Abstract
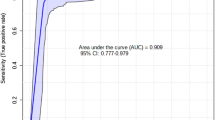
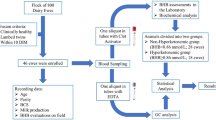
Fetal mortality significantly impacts reproductive efficiency in sheep, with approximately 30% of fetuses failing to survive until lambing. In multifetal gestation, partial litter loss (PLL) occurs in 28% to 37% of cases. The biochemical composition of allantoic fluid (ALF), which plays a key role in metabolic regulation and fetal waste clearance, has been shown in other species to reflect fetal development and maturity. However, ALF metabolomics in sheep, especially in relation to PLL, remains unexplored. This study investigated late-gestation fetal mortality in multifetal sheep pregnancies by analyzing maternal metabolic status, placental gene expression, and ALF composition to identify potential metabolic compounds associated with PLL. The study was conducted on 25 ewes, of which 14 experienced PLL, and 11 had vital litters (VL). Plasma analysis revealed that VL ewes have higher β-hydroxybutyrate concentrations than PLL ewes, whereas plasma insulin and cortisol levels are higher in PLL ewes. Analysis of gene expression in the placental cotyledon demonstrated increased androgen receptor expression in PLL pregnancies and trends toward elevated solute carrier family 2 member 1, and reduced nuclear receptor subfamily 3 group C member 1 expression. Insulin-like growth factor 2 expression tended to be higher in VL placentas. Allantocentesis was performed on 25 ewes with multifetal pregnancies (11 VL and 14 PLL). ALF samples were collected from 74 fetuses at 140.4 ± 2.0 days in pregnancy, and 41 samples were selected for metabolomic profiling. Using genomic analysis, 25 ALF samples were confidently matched to individual lambs. Untargeted lipidomics of ALF from 14 lambs in PLL (7 born alive vs. 7 stillborn) revealed 24 differentially abundant lipid compounds. Stillborn lambs exhibited lower phosphatidylglycerol (PG) 15:0_20:1, but higher levels of sphingomyelin (SM), lysophosphatidylcholine (LPC), ether-linked phosphatidylethanolamines (PEs), lysophosphatidylethanolamines (LPEs), and monogalactosyldiacylglycerols (MGDGs). Collectively, PLL in multifetal ewe pregnancies is linked to maternal metabolic imbalance, elevated cortisol levels, dysregulated placental gene expression, and altered ALF lipid profiles in stillborn fetuses. Elevated SM and LPC, and lower PG, suggest immature lungs. These molecular and metabolic signatures may provide new insights into the mechanisms underlying PLL in late-gestation multifetal pregnancies.
Similar content being viewed by others
Data availability
All data are presented in the paper.
References
Bolet, G. Timing and Extent of Embryonic Mortality in Pigs Sheep and Goats: Genetic Variability. Embryonic Mortal Farm Anim. (Springer, Dordrecht, 12–43). Available from: https://link.springer.com/chapter/ (1986).
Quinlivan, T. D., Martin, C. A., Taylor, W. B. & Cairney, I. M. Estimates of pre- and perinatal mortality in the New Zealand Romney Marsh ewe. II. Pre- and perinatal loss in those ewes that conceived to second service and those that returned to second service and were mated a third time. J Reprod Fertil. Bioscientifica Ltd; ;11:391–8. (1966). Available from: https://rep.bioscientifica.com/view/journals/rep/11/3/jrf_11_3_009.xml
Dixon, A. B. et al. Patterns of late embryonic and fetal mortality and association with several factors in sheep 1. J Anim Sci. ;85:1274–84. (2007). Available from: https://academic.oup.com/jas/article-abstract/85/5/1274/4777701
Alon, T., Rosov, A., Lifshitz, L. & Moallem, U. Male fetuses negatively affect the vitality of the litter and the dam’s metabolic and physiological state in multifetal pregnant ewe. PLoS One. Public Library of Science; ;18:1–11. (2023). Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0285338
Gootwine, E. Variability in the rate of decline in birth weight as litter size increases in sheep. Anim. Sci. J. 81, 393–8 (2005).
Gootwine, E., Spencer, T. E. & Bazer, F. W. Litter-size-dependent intrauterine growth restriction in sheep. Animal 1, 547–564 (2007).
Hillman, S. C., Morris, R. K. & Kilby, M. D. Single twin demise: Consequence for survivors. Semin. Fetal. Neonatal. Med. 15, 319–26 (2010).
Gootwine, E., Reicher, S. & Rozov, A. Prolificacy and lamb survival at birth in Awassi and Assaf sheep carrying the FecB (Booroola) mutation. Anim. Reprod. Sci. 108, 402–11 (2008).
Beachler, T. M. et al. Metabolomic Profile of Allantoic and Amniotic Fluid in Late-term Gestational Mares Characterized by 1H-nuclear Magnetic Resonance Spectroscopy. J Equine Vet Sci. W.B. Saunders; ;94:103235. (2020). Available from: https://www.sciencedirect.com/science/article/pii/S0737080620303269?utm_source=chatgpt.com
Petit, A. et al. Allantoic fluid metabolome reveals specific metabolic signatures in chicken lines different for their muscle glycogen content. Sci Rep. Nature Research; ;13:1–15. (2023). Available from: https://www.nature.com/articles/s41598-023-35652-0
Sahraei, H., Mogheiseh, A., Nazifi, S., Divar, M. R. & Iraji, F. Canine and feline foetal fluids: Volume, hormonal and biochemical characterization during pregnancy. Vet Med Sci. John Wiley and Sons Inc; ;10. (2024). Available from: https://pubmed.ncbi.nlm.nih.gov/38654677/
Bell, A. W. Regulation of organic nutrient metabolism during transition from late pregnancy to early lactation. J Anim Sci [Internet]. Oxford University Press; [cited 2018 Sep 17];73:2804. (1995). Available from: https://academic.oup.com/jas/article/73/9/2804-2819/4632911
Moallem, U., Rozov, A., Gootwine, E. & Honig, H. Plasma concentrations of key metabolites and insulin in late-pregnant ewes carrying 1 to 5 fetuses. J Anim Sci [Internet]. ;90:318–24. (2012). Available from: https://academic.oup.com/jas/article/90/1/318/4764617
Seckl, J. R. & Holmes, M. C. Mechanisms of disease: Glucocorticoids, their placental metabolism and fetal programming of adult pathophysiology. Nat Clin Pract Endocrinol Metab. Nature Publishing Group; ;3:479–88. (2007). Available from: https://www.nature.com/articles/ncpendmet0515
Dy, J., Guan, H., Sampath-Kumar, R., Richardson, B. S. & Yang, K. Placental 11β-Hydroxysteroid Dehydrogenase Type 2 is Reduced in Pregnancies Complicated with Idiopathic Intrauterine Growth Restriction: Evidence That This is Associated With an Attenuated Ratio of Cortisone to Cortisol in the Umbilical Artery. Placenta. Placenta; ;29:193–200. (2008). Available from: https://pubmed.ncbi.nlm.nih.gov/18061258/
Sloboda, D. M., Newnham, J. P. & Challis, J. R. G. Effects of repeated maternal betamethasone administration on growth and hypothalamic-pituitary-adrenal function of the ovine fetus at term. J Endocrinol. Society for Endocrinology; ;165:79–91. (2000). Available from: https://pubmed.ncbi.nlm.nih.gov/10750038/
Fowden, A. L., Vaughan, O. R. & Forhead, A. J. Early-life programming of livestock metabolism by glucocorticoids. J Dev Orig Health Dis. Cambridge University Press; ;16:e16. (2025). Available from: https://www.cambridge.org/core/journals/journal-of-developmental-origins-of-health-and-disease/article/earlylife-programming-of-livestock-metabolism-by-glucocorticoids/BFCB6C72DEE48E9DCDDC8543374A5C73
Meakin, A. S., Gough, M., Saif, Z. & Clifton, V. L. An ex vivo approach to understanding sex-specific differences in placental androgen signalling in the presence and absence of inflammation. Placenta. W.B. Saunders; ;120:49–58. (2022). Available from: https://linkinghub.elsevier.com/retrieve/pii/S0143400422000558
Illsley, N. P. Glucose Transporters in the Human Placenta. Placenta. W.B. Saunders; ;21:14–22. (2000). Available from: https://www.sciencedirect.com/science/article/abs/pii/S0143400499904484?via%3Dihub
Jansson, T., Wennergren, M. & Illsley, N. P. Glucose transporter protein expression in human placenta throughout gestation and in intrauterine growth retardation. J. Clin. Endocrinol. Metab. 77, 1554–62. https://doi.org/10.1210/jcem.77.6.8263141 (1993).
Brett, K. E., Ferraro, Z. M., Yockell-Lelievre, J., Gruslin, A. & Adamo, K. B. Maternal–Fetal Nutrient Transport in Pregnancy Pathologies: The Role of the Placenta. Int J Mol Sci. Vol 15, Pages 16153–16185. Multidisciplinary Digital Publishing Institute; 2014;15:16153–85. (2014). Available from: https://www.mdpi.com/1422-0067/15/9/16153/htm
Moisiadis, V. G. & Matthews, S. G. Glucocorticoids and fetal programming part 1: outcomes. Nat Rev Endocrinol 2014 107. Nature Publishing Group; ;10:391–402. (2014). Available from: https://www.nature.com/articles/nrendo.2014.73
Sibley, C. P. et al. Placental-specific insulin-like growth factor 2 (Igf2) regulates the diffusional exchange characteristics of the mouse placenta. Proc. Natl. Acad. Sci. U. S. A. 101, 8204–8. https://doi.org/10.1073/pnas.0402508101 (2004).
Sferruzzi-Perri, A. N., Sandovici, I., Constancia, M. & Fowden, A. L. Placental phenotype and the insulin-like growth factors: Resource allocation to fetal growth. J. Physiol. 595, 5057–93. https://doi.org/10.1113/JP273330 (2017).
Mellor, D. J. & Slater, J. S. Daily changes in amniotic and allantoic fluid during the last three months of pregnancy in conscious, unstressed ewes, with catheters in their foetal fluid sacs. J. Physiol. 217, 573–604. https://doi.org/10.1113/jphysiol.1971.sp009587 (1971).
Shandley, L., Alcorn, D. & Wintour, E. M. Ovine amniotic and allantoic epithelia across gestation. Anat. Rec. 248, 542–53. https://doi.org/10.1002/%28SICI%2910970185%28199708%29248%3A4%3C542%3A%3AAID-AR6%3E3.0.CO%3B2-N (1997).
Haffaf, S. & Benallou, B. Changes in energetic profile of pregnant ewes in relation with the composition of the fetal fluids. Asian Pac J Trop Biomed. No longer published by Elsevier; ;6:256–8. (2016). Available from: https://www.sciencedirect.com/science/article/pii/S2221169115002786
Khatun, A., Wani, G. M., Bhat, J. I. A., Choudhury, A. R. & Khan, M. Z. Biochemical indices in sheep during different stages of pregnancy. Asian J. Anim. Vet. Adv. 6, 175–81 (2011).
Tabatabaei, S. Gestational variations in the biochemical composition of the fetal fluids and maternal blood serum in goat. Comp Clin Path. Springer; ;21:1305–12. Available from: https://link.springer.com/article/ (2012). https://doi.org/10.1007/s00580-011-1286-4
Furse, S. Is phosphatidylglycerol essential for terrestrial life? J Chem Biol Springer; 2016;10:1–9. Available from: https://link.springer.com/article/ (2016). 101 https://doi.org/10.1007/s12154-016-0159-3
Goerke, J. Pulmonary surfactant: functions and molecular composition. Biochim Biophys Acta - Mol Basis Dis. Elsevier; ;1408:79–89. (1998). Available from: https://www.sciencedirect.com/science/article/pii/S092544399800060X?via%3Dihub
Zhuo, R., Rong, P., Wang, J., Parvin, R. & Deng, Y. The Potential Role of Bioactive Plasmalogens in Lung Surfactant. Front. Cell. Dev. Biol. Front. Media S A. 9, 618102 (2021). Available from: www.frontiersin.org.
Sampaio, J. L. et al. Membrane lipidome of an epithelial cell line. Proc Natl Acad Sci U S A. National Academy of Sciences; ;108:1903–7. (2011). Available from: https://doi.org/10.1073/pnas.1019267108?download=true
Quehenberger, O. et al. Lipidomics reveals a remarkable diversity of lipids in human plasma1. J Lipid Res. Elsevier; ;51:3299–305. (2010). Available from: https://www.jlr.org/action/showFullText?pii=S0022227520409630
Atilla-Gokcumen, G. E. et al. Dividing cells regulate their lipid composition and localization. Cell. Elsevier B.V.; ;156:428–39. (2014). Available from: https://www.cell.com/action/showFullText?pii=S0092867413015493
Lemke, A. et al. Human amniotic membrane as newly identified source of amniotic fluid pulmonary surfactant. Sci Reports 2017 71. Nature Publishing Group; ;7:1–13. (2017). Available from: https://www.nature.com/articles/s41598-017-06402-w
Bustos, R. et al. Significance of phosphatidylglycerol in amniotic fluid in complicated pregnancies. Am J Obstet Gynecol. Am J Obstet Gynecol; (1979). ;133:899–903 Available from: https://pubmed.ncbi.nlm.nih.gov/107803/
Skjáraasen, J. & Stray-Pedersen, S. Amniotic fluid phosphatidylinositol and phosphatidylglycerol: I. Normal pregnancies. Acta Obstet Gynecol Scand. John Wiley & Sons, Ltd; ;58:225–229. (1979). Available from: /doi/https://doi.org/10.3109/00016347909154038.
Hallman, M., Spragg, R., Harrell, J. H., Moser, K. M. & Gluck, L. Evidence of lung surfactant abnormality in respiratory failure. Study of bronchoalveolar lavage phospholipids, surface activity, phospholipase activity, and plasma myoinositol. J. Clin. Invest. 70, 673–83 (1982).
Hallman, M., Feldman, B. H., Kirkpatrick, E. & Gluck, L. Absence of phosphatidylglycerol (PG) in respiratory distress syndrome in the newborn: Study of the minor surfactant phospholipids in newborns. Pediatr Res. Nature Publishing Group; ;11:714–20. (1977). Available from: https://www.nature.com/articles/pr19771331
Numataa, M., Chua, H. W., Dakhamab, A. & Voelkera, D. R. Pulmonary surfactant phosphatidylglycerol inhibits respiratory syncytial virus-induced inflammation and infection. Proc Natl Acad Sci U S A. National Academy of Sciences; ;107:320–5. (2010). Available from: https://doi.org/10.1073/pnas.0909361107?download=true
Gluck, L., Kulovich, M. V., Borer, R. C. & Keidel, W. N. The interpretation and significance of the lecithin/sphingomyelin ratio in amniotic fluid. Am J Obstet Gynecol. ;120:142–55. (1974). Available from: https://linkinghub.elsevier.com/retrieve/pii/000293787490194X
Gluck, L. et al. Diagnosis of the respiratory distress syndrome by amniocentesis. Am. J. Obstet. Gynecol. 109, 440–5 (1971).
Ogawa, Y. Fetal pulmonary surfactant in amniotic fluid of ewes. Tohoku J. Exp. Med. 108, 307–15 (1972).
Fujiwara, T., Adams, F. H. & Scudder, A. Fetal lamb amniotic fluid: Relationship of lipid composition to surface tension. J Pediatr. Mosby; ;65:824–30. (1964). Available from: https://www.sciencedirect.com/science/article/pii/S0022347664800044
Cullis, P. R., Van Dijck, P. W. M., De Kruijff, B. & De Gier, J. Effects of cholesterol on the properties of equimolar mixtures of synthetic phosphatidylethanolamine and phosphatidylcholine. A 31P NMR and differential scanning calorimetry study. Biochim Biophys Acta - Biomembr. Elsevier; ;513:21–30. (1978). Available from: https://www.sciencedirect.com/science/article/pii/0005273678901086?via%3Dihub
Chen, X., Hyatt, B. A., Mucenski, M. L., Mason, R. J. & Shannon, J. M. Identification and characterization of a lysophosphatidylcholine acyltransferase in alveolar II cells. Proc. Natl. Acad. Sci. U S Natl. Acad. Sci. 103, 11724–11729. https://doi.org/10.1073/pnas.0604946103?download=true (2006). Available from: /doi/.
Han, S. & Mallampalli, R. K. Sizing up surfactant synthesis. Cell Metab. Cell Press; ;20:195–6. (2014). Available from: https://www.cell.com/action/showFullText?pii=S1550413114003234
Purandare, N. et al. Molecular mechanisms regulating lysophosphatidylcholine acyltransferase 1 (LPCAT1) in human pregnancy. Placenta. W.B. Saunders; ;106:40–8. (2021). Available from: https://www.sciencedirect.com/science/article/pii/S0143400421000473?via%3Dihub
Kramer, K. J. et al. Lysophosphatidylcholine acyltransferase 1 protein is present in maternal blood in the third trimester and is upregulated by antenatal corticosteroids. J Perinat Med. De Gruyter Open Ltd; ;51:432–4. Available from: https://www.degruyterbrill.com/document/doi/ (2023). https://doi.org/10.1515/jpm-2022-0150/html
Tyden, O., Berne, C. & Eriksson, U. Lung maturation in fetuses of diabetic rats. Pediatr. Res. 14, 1192–1195 (1980).
Mohany, K. M. et al. The association of LPCAT1-rs9728 polymorphism with cord blood IL-10, MIF, and VEGF levels in neonatal respiratory distress syndrome: a case–control study. Egypt J Bronchol 2024 181. SpringerOpen; ;18:1–10. Available from: https://ejb.springeropen.com/articles/ (2024). https://doi.org/10.1186/s43168-024-00273-2
Carter, S. W. D. et al. Maternal plasma cell-free RNA as a predictive test for fetal lung maturation. BMC Med. BioMed Central Ltd; ;23:1–20. Available from: https://bmcmedicine.biomedcentral.com/articles/ (2025). https://doi.org/10.1186/s12916-025-04256-y
Welch, R. A., Shaw, M. K. & Welch, K. C. Amniotic fluid LPCAT1 mRNA correlates with the lamellar body count. J Perinat Med. Walter de Gruyter GmbH; ;44:531–2. (2016). Available from: https://pubmed.ncbi.nlm.nih.gov/25968427/
Clifton, V. L. & Review Sex and the Human Placenta: Mediating Differential Strategies of Fetal Growth and Survival. Placenta. Placenta; ;31. (2010). Available from: https://pubmed.ncbi.nlm.nih.gov/20004469/
Melamed, N., Yogev, Y. & Glezerman, M. Fetal gender and pregnancy outcome. J. Matern. Fetal. Neonatal. Med. 23, 338–344 (2010).
Malitsky, S. et al. Viral infection of the marine alga Emiliania huxleyi triggers lipidome remodeling and induces the production of highly saturated triacylglycerol. New Phytol. 210, 88–96. https://doi.org/10.1111/nph.13852 (2016).
Zheng, L. et al. Fumarate induces redox-dependent senescence by modifying glutathione metabolism. Nat Commun. Nature Publishing Group; ;6:6001. (2015). Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC4340546/
Bendikov-Bar, I., Malitsky, S., Itkin, M., Rusal, M. & Sagi, D. Metabolomic Changes Are Predictive of Aging in Laying Hens. Journals Gerontol - Ser A Biol Sci Med Sci. Oxford University Press; ;76:1757–68. (2021). Available from: https://pubmed.ncbi.nlm.nih.gov/33978733/
Thermo Fisher Scientific. LipidSearch. San Jose (Thermo Fisher Scientific, 2016). Available from: www.thermofisher.com.
Thermo Fisher Scientific. TraceFinder Software. Waltham (Thermo Fisher Scientific Inc, 2019). Available from: www.thermofisher.com.
Waters Corporation & Milford MassLynx Software with TargetLynx XS (Waters Corporation, 2017). Available from: www.waters.com.
Anelli, G. M. et al. Placental ERRγ-CYP19 expressions and circulating 17-Beta Estradiol in IUGR pregnancies. Front Pediatr [Internet]. Front. Media S A. 7, 447158 (2019). from: www.frontiersin.org.
Winterhager, E. & Gellhaus, A. Transplacental nutrient transport mechanisms of intrauterine growth restriction in rodent models and humans. Front Physiol [Internet]. Frontiers Media S.A.; ;8:308030. (2017). Available from: https://www.nichd.nih
Moisiadis, V. G. & Matthews, S. G. Glucocorticoids and fetal programming part 2: mechanisms. Nat Rev Endocrinol 2014 107. Nature Publishing Group; ;10:403–11. (2014). Available from: https://www.nature.com/articles/nrendo.2014.74
Grammatopoulos, D. K. Placental corticotrophin-releasing hormone and its receptors in human pregnancy and labour: Still a scientific enigma. J. Neuroendocrinol. 20, 432–8. https://doi.org/10.1111/j.1365-2826.299008.01660.x (2008).
Parsons, A. M. & Bouma, G. J. A Potential Role and Contribution of Androgens in Placental Development and Pregnancy. Life. MDPI AG; ;11:644. (2021). Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8305703/
SAS SAS User’s Guide: Statistics. Cary, NC, USA; 2002. (2002).
R Core Team. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing. (2025). Available from: https://www.r-project.org/
Acknowledgements
The authors wish to thank the entire staff in charge of the experimental flock at the Volcani Center, Rishon LeZion, Israel, for their assistance with animal care.This sentence is added to the manuscript.
Funding
This research was financially supported by the Chief Scientist of the Ministry of Agriculture, Rishon LeZion – grant number 362081023.
Author information
Authors and Affiliations
Contributions
**TA: ** Methodology, Conceptualization, Project administration, Ultrasound monitoring, Data curation, Visualization, Laboratory analysis, Formal analysis, Validation, and Writing. **MR** : Allantocentesis. **JS** : Data curation, Visualization **. PDSS** : Laboratory analysis, Formal analysis. **AR** : Data curation, Animal care, Formal analysis, Methodology. **LL** : Data curation, Laboratory analysis, Methodology, Validation. **GK** : Laboratory analysis, Formal analysis. **MG** : Genomic data processing. **SM** : Formal analysis, Metabolomics, Lipidomics. **MI** : Formal analysis, Metabolomics, Lipidomics **. RR** : Data processing, Statistical analysis. **UM: ** Conceptualization, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Visualization, Writing – review & editing. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The Volcani Center Animal Care Committee approved all of the procedures performed on the animals in the present study (approval no. 981/23 IL). Consent to participate is not relevant.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Alon, T., Ross, M., Spirer, J. et al. Lipidomics of allantoic fluid collected by allantocentesis indicates a possible mechanism underlying partial litter loss in multifetal pregnancies of ewes. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44636-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44636-9