Abstract
This multicenter study evaluated the prognostic impact of acute kidney injury (AKI) on one-year survival in patients undergoing Impella-supported high-risk percutaneous coronary intervention (hrPCI), and whether Impella use offers renal protection. A total of 470 patients from two independent cohorts were analyzed: the IMPELLA-PL group (n = 249, derivation cohort) and the Dresden group (n = 221, validation cohort). Patients with cardiogenic shock or chronic dialysis were not included. AKI—defined as a ≥ 0.5 mg/dL increase in serum creatinine within 72 h—occurred in 13.3% and 14.9% of patients, respectively, which was significantly lower than the predicted 26.1% risk based on the Mehran Risk Score. AKI was an independent predictor of one-year mortality in both the derivation (HR: 2.75; 95% CI: 1.11–6.82) and validation (HR: 2.16; 95% CI: 1.12–4.10) cohorts. Upfront Impella implantation and higher baseline glomerular filtration rate were independently associated with a reduced risk of AKI in both cohorts. The adverse effect of AKI on long-term survival was significant in males, but not in females. These findings indicate that although AKI remains a strong prognostic marker following hrPCI, early Impella support may mitigate this risk. The results also suggest potential sex-related differences in susceptibility to renal complications following complex coronary interventions.
Similar content being viewed by others
Introduction
Acute kidney injury (AKI) is a serious complication of percutaneous coronary intervention (PCI) and is associated with an increased risk of bleeding, myocardial infarction, and death1. The likelihood of AKI increases with age, comorbidities, and the severity of patient’s condition, especially in the presence of hemodynamic instability2.
Mechanical circulatory support (MCS) devices augment blood flow and improve organ perfusion; however, their use is associated with major complications, particularly those related to vascular access3. The Impella pump (Abiomed Inc., J&J Medtech, Danvers, MA) is an MCS device that delivers high blood flow and significantly unloads the left ventricle, improving survival in patients with cardiogenic shock due to myocardial infarction4. However, its use in this setting has been linked to an increased risk of AKI and the need for renal replacement therapy4,5. Conversely, it has been suggested that in patients without cardiogenic shock undergoing high-risk PCI (hrPCI), Impella pumps may exert a protective effect against AKI6,7. Whether this observation is reproducible remains uncertain. Notably, little is known about the long-term prognostic impact of AKI in patients undergoing Impella-supported hrPCI, or whether this impact differs between genders.
In this study, we analyzed two independent populations of patients who underwent Impella-supported hrPCI, using one as a derivation cohort and the other as a validation cohort to assess the risk and prognostic impact of AKI. Given that men have been shown to be at higher risk of developing AKI than women8, we additionally examined potential sex-related differences in the prognostic impact of AKI. The primary objective was to evaluate the prognostic significance of AKI on one-year mortality in patients without cardiogenic shock at baseline who underwent complex hrPCI with Impella support. The secondary objectives were: (i) to compare the observed prevalence of AKI after hrPCI with the predicted risk based on the Mehran risk score; (ii) to identify determinants of AKI in this population; and (iii) to evaluate the impact of AKI on survival by sex.
Methods
Study population
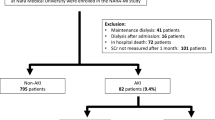
The study population consisted of two cohorts: the IMPELLA-PL group, which served as the derivation cohort, and the Dresden group, which served as the validation cohort. The IMPELLA-PL registry is a nationwide, multicenter, retrospective study that includes patients who received Impella support for cardiogenic shock or hrPCI in 20 Polish interventional cardiology centers that had performed at least five Impella-assisted procedures9. Clinical characteristics, procedural data, and outcomes of consecutive patients from 2014 to December 2021 were retrospectively documented in a secure, password-protected database. Follow-up data at 12 months were obtained through a review of hospital records and outpatient medical documentation. The quality of the collected data was monitored by an independent Study Monitoring Committee10. The registry includes 308 patients divided into two subgroups: those undergoing Impella-assisted revascularization and those receiving Impella support for cardiogenic shock9,10. In this study, only the first subgroup—comprising hemodynamically stable patients with severe coronary artery disease who underwent elective or urgent hrPCI—was considered. Patients presenting with primary cardiogenic shock (55 patients) and those undergoing chronic dialysis (4 patients) were excluded. Cardiogenic shock was defined as the presence of cardiac disease accompanied by systolic blood pressure < 90 mmHg for more than 15 min or the need for catecholamine support to maintain systolic blood pressure ≥ 90 mmHg, together with clinical signs of impaired end-organ perfusion, as indicated by elevated serum lactate levels. The final study group consisted of 249 patients.
The Dresden group consisted of patients derived from the Dresden Impella Registry, an ongoing registry of a high-volume center that consecutively includes cardiogenic shock and hrPCI patients who received Impella11,12. Implantation of Impella was typically performed in a cardiac catheterization laboratory by an experienced interventional cardiologist (> 20 implantations per year). After exclusion of patients presenting with primary cardiogenic shock (as defined in the IMPELLA-PL cohort) and those on chronic dialysis, the final study population consisted of 221 patients undergoing high-risk PCI.
In both cohorts, the decision to perform hrPCI with Impella support was based on a structured Heart Team assessment incorporating objective clinical and anatomical high-risk features. These included severe left ventricular dysfunction, complex multivessel PCI with anticipated hemodynamic compromise, unprotected left main or last remaining vessel disease, extensive coronary calcification requiring atherectomy, and high overall anatomical complexity. The combination of these factors, rather than a single predefined cut-off, guided the decision to use mechanical circulatory support. The decision to perform bailout Impella implantation was at the discretion of the operator performing hrPCI and was primarily driven by hemodynamic deterioration requiring catecholamine administration.
The study was approved by the Bioethics Committee of the Medical University of Warsaw. All methods in this study were performed in accordance with the relevant guidelines and regulations. The need for informed consent was waived by the Bioethics Committee of the Medical University of Warsaw.
Renal function
Using the pre-procedure serum creatinine level as the baseline, the estimated glomerular filtration rate (eGFR) (in ml/min/1.73 m2) was calculated according to the Modification of Diet in Renal Disease (MDRD) equation, which classified patients as having either normal baseline renal function (eGFR ≥ 60 ml/min/1.73 m2) or chronic kidney disease (eGFR < 60 ml/min/1.73 m2).
Acute kidney injury was defined as an increase in serum creatinine of ≥ 0.5 mg/dL (44 μmol/L) or ≥ 25% from baseline within 72 h after high-risk PCI, using the highest recorded creatinine value during this period13,14. The threshold creatinine level for AKI corresponded to that defined in the Mehran risk score, a tool used to predict the risk of AKI following hrPCI. This enabled a comparison between the observed and predicted prevalence of AKI, as estimated by the Mehran risk model and expressed as the percentage probability of developing AKI2. In line with the predicted risk categories, patients were classified as low (7.5%), moderate (14%), high (26.1%), and very high risk (57.3%) for AKI.
Additionally, the sensitivity of AKI’s prognostic power was further assessed using the Kidney Disease Improving Global Outcomes (KDIGO) working group criteria. According to these criteria, AKI was defined as an increase in plasma creatinine of at least 0.3 mg/dL (26.5 μmol/L) over baseline within 48 h after hrPCI15.
Statistical analysis
Categorical data are presented as frequency counts and percentages, while continuous data are expressed as mean ± standard deviation (SD) or median with interquartile range [IQR], as appropriate. Categorical variables were compared using the chi-squared test or Fisher’s exact test, while continuous variables were analyzed using Student’s t-test or the Mann–Whitney U test for non-normally distributed data. In the derivation analysis, baseline patient characteristics, pre-procedural diagnostic results, procedural details, and complications were considered as candidate variables for inclusion in the risk factor model for one-year mortality. Univariable Cox regression analysis was performed to identify variables significantly associated with mortality, followed by multivariable backward stepwise Cox regression, which determined the independent predictors of one-year mortality, adjusted for age and left ventricular ejection fraction. The model was subsequently tested in the validation cohort to evaluate its performance. A similar approach was applied to identify independent predictors of AKI in the derivation cohort, utilizing univariable and multivariable logistic regression analyses, adjusted for age, with the final model undergoing validation in the validation cohort. For the pooled analysis of males and females from both the derivation and validation cohorts, internal validation was performed using the bootstrapping method. Statistical significance was defined as a p-value < 0.05. Statistical analyses were performed using IBM SPSS Statistics, version 30.0.0.0 (172).
Results
Baseline characteristics and procedural details are presented in Table 1. Patients in the IMPELLA-PL and Dresden cohorts differed significantly in many variables; however, there was no significant difference in baseline creatinine levels or AKI prevalence (Table 1). The median [IQR] creatinine increase after hrPCI was lower in the IMPELLA-PL group than in the Dresden group (0.01 [0.28] mg/dL vs. 0.1 [0.36] mg/dL, p < 0.001), including among patients with AKI (0.73 [1.12] mg/dL vs. 1.28 [1.61] mg/dL, p < 0.05). Although the Mehran risk score was lower in IMPELLA-PL patients, renal replacement therapy after hrPCI was more frequent in this group (Table 1). There was no significant difference in the 1-year mortality rate between the IMPELLA-PL and Dresden populations (18.5% vs. 23.5%, p = 0.073); however, the hospital stay was significantly longer in the IMPELLA-PL cohort (11 [11] vs. 7 [8] days, p < 0.001). Table 2 presents the differences between patients with and without AKI. In both cohorts, patients who developed AKI more frequently had chronic kidney disease, higher creatinine levels and Mehran risk scores, and lower eGFR at baseline. Additionally, several cohort-specific differences between the AKI and non-AKI groups were observed, including left ventricular end-diastolic diameter, upfront Impella use, renal replacement therapy after hrPCI, atrial fibrillation, EuroSCORE II, red blood cell count, troponin level, contrast volume and hemoglobin drop after hrPCI (Table 2).
Determinants of one-year mortality
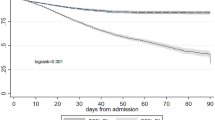
To identify independent predictors of 1-year mortality, the IMPELLA-PL group served as the derivation cohort, while the Dresden group was used as the validation cohort. In IMPELLA-PL patients, univariable Cox regression analysis identified variables from Table 1 that were significantly associated with 1-year mortality. These included diabetes mellitus, atrial fibrillation, prior stroke, EuroSCORE II, pre-procedural white blood cell count and creatinine level, NYHA class, severe aortic valve stenosis, the number of significantly stenosed coronary arteries, Mehran risk score, catecholamine use, and AKI. Subsequently, multivariable Cox regression analysis determined independent predictors of mortality, adjusting for age and LVEF ≤ 35%, and the final model was externally validated in the Dresden group (Table 3 and 4). AKI was a significant predictor of mortality in both populations (Fig. 1). Additionally, atrial fibrillation, pre-procedural creatinine level, the number of significantly stenosed coronary arteries, and catecholamine use were independent predictors in the derivation group (Table 3), whereas LVEF ≤ 35% was an independent predictor in the validation group (Table 4).
Kaplan–Meier survival curves (unadjusted) comparing patients without acute kidney injury (No AKI) and with acute kidney injury (AKI): (A) IMPELLA-PL cohort; (B) Dresden cohort.
Effect of Impella on AKI
To assess the effect of the Impella pump on AKI, the observed AKI prevalence after hrPCI was compared to the predicted risk based on the Mehran risk score. AKI incidence was significantly lower than predicted in both the overall IMPELLA-PL and Dresden cohorts. Across different Mehran risk categories, there was a general tendency for actual risk to be lower than predicted, with a statistically significant difference observed in very high-risk IMPELLA-PL patients and moderate-risk Dresden patients (Fig. 2).
Predicted vs. observed rates of acute kidney injury in the overall groups and across renal risk categories based on the Mehran Risk Score: (A) IMPELLA-PL cohort; (B) Dresden cohort.
Since the majority of patients in the Dresden cohort received the Impella 2.5 device, its impact on AKI and survival was assessed in the pooled data analysis, which showed no significant effect on these outcomes (p = 0.47 and p = 0.11, respectively).
Determinants of AKI
Determinants of AKI in the IMPELLA-PL cohort were identified using univariable logistic regression to select predictor candidates from Table 1, followed by multivariable regression adjusted for age (Table 5). The model was then validated in the Dresden cohort (Table 6). In both cohorts, higher baseline eGFR and Impella pump implantation before hrPCI were independent determinants of a lower risk of AKI.
AKI in males and females
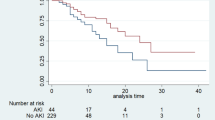
In a pooled analysis of 470 patients from both cohorts, AKI occurred in 53 of 373 males (14.2%) and 13 of 97 females (13.4%) (p = 1.00). During the 1-year follow-up, 86 males (23.1%) and 12 females (12.4%) died (p = 0.08, log-rank test). AKI was a significant predictor of 1-year mortality in males but not in females (Fig. 3). In a multivariable Cox regression analysis adjusted for age, LVEF ≤ 35%, and variables from the model in Table 3, AKI remained an independent predictor of mortality in males (HR: 2.16, 95% CI: 1.1–3.93, p < 0.05), whereas no such association was observed in females (HR: 2.52, 95% CI: 0.37–17.22, p = 0.35). Internal validation using bootstrapping (10,000 samples) confirmed the significant impact of AKI on mortality in males (p < 0.05) but not in females (p = 0.25). However, the interaction between sex and AKI in relation to 1-year survival was not statistically significant (p = 0.4).
Kaplan–Meier survival curves (unadjusted) comparing patients without acute kidney injury (No AKI) and with acute kidney injury (AKI): (A) Males; (B) Females.
The observed prevalence of AKI in males (14.2%) and females (13.4%) treated with the Impella pump was significantly lower than the predicted rate of 26.1% for both sexes based on the Mehran risk score (p < 0.001 and p < 0.05, respectively).
Sensitivity analysis with KDIGO criteria
Additional analyses were performed to assess the sensitivity of AKI’s predictive power to the KDIGO definition of AKI. According to the KDIGO criteria, AKI is defined as an increase in creatinine ≥ 0.3 mg/dL within 48 h after the procedure15. In line with these criteria, AKI was recognized in 22.9% of the IMPELLA-PL group and 24.9% of the Dresden group (p = 0.66). Applying the KDIGO criteria for the models presented in Tables 3 and 4, resulted in a borderline statistical significance of AKI’s predictive impact, with HR: 2.16 (95% CI: 0.98–4.73, p = 0.055) in the IMPELLA-PL cohort and HR: 1.75 (95% CI: 0.95–3.21, p = 0.073) in the Dresden cohort. A similar trend was observed in the pooled analysis of males, where the prognostic power of AKI using the KDIGO criteria became borderline (HR: 1.69, 95% CI: 0.97–2.93, p = 0.063), while it remained non-significant in females (HR: 1.62, 95% CI: 0.32–8.15, p = 0.56).
Discussion
This study shows that in patients without cardiogenic shock, AKI associated with complex PCI is a significant and independent determinant of one-year mortality. The use of the Impella pump in this setting reduced the actual risk of AKI compared to the expected risk based on the well-validated Mehran risk score. Upfront Impella implantation and preserved eGFR were identified as independent protective factors against AKI following high-risk PCI. The detrimental impact of AKI on survival was particularly pronounced in men.
The impact of mechanical circulatory support on AKI development is difficult to ascertain, primarily because MCS is often employed in the setting of cardiogenic shock, which itself is a strong risk factor for AKI16. In the DanGer Shock trial, the use of the Impella pump was associated with a significant increase in both AKI and the need for renal replacement therapy compared to the control arm4. In turn, both AKI and renal replacement therapy were associated with higher mortality at 180 days. Despite this, Impella use improved survival in the overall DanGer Shock cohort, including among patients who developed AKI and required renal replacement therapy. Further analysis revealed that Impella-related suction events, higher pump speed, and longer duration of support were associated with an increased risk of AKI in the Impella group5.
The renal effects of Impella in cardiogenic shock appear to contrast with our findings, where the prevalence of AKI in Impella-supported hrPCI was significantly lower than expected. Notably, the risk factors for AKI identified in the DanGer Shock trial were minimized during hrPCI in our cohorts. In particular, the AUTO mode of the Impella—the default setting during the first three hours of operation—might have mitigated suction events and appropriately limited pump speed. Moreover, the duration of support was generally shorter than that typically required in patients with cardiogenic shock. Additionally, mechanical circulatory support may have prevented severe hemodynamic instability and maintained renal perfusion during complex coronary interventions, thereby preserving renal function17. This mechanism likely explains why upfront Impella use before hrPCI was independently associated with a lower risk of AKI compared to bailout implantation during hemodynamic compromise. Furthermore, renal congestion—currently recognized as a central mechanism underlying worsening renal function in heart failure—may be mitigated by microaxial mechanical support. Through reductions in left ventricular filling pressures and central venous congestion, this approach may improve renal perfusion and hemodynamic stability17,18,19. Preclinical data further support this hypothesis, indicating that Impella-mediated ventricular unloading is associated with reduced renal sympathetic activity and improved renal blood flow20.
In the general PCI patient population, the incidence of acute kidney injury (AKI) is estimated to be approximately 7%16. In high-risk groups, the prevalence of AKI is expected to be considerably higher, as demonstrated in our study, where the anticipated risk was around 26%. However, the actual incidence was significantly lower in both the derivation and validation cohorts (13.3% and 14.9%, respectively), suggesting a potential renoprotective effect of the Impella pump. Recently, Flaherty et al. reported a significant reduction in AKI incidence compared with the predicted risk in patients undergoing high-risk PCI (hrPCI) with Impella support. However, they did not assess the impact of AKI on patient prognosis7. In our population treated with Impella-supported hrPCI, AKI was associated with significantly worse survival over the one-year observation period. Previous studies in the general PCI population have demonstrated that AKI—particularly when resulting in persistent renal dysfunction—is a significant risk factor for both short- and long-term mortality16,21,22. The occurrence of AKI following PCI in the setting of acute coronary syndrome has also been linked to higher rates of target lesion revascularization, major bleeding, and ultimately, increased one-year mortality23. Nevertheless, the impact of AKI on survival following interventions with Impella support has not been previously reported. Furthermore, the mechanisms underlying the long-term effects of AKI on mortality remain incompletely understood. AKI may contribute to persistent or progressive kidney function loss, thereby increasing the risk of end-stage renal disease in the future15,21,24. In turn, renal dysfunction may promote the development of coronary artery disease, further elevating the risk of death25.
In our study, the detrimental prognostic impact of AKI was more pronounced in males than in females, despite a similar incidence of AKI in both sexes. However, these findings should be considered exploratory and interpreted with caution, as the female subgroup experienced very few adverse events (13 cases of AKI and 12 deaths), resulting in wide confidence interval (HR 2.52, 95% CI 0.37–17.22). Therefore, the lack of statistical significance in women may reflect limited statistical power rather than a true absence of effect. Nevertheless, this observation warrants further investigation, as men have been shown to be at higher risk of developing AKI compared with women. This difference persists even after adjustment for age, baseline eGFR, and comorbidities and has been associated, at least in part, with increased mortality8. Experimental and clinical studies suggest that the protective effects of estrogen in women—particularly during the reproductive period—may contribute to sex-related differences in renal vulnerability26.
The definition of AKI in this study corresponded to that employed in Mehran risk score in order to enable a comparison between the observed prevalence of AKI after hrPCI and the predicted risk based on the Mehran risk score. In the sensitivity analysis, a lower threshold for serum creatinine increase (≥ 0.3 mg/dL), combined with a shorter detection window (48 h) as defined by the KDIGO criteria, demonstrated weaker predictive power of AKI compared to the threshold used in our study (≥ 0.5 mg/dL within 72 h). These findings align with previous reports indicating that the risk of long-term adverse outcomes increases progressively with the severity of post-PCI AKI24. Accordingly, identifying more severe renal impairment over an extended detection period (comparing to KDIGO criteria) provided stronger prognostic value in our analysis. Although Impella use appears to mitigate the risk of kidney injury, properly defining the cutoff point for clinically relevant AKI remains essential for appropriate patient surveillance.
The impact of AKI on long-term survival following Impella-supported hrPCI has not been previously reported, and our study provides new insight into the prognostic significance of acute renal complications in this setting. In particular, it presents evidence supporting a potential protective effect of Impella against AKI during complex coronary interventions in patients primarily without cardiogenic shock.
Limitations
Despite its important findings, this study has several limitations. First, its observational design limits the establishment of a causal relationship between Impella use and the reduced incidence of AKI. The Mehran score may overestimate the predicted risk of AKI in our study, and therefore, the comparison between predicted and observed AKI rates should be interpreted cautiously and considered exploratory. Nonetheless, using the Mehran risk score allowed us to place our findings within a well-established risk framework and to provide indirect insight into the potential impact of Impella support on renal outcomes in this high-risk population.
Second, the study population consisted primarily of patients without cardiogenic shock, which limits the generalizability of the results to broader high-risk populations treated with the Impella pump.
Third, unmeasured confounders—such as hydration protocols and operator technique—may have influenced renal outcomes and survival, underscoring the need for prospective, randomized studies to validate these findings.
Fourth, the observed sex differences in the prognostic impact of AKI should be considered exploratory only, as the very limited number of events in the female group may have resulted in insufficient statistical power.
Fifth, baseline renal function was assessed using the MDRD equation; although CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) formula is currently recommended15, the retrospective nature of the dataset required consistency with the originally recorded variables.
Finally, although the decision to perform hrPCI with Impella support incorporated objective clinical and anatomical high-risk features, the absence of prospectively predefined and uniformly applied standardized criteria represents a limitation, as it may introduce selection bias and limit full reproducibility. Nevertheless, the overall robustness of the findings was supported by the use of separate derivation and validation cohorts.
Conclusions
Acute kidney injury following complex PCI is a significant and independent risk factor for one-year mortality, with the risk probably pronounced in males. The use of Impella during PCI may potentially reduce the risk of AKI, with the greatest benefit observed when the device is implanted prior to coronary intervention.
Data availability
The data underlying this article will be shared on reasonable request to the first and last author with the allowance of the participating investigators.
References
Tsai, T. T. et al. Contemporary incidence, predictors, and outcomes of acute kidney injury in patients undergoing percutaneous coronary interventions: Insights from the NCDR Cath-PCI registry. JACC Cardiovasc. Interv. 7, 1–9 (2014).
Mehran, R. et al. A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: Development and initial validation. J. Am. Coll. Cardiol. 44, 1393–1399 (2004).
Van Edom, C. J. et al. Management of bleeding and hemolysis during percutaneous microaxial flow pump support: A practical approach. JACC Cardiovasc. Interv. 16, 1707–1720 (2023).
Møller, J. E. et al. Microaxial flow pump or standard care in infarct-related cardiogenic shock. N. Engl. J. Med. 390, 1382–1393 (2024).
Zweck, E. et al. Microaxial flow pump use and renal outcomes in infarct-related cardiogenic shock: A secondary analysis of the DanGer shock trial. Circulation 150, 1990–2003 (2024).
Flaherty, M. P. et al. Hemodynamic Support with a microaxial percutaneous left ventricular assist device (impella) protects against acute kidney injury in patients undergoing high-risk percutaneous coronary intervention. Circ Res. 120, 692–700 (2017).
Flaherty, M. P. et al. Impella support and acute kidney injury during high-risk percutaneous coronary intervention: The global cVAD renal protection study. Catheter Cardiovasc. Interv. 95, 1111–1121 (2020).
Loutradis, C. et al. Acute kidney injury is more common in men than women after accounting for socioeconomic status, ethnicity, alcohol intake and smoking history. Biol. Sex Differ. 12, 30 (2021).
Pietrasik, A. et al. Mechanical circulatory support for high-risk percutaneous coronary interventions and cardiogenic shock: Rationale and design of the multicenter, investigator-initiated IMPELLA-PL registry. Cardiol. J. 29, 866–871 (2022).
Pietrasik, A. et al. Multicenter registry of Impella-assisted high-risk percutaneous coronary interventions and cardiogenic shock in Poland (IMPELLA-PL). Kardiol. Pol. 81, 1103–1112 (2023).
Mierke, J. et al. Sex-related differences in outcome of patients treated with microaxial percutaneous left ventricular assist device for cardiogenic shock. Heart Lung Circ. 33, 1670–1679 (2024).
Mangner, N. et al. DanGer shock-like profile predicts the outcome in ST-elevation myocardial infarction-related cardiogenic shock. ESC Heart Fail. https://doi.org/10.1002/ehf2.15269 (2025) (Epub ahead of print).
Weisbord, S. D. et al. Outcomes after angiography with sodium bicarbonate and acetylcysteine. N. Engl. J. Med. 378, 603–614 (2018).
Brar, S. S. et al. Haemodynamic-guided fluid administration for the prevention of contrast-induced acute kidney injury: The POSEIDON randomised controlled trial. Lancet 383, 1814–1823 (2014).
Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO. Clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013(3), 1–150 (2012).
Uzendu, A. et al. Contemporary methods for predicting acute kidney injury after coronary intervention. JACC Cardiovasc. Interv. 16, 2294–2305 (2023).
Patsalis, N. et al. Renal protection and hemodynamic improvement by Impella® Microaxial pump in patients with cardiogenic shock. J. Clin. Med. 11, 6817 (2022).
Tamayo-Gutierrez, A. & Ibrahim, H. N. The kidney in heart failure: The role of venous congestion. Methodist Debakey Cardiovasc. J. 18, 4–10 (2022).
Raina, R. et al. An update on the pathophysiology and treatment of cardiorenal syndrome. Cardiol. Res. 11, 76–88 (2020).
Warnakulasuriya, T., George, B., Lever, N. & Ramchandra, R. Mechanical circulatory support reduces renal sympathetic nerve activity in an ovine model of acute myocardial infarction. Clin. Auton. Res. 35, 193–203 (2025).
Nakamura, T. et al. Prognostic impact and predictors of persistent renal dysfunction in acute kidney injury after percutaneous coronary intervention for acute myocardial infarction. Sci. Rep. 14, 6299 (2024).
Jiang, M. Y. Impact of acute kidney injury and baseline renal impairment on prognosis among patients undergoing percutaneous coronary intervention. Acta Cardiol. Sin. 36, 223–232 (2020).
Giacoppo, D. et al. Impact of contrast-induced acute kidney injury after percutaneous coronary intervention on short- and long-term outcomes: Pooled analysis from the HORIZONS-AMI and ACUITY trials. Circ. Cardiovasc. Interv. 8, e002475 (2015).
James, M. T. et al. Associations between acute kidney injury and cardiovascular and renal outcomes after coronary angiography. Circulation 123, 409–416 (2011).
Di Angelantonio, E. et al. Chronic kidney disease and risk of major cardiovascular disease and non-vascular mortality: Prospective population based cohort study. BMJ 341, c4986 (2010).
Golestaneh, L. et al. Sex, acute kidney injury, and age: A prospective cohort study. Am. J. Kidney Dis. 85, 329-338.e1 (2025).
Author information
Authors and Affiliations
Contributions
J.S. collected and analyzed the data as well as wrote the main manuscript; N.M. collected and analyzed the data as well as reviewed and corrected the manuscript; F.W., A.P., A.L., A.G., A.C., T.P., D.O., M.G., S.H., G.B., J.M., W.B., S.J., A.S., P.B., A.W., M.W., Ł.R., S.B., A.P., M.K., R.G., W.K., K.R., M.P., P.K., P.W., S.G., A.K., M.Ł., W.S., M.Z., P.P., S.W., A.W., K.Ś., A.T., M.P., J.H., K.K., M.H., M.G., J.Z.P., M.F., R.R., P.K., D.C., A.O., W.W., J.L., A.L., and J.K. collected the data and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.S. is an Impella proctor and has received consulting fees from Abiomed / Johnson & Johnson. A.P. Principal Investigator of the Impella-PL Registry, has received an unrestricted educational grant from Abiomed and has served on the speakers bureau for Abiomed. J.L. has received consulting fees from Abiomed / Johnson & Johnson. A.L. reports honoraria or consultancy fees from Edwards Lifesciences, Boston Scientific, Abiomed, Novartis, Pfizer, Astra Zeneca, Boehringer, Abbott, MSD, Corvia, Daiichi and Meril; received grant support form Edwards Lifesciences and Novartis, and owns stock options from Transverse Medical, Picardia and Filterlex. N.M. has received an educational and research grant from Abiomed to his institution, outside the submitted work; has received an educational grant from Boston Scientific to his institution, outside the submitted work; and has received personal fees from Abbott, Abiomed, Amgen, AstraZeneca, B. Braun, Biotronik, Boston Scientific, Cordis, Edwards Lifesciences, Inari, Medtronic, Pfizer/BMS, and Shockwave, outside the submitted work. Other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sacha, J., Woitek, F.J., Pietrasik, A. et al. Prognostic impact of acute kidney injury on one-year survival in patients undergoing high-risk PCI with Impella support. Sci Rep 16, 10625 (2026). https://doi.org/10.1038/s41598-026-44724-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-44724-w