Abstract
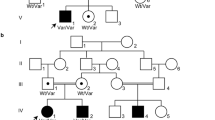
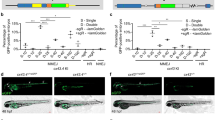
Biallelic mutations in human GON4L have been linked to short stature, craniofacial abnormalities, and microcephaly, yet the underlying mechanisms remain unclear. GON4L is a nuclear protein composed of a YY1AP1-like region, two PAH domains, and one SANT domain, all implicated in transcriptional or chromatin regulation. Here, we define the function of Gon4la (a zebrafish ortholog of human GON4L) in post-embryonic growth and tissue homeostasis. We generated three gon4la mutant lines using CRISPR-Cas9, each predicted to express truncated proteins lacking the C-terminal PAH/SANT domains and differing in N-terminal YY1AP1-like region retention. All gon4la mutants displayed proportionate dwarfism with intestinal and pancreatic abnormalities, accompanied by expanded progenitor proliferation and impaired epithelial differentiation. Only mutants completely lacking the N-terminal YY1AP1-like region exhibited reduced igf1a expression, elevated growth hormone pathway activity, and increased mortality, consistent with endocrine dysregulation. Our findings demonstrate that distinct domains of Gon4la contribute differentially to systemic growth and metabolic organ homeostasis. Zebrafish gon4la mutants provide a tractable vertebrate model for dissecting mechanisms of GON4L-related developmental disorder, Li-Takada-Miyake syndrome (LTMS), and for functional testing of rare GON4L variants.
Similar content being viewed by others
Data availability
The authors confirm that the data supporting this study are available within the article. Original data that support the findings of this study are available from the corresponding author upon reasonable request. The gon4la sequence has been deposited in the NCBI GenBank (accession number: PX525845) and ENA (accession number: ERP184456).
References
Ornitz, D. M. & Legeai-Mallet, L. Achondroplasia: Development, pathogenesis, and therapy.. Dev. Dyn. 246, 291–309. https://doi.org/10.1002/dvdy.24479 (2017).
Mastromauro, C., Giannini, C. & Chiarelli, F. Short stature related to growth hormone insensitivity (GHI) in childhood. Front. Endocrinol. (Lausanne) 14, 1141039. https://doi.org/10.3389/fendo.2023.1141039 (2023).
Dehkhoda, F., Lee, C. M. M., Medina, J. & Brooks, A. J. The growth hormone receptor: Mechanism of receptor activation, cell signaling, and physiological aspects. Front. Endocrinol. (Lausanne) 9, 35. https://doi.org/10.3389/fendo.2018.00035 (2018).
Gabriele, M. et al. YY1 haploinsufficiency causes an intellectual disability syndrome featuring transcriptional and chromatin dysfunction. Am. J. Hum. Genet. 100, 907–925. https://doi.org/10.1016/j.ajhg.2017.05.006 (2017).
Gabriele, M., Lopez Tobon, A., D’Agostino, G. & Testa, G. The chromatin basis of neurodevelopmental disorders: Rethinking dysfunction along the molecular and temporal axes.. Prog. Neuropsychopharmacol. Biol. Psychiatry 84, 306–327. https://doi.org/10.1016/j.pnpbp.2017.12.013 (2018).
Li, S. et al. Biallelic loss-of-function variants in GON4L cause microcephaly and brain structure abnormalities.. NPJ Genom. Med. 9, 55. https://doi.org/10.1038/s41525-024-00437-5 (2024).
Schwarzenbacher, H. et al. A frameshift mutation in GON4L is associated with proportionate dwarfism in Fleckvieh cattle.. Genet. Sel. Evol. 48, 25 (2016).
Ohtomo, T., Horii, T., Nomizu, M., Suga, T. & Yamada, J. Molecular cloning of a structural homolog of YY1AP, a coactivator of the multifunctional transcription factor YY1. Amino Acids 33, 645–652 (2007).
Verheul, T. C. J., van Hijfte, L., Perenthaler, E. & Barakat, T. S. The why of YY1: Mechanisms of transcriptional regulation by Yin Yang 1.. Front. Cell Dev. Biol. 8, 592164. https://doi.org/10.3389/fcell.2020.592164 (2020).
Lu, P. et al. The developmental regulator protein Gon4l associates with protein YY1, co-repressor Sin3a, and Histone Deacetylase 1 and mediates transcriptional repression. J. Biol. Chem. 286, 18311–18319 (2011).
Spronk, C. A. et al. The Mad1-Sin3B interaction involves a novel helical fold. Nat. Struct. Biol. 7, 1100–1104 (2000).
Boyer, L. A. et al. Essential role for the SANT domain in the functioning of multiple chromatin remodeling enzymes.. Mol. Cell 10, 935–942 (2002).
Wang, C. Y. et al. YY1AP, a novel co-activator of YY1. J. Biol. Chem. 279, 17750–17755 (2004).
McMenamin, S. K., Minchin, J. E., Gordon, T. N., Rawls, J. F. & Parichy, D. M. Dwarfism and increased adiposity in the gh1 mutant zebrafish Vizzini. Endocrinology 154, 1476–1487. https://doi.org/10.1210/en.2012-1734 (2013).
Yao, L., Chen, J., Wu, X., Jia, S. & Meng, A. Zebrafish cdc6 hypomorphic mutation causes Meier-Gorlin syndrome-like phenotype. Hum. Mol. Genet. 26, 4168–4180. https://doi.org/10.1093/hmg/ddx305 (2017).
Colgan, D. F., Goodfellow, R. X. & Colgan, J. D. The transcriptional regulator GON4L is required for viability and hematopoiesis in mice. Exp. Hematol. 98, 25–35. https://doi.org/10.1016/j.exphem.2021.04.001 (2021).
Williams, M. L. K. et al. Gon4l regulates notochord boundary formation and cell polarity underlying axis extension by repressing adhesion genes. Nat. Commun. 9, 1319 (2018).
Lim, C.-H., Chong, S.-W. & Jiang, Y.-J. Udu deficiency activates DNA damage checkpoint. Mol. Biol. Cell 20, 4183–4193. https://doi.org/10.1091/mbc.E09-02-0109 (2009).
Liu, Y. et al. The zebrafish udu gene encodes a novel nuclear factor and is essential for primitive erythroid cell development. Blood 110, 99–106. https://doi.org/10.1182/blood-2006-11-059204 (2007).
Cubbage, C. C. & Mabee, P. M. Development of the cranium and paired fins in the zebrafish Danio rerio (Ostariophysi, Cyprinidae). J. Morphol. 229, 121–160. (1996).
Charles, J. F. et al. Utility of quantitative micro-computed tomographic analysis in zebrafish to define gene function during skeletogenesis. Bone 101, 162–171. https://doi.org/10.1016/j.bone.2017.05.001 (2017).
Zeng, N. et al. Sexual dimorphic effects of igf1 deficiency on metabolism in zebrafish. Front. Endocrinol. (Lausanne) 13, 879962. https://doi.org/10.3389/fendo.2022.879962 (2022).
Capozzi, A., Casa, S. D., Altieri, B. & Pontecorvi, A. Low bone mineral density in a growth hormone deficient (GHD) adolescent. Clin. Cases Miner. Bone Metab. 10, 203–205 (2013).
Barker, N. Adult intestinal stem cells: Critical drivers of epithelial homeostasis and regeneration. Nat. Rev. Mol. Cell Biol. 15, 19–33. https://doi.org/10.1038/nrm3721 (2014).
Peng, Z., Bao, L., Shi, B. & Shi, Y. B. Protein arginine methyltransferase 1 is required for the maintenance of adult small intestinal and colonic epithelial cell homeostasis. Int. J. Biol. Sci. 20, 554–568. https://doi.org/10.7150/ijbs.89958 (2024).
Tavakoli, S., Zhu, S. & Matsudaira, P. Cell clusters containing intestinal stem cells line, the zebrafish intestine intervillus pocket. iScience 25, 104280. https://doi.org/10.1016/j.isci.2022.104280 (2022).
Anderson, J. L. et al. mRNA processing in mutant zebrafish lines generated by chemical and CRISPR-mediated mutagenesis produces unexpected transcripts that escape nonsense-mediated decay. PLoS Genet. 13, e1007105. https://doi.org/10.1371/journal.pgen.1007105 (2017).
Blättler, S. M. et al. Yin Yang 1 deficiency in skeletal muscle protects against rapamycin-induced diabetic-like symptoms through activation of insulin/IGF signaling. Cell Metab. 15, 505–517. https://doi.org/10.1016/j.cmet.2012.03.008 (2012).
Perekatt, A. O. et al. YY1 is indispensable for Lgr5+ intestinal stem cell renewal. Proc. Natl. Acad. Sci. U S A 111, 7695–7700. https://doi.org/10.1073/pnas.1400128111 (2014).
Fagerberg, L. et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol. Cell. Proteomics 13, 397–406. https://doi.org/10.1074/mcp.M113.035600 (2014).
You, M.-S. et al. A sketch of the Taiwan Zebrafish Core Facility. Zebrafish 13, S24–S29 (2016).
Kimmel, C. B., Ballard, W. W., Kimmel, S. R., Ullmann, B. & Schilling, T. F. Stages of embryonic development of the zebrafish. Dev. Dyn. 203, 253–310 (1995).
Tsai, S. M., Chu, K. C. & Jiang, Y. J. Newly identified Gon4l/Udu-interacting proteins implicate novel functions. Sci. Rep. 10, 14213. https://doi.org/10.1038/s41598-020-70855-9 (2020).
Hoshijima, K. et al. Highly efficient CRISPR-Cas9-based methods for generating deletion mutations and F0 embryos that lack gene function in zebrafish. Dev. Cell 51, 645-657.e644. https://doi.org/10.1016/j.devcel.2019.10.004 (2019).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675. https://doi.org/10.1038/nmeth.2089 (2012).
Ding, Y. et al. Computational 3D histological phenotyping of whole zebrafish by X-ray histotomography. Elife https://doi.org/10.7554/eLife.44898 (2019).
Acknowledgements
We thank the Taiwan Mouse Clinic, Academia Sinica and Taiwan Animal Consortium for their technical support in micro-CT imaging. We are grateful to the staff at the Zebrafish Facility of NHRI for their efforts in maintaining fish stocks. We thank the Pathology Core Facility of NHRI for HE staining.
Funding
This work was supported by the National Health Research Institutes, Taiwan (MG-111-PP-08, MG-112-PP-08, MG-113-PP-08, and BN-114-PP-31) and grants from the Ministry of Science and Technology/National Science and Technology Council, Taiwan (MOST 106-2311-B-400-003-MY3, MOST 111-2740-B-400-001-, NSTC 112-2320-B-400-011-MY3, and NSTC 113-2740-B-400-001-).
Author information
Authors and Affiliations
Contributions
SMT: conceptualization, methodology, formal analysis, investigation, writing—original draft, visualization; CHH: conceptualization, investigation, resources; ICC: investigation, resources; WNL: investigation, visualization, writing—original draft; JKC: resources; YJJ: conceptualization, funding acquisition, resources, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All procedures involving zebrafish and their embryos were approved by the NHRI Institutional Animal Care and Use Committee (NHRI-IACUC-106063-A, NHRI-IACUC-110132-AC1, and NHRI-IACUC-112019-A), and carried out in accordance with the approved guidelines.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tsai, SM., Hsu, CH., Chiang, IC. et al. Zebrafish gon4la mutants recapitulate human GON4L-related growth disorders and reveal novel metabolic organs abnormalities. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44674-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44674-3