, the images were taken by Micro-computed tomography (µCT) with the µCT imaging system (AX2000 CT scanner, Always Imaging, Shanghai). Each specimen was scanned for 5 min (voltage 120 kV, current 100 µA) along the anatomical axis of the cranial parietal bone at ambient temperature, the Microfocus X-ray Source (FineTec 160 kV, Germany) along with an X-ray detector (NDT 1717 M; pixel size:139 μm; pixel number: 3072 × 3072: voxel size: 14 μm) help to generate continuous cross-sectional images (image resolution:15.6 μm; time of exposure:500 ms). The cylindrical region of interest (ROI; Ø5 × 5 mm) centering on the bone defect region was set, the newly formed bone tissue in the ROI was determined and distinguished (from biomaterials) using quantitative image analysis based on the density difference. The certain trabecular morphological parameters like bone volume fraction (BV/TV%) and trabecular number (Tb.N; 1/mm) were calculated using VG Studio MAX.
Histological progressing
The granule-filled cranial parietal specimens were incubated in 10% buffered formaldehyde over one week for fixation and then rinsed with pure water overnight. After dehydration in a graded series of ethanol (70%, 80%, 95%, 100%, 100%, 100%), these specimens were cleared with Xylene and embedded in polymethyl methacrylate (PMMA), dibutyl phthalate and bis(tert-butyldioxyisopropyl) benzene. After polymerization, the specimens were divided into four parts according to different material granule filled areas. Along the anatomical axis of the cranial parietal bone by means of a saw microtome (Leica, SP1600, Nussloch, Germany), each specimen which contains the bone tissue and implants were cut into 6 ~ 8 parts as thin section for further study, with each section having a thickness of 5 μm. To prevent from the impurities and scratches, these thin sections were finely ground through a polishing machine (MP-2B), after that the sections were stained with H&E and Masson trichrome staining.
Statistic analysis
The numerical data were shown as the mean value ± SD (standard deviation) and some quantitative data was compared and analyzed by using one-way analysis of variance (ANOVA). The value of p < 0.05 was considered to be significant. The SPSS 19.0 software (SPSS, Chicago, IL, USA) was used for all statistical analysis.
Results
Primary characterization of the bioceramic granules
According to the quantitative ICP analysis results, the as-calcined bioceramic granules indicated that the Zn/Ca and P/Si molar ratio was 6.10% and 7.85% for the CSi-Zn6 and CSi-P8, respectively. Both the Zn/Ca ratio (6.10%) and the P/Si ratio (7.85%) were basically consistent with the designed ratio. As expected, the bioceramic fibers were prepared and then as-cut cylindrical granules could be easily prepared through our self-designed nozzles and cut system and followed by sintering treatment (Fig. 1A). Besides, the decrease in diameter of the bioceramic fibers were easily observed during sintering treatment (Fig. 1B). Because the main components of the four group materials were consistent, the diameter between groups was no significant difference during thermal treatment. The XRD patterns of four groups of bioceramic granules were showed in Fig. 1C. For the XRD results of CSi with foreign ion-doped (8P/6Zn-3:0, 8P/6Zn-2:1, 8P/6Zn-1:2, 8P/6Zn-0:3), the phase presenting was mainly consistent with the phase of CSi whose peaks were at 27.88° and 29.87°/2θ, suggesting that the original crystalline phase was preserved. Also, the XRD results about foreign ion-doped CSi components were highly matched to standard values (PDF#27–0088).
FE-SEM/EDS analysis of the bioceramic granules
The SEM images were used to identify the component distribution of the bioceramic granules after sintering. As shown in Fig. 1D, the SEM images (×50 magnification) indicated the distinct granular and powder structures in the surface of the samples. However, the superfine bioceramic grains among four groups could hardly be distinguished from each other because of the same mainly composition of CSi grains, the difference caused by foreign ion-doped was barely be observed. To distinguishing the different foreign ion-doped compositions, the face-scanning EDS analysis was employed to determine the elemental distribution on the fracture surface of the nCSi granules (Fig. 2). Along the diametrical line to collect characteristic X rays of the specific elements, the line-scanning analysis for the relative content was conducted by directing the electron beam to the surface. With the percentage of CSi-Zn6 and CSi-P8 change, the EDS images showed that the corresponding changes in the content of zinc and P element while the content of Ca and Si elements were no obviously significant.
Bio-degradation and mechanical properties of the granules in vitro
The bioceramic granules were submerged in Tris buffer to present the biodegradation process in vivo (Fig. 3A). The mechanical properties of the granule were evaluated by a universal testing machine during the compression tests as indicated in Fig. 3B. As indicating by the mechanical test, Zn-doping wollastonite(8P/6Zn-1:2, 8P/6Zn-0:3)could enhance the sintering properties, retard bio-dissolution in vitro and compressive strength could be improved significantly. In contrast, P-doping may inhibit the sintering and mechanical property of wollastonite bioceramic, but could enhance the biologically active response in osteogenic cell and bone defect conditions.
The weight loss analysis of these bioceramic granules was shown in Fig. 3C to determine their bio-degradation properties. In order to evaluate the in vitro degradation properties of these ceramics, the granules were immersed (1 g; n = 4) was submerged with the 20mL Tris buffer and an initial pH ~ 7.40 at 37℃ for 1, 3, 5, and 7 days respectively. Zn-doped wollastonite significantly controls the overly rapid degradation of pure wollastonite, but P-doping can promote the degradation of wollastonite. Therefore, the combined particles of Zn and P-doped wollastonite have good sinterability and degradation properties that are more suitable for tissue regeneration. The concentration curves of calcium, silicon, zinc and phosphorus ions were shown in the Fig. 3B-E. For the curves of calcium ion, the groups of nCSi co-doped with Zn and P showed better ion release activity, while the materials doped with Zn alone had the worst ion release activity. Besides, it was worth mentioning that the zinc and phosphorus ions release curves of four groups exhibited the different release rate dependent on the doping ratio. In addition, for the release curves of silicon ions, except for the lower release rate of the 8P/6Zn-0:3 group of materials, all the other materials showed similar ion release activities. In summary, compared to doping with zinc and phosphorus alone, the simultaneous doping of two ions was more beneficial to enhance the biological release activity of the wollastonite materials.
In vitro bioactivity (apatite mineralization) evaluation
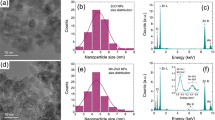
The four groups of CSi granules were submerged in the SBF for one and two weeks, respectively. And then the surface of submerged materials was examined by SEM to detect the newly formed apatite. The SEM images (Fig. 4) at high magnification (×10 000) showed that some apatite-like substance accumulating on the surface was easily found in the form of microspheres. At one week after immersion, the apatite-like grains could be clearly found among 8P/6Zn-3:0, 8P/6Zn-2:1 and 8P/6Zn-1:2 groups while the grain was barely observed in the 8P/6Zn-0:3 group. At two weeks, there were more apatite-like grains on the surface of four materials. Among the four groups, the 8P/6Zn-1:2 granules had the widest coverage and the densest accumulation degree while the number of apatite-like substances in 8P/6Zn-0:3 group was the least. As indicated by the EDS analysis, the Ca/P ratios in the surface layer of four groups were 8P/6Zn-3:0(1.47), 8P/6Zn-2:1(1.54), 8P/6Zn-1:2(1.65) and 8P/6Zn-0:3(1.82), those were close to the Ca/P ratio of hydroxyapatite.
Cell proliferation, osteogenic differentiation testing
For the results of CCK-8 testing, we found the groups of 1/8 and 1/16 concentrations showed good cell proliferation effect (Fig. 5A). So we used these two materials leaching solution concentrations for treatment in the following experiments. The Western blot results showed that the 8P/6Zn-2:1 group have the high expression of RUNX2 and β-catenin proteins (Fig. 5B), which demonstrated its good osteogenic ability while the group of 8P/6Zn-3:0 also had good performance. Besides, the results of qPCR also exhibited that the expression of osteogenesis-related RNA in group of 8P/6Zn-2:1 was better than other groups (Fig. 5C). In addition, in the anti-bacterial test, it was easy to find that, with the increase of the proportion of zinc in the material, the material showed better antibacterial effect (Fig. 5E).
After osteogenic induction of BMSCs using a 1/16 concentration of material leachate, ARS staining was performed to visually assess the formation of mineralized nodules. As indicated by Fig. 5D, compared to other groups, the groups of 8P/6Zn-2:1 and 8P/6Zn-1:2 exhibited somewhat higher staining intensity and nodule formation. In contrast, the control group, which did not receive material leachate treatment, showed a slightly lower degree and density of staining compared to the other groups. This further underscores the osteoinductive effect of the materials. In summary, the Alizarin Red S staining results suggest a potential enhancement in the osteogenic differentiation capacity of BMSCs induced by the 8P/6Zn-2:1 material leachate, which is consistent with the molecular findings from Western blot and qPCR analyses.
Systematical characterization of the granules in cranial parietal bone defects
Radiological examination by X-ray scanning and µCT analysis
The critical size cranial parietal bone defects animal model involving the materials granule implantation was shown in Fig. 6A-B. The roentgenographic analysis showed that the granules were implanted in the expected position (Fig. 6B).
To evaluate the repair process of the cranial parietal bone repair, the 2D/3D µCT analysis was performed at 8 and 16 weeks (Fig. 6C). The results showed that, at 8 weeks, except for the 8P/6Zn-3:0 group, some new bone could be observed clearly growing into the pores of the filling materials among other groups. And compared with the 8P/6Zn-0:3 group, the materials of 8P/6Zn-2:1 and 8P/6Zn-1:2 had better effect of promoting the new bone growth. At 16 weeks, the bone defects in the 8P/6Zn-2:1 and 8P/6Zn-1:2 groups filled with new bone and undegraded granules, the defects were almost invisible. Besides, the remaining materials were tightly integrated with new bone tissue among four groups.
The trabecular morphological parameters were shown in the Fig. 6D. The quantitative data was consistent with the µCT photographs as the trabecular number and bone volume fraction. At 8 weeks, the bone volume fraction of 8P/6Zn-0:3 group showed the significant difference with the other groups and the trabecular number of four groups were not significant difference. Besides, with the prolongation of repair time to 16 weeks, both the bone volume fraction and the trabecular number of 8P/6Zn-2:1 group exhibited the significant difference with others, implying the granules had better osteogenic activity.
Histological progressing
Figures 7 and 8 showed the results of the implantation biological ceramic granules and bone interface layer of HE and Masson staining histological appearance after 8 and 16 weeks. At 8 weeks, the structure of 8P/6Zn-3:0 particles were degraded, which was manifested by absorption vacuoles at the edges of the particles. While there was not significant degradation of others which were doped with Zn ions. Besides, the experimental phenomenon of some new bone ingrowth material clearance was only observed in 8P/6Zn-2:1 group while other groups were some fibrous tissues in the void. At 16 weeks, under high magnification (×40), there was a great number of new bone surrounding the 8P/6Zn-2:1 particle and the boundary between bone and particle was unclear. Besides the 8P/6Zn-3:0 particles showed good biodegradability and there were some new bone and fibrous tissue surrounding the granules but not close to it. In addition, although there were some new bones around the 8P/6Zn-1:2 particles, the fibrous tissue was more tightly bound to the materials. For the group of 8P/6Zn-0:3, the new bone was barely observed around the particle.
Schematic illustration of bioceramic granules preparation, primary characterization of the bioceramic granules and SEM images of the bioceramic granules after sintering. (A) Schematic procedure of bioceramic fiber precursor preparation, Figures were created with BioRender.com; (B) Changes in diameter of the fibrous granules; (C) XRD patterns of ion-doped CSi granules; (D) SEM images of the bioceramic powders after sintering.
Composition analysis by EDS mapping for the fibrous bioceramic granule.
The degradation evaluation in Tris buffer in vitro. (A) Schematic diagram of the ion release test; (B) Compressive strength under the uniaxial compression test; (C) Weight loss during the degradation test in vitro; (D–G) Changes of Ca, P, Zn, and Si concentrations in the Tris buffer with time.
SEM images of the bioceramic granules after immersion in SBF (1.0 g/10 ml) for 0, 7, and 14 days, respectively. The Ca/P ratios were conducted by quantitative EDS measurement as shown in red.
(A) The viability of BMSCs cultured on different concentrations of material(1/4, 1/8, 1/16 and 1/32)extract solution after 1, 3, and 7 days measured by CCK-8; (B) Expression of osteogenic proteins (LRP5, Runx-2,β-catenin and SP7) in BMSCs following a 7-day osteogenic incubation on different concentrations of material (1/8 and 1/16) extract solution; (C) Relative mRNA expression levels (normalized to β-actin) of osteogenesis-related genes (LRP5, OCN, BMP-2 and Runx-2) in BMSCs following a 7-day osteogenic incubation on different concentrations of material༈1/8 and 1/16༉extract solution; (D) the Alizarin Red S staining (ARS) performed to visually assess the formation of mineralized nodules; (E) Spread plate images of S. aureus seeding on the surface of corresponding plates with 1/16 concentrations of material extract solution. Quantitative statistics of colony count of the four groups materials against. All data are expressed as the mean ± SD. Different letters represent significant differences between groups at the same time point, n = 6; *p<0.05; **p<0.01; ***p<0.001. Group: A:8P/6Zn-3:0, B:8P/6Zn-2:1, C:8P/6Zn-1:2, D: 8P/6Zn-0:3.
Animal model of bone defects, 2D µCT reconstruction and quantitative analyses of cranial parietal bone defects filled with the nCSi bioceramic granules after implantation for 8 and 16 weeks. (A) Schematic illustration of the bone defect model; (B) bioceramic granules implantation in cranial parietal bone defects; (C) µCT images; (D) BV/TV (bone volume/total volume); Tb.N (trabecular number). *p <0.05; **p<0.01.
H&E staining histological evaluation (10 ×, 40 ×) of bone regeneration for four groups of nCSi bioceramic granules at 8 and 16 weeks. B: bone; F: fibrous tissue; CSi: wollastonite core; M: materials (granules).
Masson’s trichome staining histological evaluation (10×, 40×) of bone regeneration for four groups bioceramic granules at 8 and 16 weeks. B: bone; F: fibrous tissue; CSi: wollastonite core; M: materials.
Discussion
Undoubtedly, the synchronization of material biodegradation and new bone regeneration is beneficial for maintaining mechanical stability and eliciting subtle biological responses, thereby achieving harmonious bone regeneration. It is noteworthy that the bio-solubility (reflecting in vitro biodegradation) of bone tissue repair materials holds significant importance for healing at the bone defect site. Previous studies have indicated that calcium and silicon ions released from wollastonite, in conjunction with other trace metal ions, exert a synergistic effect on bone formation26. However, the accumulation of released metal ions in vivo may also potentially trigger adverse biological reactions, such as allergic responses, local necrosis, and inflammation. Consequently, biodegradation kinetics and ion release profiles should be carefully monitored and tailored to align with the bone healing process27.
Many methods have been applied to improve the properties of bioceramics. As reported, the tricalcium silicate bone cement was modified with wollastonite/fluorapatite glass and glass-ceramic to obtain outstanding biological activity, biocompatibility and antibacterial properties28. In our study, cylindrical bioceramic pellets with varying ion doping ratios were employed to modulate the biodegradation behavior of two types of bioceramic compositions. Based on experimental results, we observed that in in vitro SBF mineralization assays, the two groups co-doped with phosphorus (P) and zinc (Zn) ions demonstrated favorable mineralization outcomes. These materials generated abundant hydroxyapatite crystals within the simulated physiological fluid. This enhanced mineralization may be attributed to alterations in the crystal structure and/or crystallinity of wollastonite induced by the different ion dopants. Correlating these findings with the ion release profiles revealed distinct patterns: The group doped solely with zinc ions (designated as the 8P/6Zn-0:3 group) exhibited relatively low release levels of Ca²⁺, PO₄³⁻ (P), and Si⁴⁺ ions. Similarly, the Ca²⁺ ion release kinetics in the 8P/6Zn-3:0 group (doped solely with phosphorus) were also suboptimal. In contrast, the other two groups, co-doped with both phosphorus and zinc ions, exhibited significantly higher release activity of Ca²⁺ and Si⁴⁺ ions. This indicates a synergistic effect on ion release between P and Zn dopants.
Y. Peng et al. have shown that by adjusting the zinc content in wollastonite bioceramics, they can acquire excellent angiogenic and antibacterial properties, which is beneficial for bone repair29. Furthermore, due to the Zn doping, these co-doped groups also demonstrated measurable Zn²⁺ ion release, which is anticipated to confer a certain degree of antibacterial activity. The Zn-doped silicon carbonate significantly controls the rapid degradation of pure silicon carbonate. However, the P-doping can promote the degradation of silicon carbonate, resulting in the composite particles of Zn and P-doped silicon carbonate having excellent sintering properties and more suitable degradation characteristics and mechanical properties for tissue regeneration30. As for the observed biomimetic re-mineralization reaction on the surface of the bioceramic granules, it is obvious that the Ca/P ratio is below the theoretical ratio of chemically stoichiometric ratio of hydroxyapatite. This is mainly attributed to the SBF containing CO32− groups which would readily substitute some phosphate groups and produce Ca-deficient carbonate apatite mineral layer on the surface of the bioceramic granules. Meanwhile, the different amount of P and Zn dopants in the bioceramic granules would influence the sintering properties and bio-dissolution behavior in the SBF medium, and thus this factor also influence the biomimetic apatite layer composition, especially the Ca/P ratio.
Cellular-level studies revealed that in cell proliferation assays, the material group with higher phosphorus (P) ion doping exhibited a greater stimulatory effect on cell proliferation. However, compared to the control group, all four experimental groups demonstrated a proliferative effect. This outcome may be associated with the release of calcium (Ca²⁺) and silicon (Si⁴⁺) ions. Related research indicates that elevated extracellular calcium ion concentrations inhibit osteoclastogenesis31. Calcium ions participate in the regulation of bone repair through their dual effects on the activity and proliferation of both osteoblasts and osteoclasts during bone remodeling, acting via hormone-independent pathways32. Furthermore, Rico et al. demonstrated that silicon (Si) intake inhibits bone loss in ovariectomized (OVX) rats and promotes longitudinal growth of long bones, such as the femur33.
Gene and protein expression results indicated that: Regarding the expression levels of osteogenesis-related proteins, the 8P/6Zn-2:1 group demonstrated significant osteogenic-promoting effects at both 1/8 and 1/16 concentrations. This finding was corroborated by corresponding results at the gene expression level. Related research elucidates that calcium phosphate-based ceramics facilitate bone regeneration by releasing Ca²⁺ and PO₄³⁻ ions to form an appetite layer resembling bone mineral composition. This apatite layer subsequently adsorbs osteogenic proteins onto the material surface, thereby inducing bone formation34.
Additionally, in antibacterial assays against Staphylococcus aureus, the materials exhibited progressively higher antibacterial activity with increasing zinc (Zn) ion doping ratios, which is consistent with the anticipated effect. Staphylococcus aureus holds significant representativeness and typicality among Gram-positive bacteria. As a prevalent pathogenic bacterium, it is widely distributed in natural environments and the human body. Some researcher also used other kinds of bacterials to such as E. coli to meansure the antibacterial rates of biomaterials35. Extensive and systematic research has been conducted on its biological characteristics and pathogenic mechanisms, resulting in a wealth of experimental data and literature. This provides a solid theoretical foundation and reliable references for our research. This is highly advantageous for controlling experimental timelines and ensuring research efficiency. In subsequent studies, we plan to expand our scope by incorporating other representative bacterial species to more comprehensively and rigorously validate our findings.
Integrating the µCT analysis results from animal experiments, the 8P/6Zn-2:1 group demonstrated superior osteogenic activity, with the material exhibiting enhanced bone repair efficacy. With the development of technology, the µCT analysis could easily distinguish the wollastonite bioceramic granules or porous scaffold from bone tissue according to the assistance of professional software. Several previous studies have also been evaluated using such µCT by the high-level technical engineers36,37. Unfortunately, the cylindrical structural granules could not easily control the in vivo bioactivity (functional ion release dosage) and biodegradation simultaneously. Actually, we analyze the biodegradation performance of the materials by comparing the differences in the material residual ratios among different material groups and their changes over time. Histological staining further revealed the presence of more newly formed bone trabeculae bridging the particles within this group. These in vivo findings are largely consistent with the in vitro results, indicating that phosphorus- and zinc-doped wollastonite possesses a stronger capacity to stimulate osteogenesis and enhance bone repair/filling efficacy compared to traditional, non-doped wollastonite filling materials.
Conclusion
This study demonstrates that phosphorus (P) and zinc (Zn) co-doped wollastonite bioceramics exhibit synergistic effects in promoting bone regeneration while providing antibacterial protection, making them promising candidates for oral and maxillofacial bone repair. The co-doped groups (8P/6Zn-2:1 and 8P/6Zn-1:2) showed superior in vitro bioactivity, which correlated with upregulated expression of osteogenic markers (RUNX2, β-catenin) in BMSCs. Notably, Zn doping conferred dose-dependent antibacterial efficacy against Staphylococcus aureus. In vivo experiments in rabbit cranial defects revealed that 8P/6Zn-2:1 granules significantly accelerated bone regeneration. This study advances the design of multifunctional 8P/6Zn-2:1 bioceramics for infected or critical-sized bone defects, offering a clinically translatable alternative to traditional grafts. Future work should explore long-term biodegradation kinetics and clinical scalability.
Data availability
The data that supports the findings of this study are available within the article and its supplementary material.
References
Bali, Y., Singh, R., Gill, T. K., Rela, R. & Priyadarshni, P. Bone graft and intraosseous anchorage of dental implants for reconstruction of the residual alveolar ridge. J. Pharm. Bioallied Sc. 13 (5), 465. https://doi.org/10.4103/jpbs.JPBS-638-20 (2021).
Mahardawi, B. et al. The clinical efficacy of the allogenic demineralized dentin matrix graft for implant placement: a systematic review. Oral Maxillofac. Surg. 28 (2), 585–593. https://doi.org/10.1007/s10006-023-01170-6 (2024).
Gayathry, G., Fernandez, F. B., Harikrishna, V. P. R. & Manoj, K. Migration and retention of human osteosarcoma cells in bioceramic graft with open channel architecture designed for bone tissue engineering. BIOMED. MATER. 19 (6), 065009. https://doi.org/10.1088/1748-605X/ad792b (2024).
Fazeli, N. et al. Accelerated reconstruction of rat calvaria bone defect using 3D-printed scaffolds coated with hydroxyapatite/bioglass. Sci. Rep. 13, 12145. https://doi.org/10.1038/s41598-023-38146-1 (2023).
Gandolfi, M. G. et al. Calcium silicate and calcium hydroxide materials for pulpcapping: biointeractivity, porosity, solubility and bioactivityof current formulations. J. Appl. Biomater. Funct. Mater. 13 (1), 43–60. https://doi.org/10.5301/jabfm.5000201 (2015).
Jung, N. et al. Improving bone formation by guided bone regeneration using a collagen membrane with rhBMP-2: a novel concept. J. Funct. Biomater. 14, 170. https://doi.org/10.3390/jfb14030170 (2023).
Sun, D. et al. Citric acid-based hydroxyapatite composite scaffolds enhance calvarial regeneration. Sci. Rep. 4, 6912. https://doi.org/10.1038/srep06912 (2014).
Brochu, B. M. et al. Advances in bioceramics for bone regeneration: a narrative review. Biomimetics 9, 690. https://doi.org/10.3390/biomimetics9110690 (2024).
Wu, K. et al. Evaluation of microbial adhesion on 3d-printed zirconia surfaces: effects of printing angle and layer thickness. J. Dent. 160, 105855. https://doi.org/10.1016/j.jdent.2025.105855 (2025).
Tavoni, M., Dapporto, M. & Tampieri, A. Bioactive calcium phosphate-based composites for bone regeneration. J. COMPOS. SCI. 5, 227. https://doi.org/10.1002/adhm.201800700 (2021).
Zhou, Y., Wu, C. & Chang, J. Bioceramics to regulate stem cells and their microenvironment for tissue regeneration. Mater. Today. 24, 41–56. https://doi.org/10.1186/s13287-018-1130-8 (2019).
Kang, Z. et al. Three-dimensional printing of CaTiO3 incorporated porous β-Ca2SiO4 composite scaffolds for bone regeneration. Appl. Mater. Today. 16, 132–140. https://doi.org/10.3233/BME-151263 (2019).
Toraya, H. & Yamazaki, S. Simulated annealing structure solution of a new phase of dicalcium silicate Ca(2)SiO(4) and the mechanism of structural changes from alpha-dicalcium silicate hydrate to alpha(L)’-dicalcium silicate via the new phase. Acta Crystallogr. B. 58 (4), 613–621. https://doi.org/10.1107/s0108768102005189 (2002).
Piste, P. & Sayaji, D. Avinash M.Calcium and its role in human body. Int. J. Res. Pharm. Biomed. Sci. 4, 2229–3701. https://doi.org/10.1177/02676591211073779 (2012).
Jugdaohsingh, R. Silicon and bone health. J. Nutr. Health Aging. 11 (2), 99–110. https://doi.org/10.1016/j.jneb.2006.09.001 (2007).
Nielsen, F. H. Update on possible nutritional importance of silicon. J. Trace Elem. Med. Bio. 28 (4), 379–382. https://doi.org/10.1016/j.jtemb.2014.06.024 (2014).
Ma, J., Chen, C. Z., Wang, D. G., Jiao, Y. & Shi, J. Z. Effect of magnesia on the degradability and bioactivity of sol-gel derived SiO2-CaO-MgO-P2O5 system glasses. COLLOID Surf. B. 81 (1), 87–95. https://doi.org/10.1016/j.colsurfb.2010.06.022 (2010).
Sun, M. et al. Systematical evolution of mechanically strong 3D printed dilute magnesium doping walloastonite scaffolds on osteogenic capacity in rabbit calvarial defects. Sci. Rep. 6, 34029. https://doi.org/10.1038/srep34029 (2016).
Li, K. et al. Chemical stability and antimicrobial activity of plasma sprayed bioactive Ca2ZnSi2O7 coating. J. Mater. Sci. Mater. Med. 22 (12), 2781–2789. https://doi.org/10.1007/s10856-011-4454-9 (2011).
Zhang, Z. et al. Design of a biofluid-absorbing bioactive sandwich-structured Zn-Si bioceramic composite wound dressing for hair follicle regeneration and skin burn wound healing. Bioact Mater. 6 (7), 1910–1920. https://doi.org/10.1016/j.bioactmat.2020.12.006 (2021).
Apanasevich, V. et al. Morphological characteristics of the osteoplastic potential of synthetic CaSiO3/HAp powder biocomposite. J. Funct. Biomater. 11 (4). https://doi.org/10.3390/jfb11040068 (2020).
Wang, X. et al. Core-shell-typed selective-area ion doping wollastonite bioceramic fibers enhancing bone regeneration and repair in situ. Appl. Mater. Today. 32, 101849. https://doi.org/10.1016/j.apmt.2023.101849 (2023).
GhoraiA, S. K. et al. A judicious approach of exploiting polyurethane-urea based electrospun nanofibrous scaffold for stimulated bone tissue regeneration through functionally nobbled nanohydroxyapatite. Chem. Eng. J. 2 (429). https://doi.org/10.1016/j.cej.2021.132179 (2022).
Xie, J. et al. Simultaneous mechanical property and biodegradation improvement of wollastonite bioceramic through magnesium dilute doping. J. Mechan Behav. Biomed. Mater. 54, 60–71. https://doi.org/10.1016/j.jmbbm.2015.09.012 (2016).
Memenga-Nicksch, S., Marschner, F. & Thomas, N. H. Systematic review and meta-analysis on marginal bone loss of dental implants placed in augmented or pristine bone sites: Findings from clinical long-term studies. J. Dent. 158, 105808. https://doi.org/10.1016/j.jdent.2025.105808 (2025).
Nicolas, T. et al. Multiparametric influence of 3D-printed organo-mineral scaffolds on bone regeneration. Sci. Rep. 14, 20848. https://doi.org/10.1038/s41598-024-71698-4 (2024).
Raucci, M. G., Guarino, V. & Ambrosio, L. Biomimetic strategies for bone repair and regeneration. J. Funct. Biomater. 3, 688–705. https://doi.org/10.3390/jfb3030688 (2012).
Abd El-Hamid, H. K., Fayad, A. M. & Elwan, R. L. Evaluation of bioactivity, biocompatibility, and antibacterial properties of tricalcium silicate bone cement modified with wollastonite/ fluorapatite glass and glass-ceramic. Ceram. Int. 50 (14), 25322–25332. https://doi.org/10.1016/j.ceramint.2024.04.262 (2024).
Peng, Y. et al. Tuning zinc content in wollastonite bioceramic endowing outstanding angiogenic and antibacterial functions beneficial for orbital reconstruction. Bioactive Mater. 36, 551–564. https://doi.org/10.1016/j.bioactmat.2024.02.027 (2024).
Zhuang, C. et al. Core-shell-structured nonstoichiometric bioceramic microspheres for improving osteogenic capability. J. Mater. Chem. B. 5, 8944–8956. https://doi.org/10.1039/c7tb02295f (2017).
Cianferotti, L., Gomes, A. R., Fabbri, S., Tanini, A. & Brandi, M. L. The calcium-sensing receptor in bone metabolism: from bench to bedside and back. Osteoporos. Int. 26 (8), 2055–2071. https://doi.org/10.1007/s00198-015-3203-1 (2015).
Marie, P. The calcium-sensing receptor in bone cells: a potential therapeutic target in osteoporosis. Bone 46, 571–576. https://doi.org/10.1016/j.bone.2009.07.082 (2009).
Rico, H. et al. Effect of silicon supplement on osteopenia induced by ovariectomy in rats. Calcif Tissue Int. 66 (1), 53–55. https://doi.org/10.1007/s002230050010 (2000).
Tang, Z., Li, X., Tan, Y., Fan, H. & Zhang, X. The material and biological characteristics of osteoinductive calcium phosphate ceramics. Regen Biomater. 5 (1), 43–59. https://doi.org/10.1093/rb/rbx024 (2018).
Ghorai, S. K., Maji, S., Subramanian, B., Maiti, T. K. & Chattopadhyay, S. Coining attributes of ultra-low concentration graphene oxide and spermine: an approach for high strength, anti-microbial and osteoconductive nanohybrid scaffold for bone tissue regeneration. Carbon 141, 370–389. https://doi.org/10.1016/j.carbon.2018.09.062 (2019).
Fu, J. et al. Core–shell biphasic microspheres with tunable density of shell micropores providing tailorable bone regeneration. Tissue Eng. Part. A. 25, 588. https://doi.org/10.1089/ten.tea.2018.0174 (2019).
Shao, H. et al. Custom repair of mandibular bone defects with 3D printed bioceramic scaffolds. J. Dent. Res. 97, 68. https://doi.org/10.1177/0022034517734846 (2018).
Acknowledgements
We would like to thank Dr. Binji Cao for his technical assistance in manuscript writing.
Funding
This study received financial support from the Medical and Health Technology Project of Hangzhou (A20230968, Xiurong Ke), the Medical and Health Research Project of Zhejiang Province (2025KY174, Xiurong Ke), Key Laboratory of Oral Biomedical Research of Zhejiang Province Foundation (2021M001, Lingling Dong), and National Natural Science Foundation of China (82101649, Lijie Fan).
Author information
Authors and Affiliations
Contributions
Dong, L. & Li, Y.: material preparation, data analysis and writing-original draft; Dong, L. & Feng, Y.: *in vivo* experiments and harvest of femoral bone specimens; Feng, Y. & Wang, X: suggestions on the animal bone defect model; Liu, Y. & Ke, X.: XRD, EDS and X-ray analysis; Dong, L. & Wang X.: μ-CT investigation and statistic analysis; Ke, X. & Fan, L.: supervision and funding acquisition; Yu, Y. & Ke, K.: proofread the paper; Fan, L. & Ke, X: experimental guidance and writing-review & editing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical statement
All of the animal experiments of this study were strictly performed in accordance with the ethical standards of the ARRIVE guidelines (PLoS Bio 8(6), e1000412,2010). The animal studies of this study were registered under ethical committee approval number ZJU21202 and ZJU20220475 by the Ethics Committee of Zhejiang University respectively. The anesthesia and euthanasia methods of this study were performed in accordance with the American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals (2020). The sample size is calculated based on pre-experimental data (n = 6, using random grouping). All data were expressed as the mean ± SD and analyzed for variance using SPSS 26.0, with a significance level of p < 0.05.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dong, L., Li, Y., Feng, Y. et al. Co-incorporation of Phosphorus and zinc into wollastonite ceramic granules synergically facilitating thin-walled structures regeneration. Sci Rep 16, 13668 (2026). https://doi.org/10.1038/s41598-026-44387-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-44387-7