Abstract
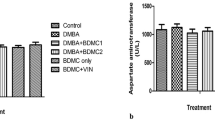
7,12-Dimethylbenz[a]anthracene (DMBA) induces reactive oxygen species (ROS) formation, oxidative stress, and apoptosis in target tissues. Royal jelly (RJ) is rich in flavonoids, phenolic acids, and peptides. This study evaluated whether RJ can mitigate DMBA-induced oxidative, apoptotic, and histopathological changes in the rat mammary gland and skin. Female Wistar rats were divided into four groups, including the Control (no treatment), RJ (300 mg/kg/week, orally), DMBA (single dose 80 mg/kg, IP), and DMBA + RJ (same doses). After 8 weeks, mammary gland and skin specimens were collected. Oxidative stress markers were quantified in tissue homogenates. Apoptotic gene and protein expression were measured by qRT-PCR and Western blot, respectively. Ki67 expression was assessed by immunohistochemistry. Additionally, histopathological evaluation (H&E stain) was conducted. DMBA significantly increased MDA, reduced GPx, SOD, and TAC. Pro-apoptotic markers were upregulated, while Bcl2 was downregulated. Histopathology revealed vacuolar degeneration, necrosis, mitotic figures, and corpora amylacea in the mammary gland, and epidermal hyperplasia, mitotic figures, follicular hyperplasia, and focal necrosis in skin. RJ co-treatment restored GPx and SOD levels to almost those of the control group, reduced MDA, decreased p53 and Bax to near-control levels, and increased Bcl2 to the approximate level of the control group. Moreover, RJ treatment normalized the mammary gland histologically, while skin showed attenuated necrosis and decreased epidermal hyperplasia and mitotic index. RJ effectively countered DMBA-induced oxidative stress and apoptosis in the rat mammary gland while preserving tissue integrity. In skin, RJ mitigated oxidative damage and reduced proliferation. These outcomes suggest RJ’s potential as an adjuvant antioxidant in glandular disorders, while showing the anticarcinogenic potential.
Similar content being viewed by others
Data availability
Data are available upon request from the corresponding author.
References
Hakkak, R. & Melnyk, S. Soy protein diet changes metabolic profile in liver of 7, 12-dimethylbenz (α) anthracene (DMBA)-induced mammary tumors in obese zucker rats. Cancer Res. 84 (6_Supplement), 759–759 (2024).
Lim, E. L. & DeGregori, J. The Nature and Nurture of Carcinogenesis. Cancer Discov. 15 (6), 1090–1092 (2025).
Conney, A. H. Induction of microsomal enzymes by foreign chemicals and carcinogenesis by polycyclic aromatic hydrocarbons: G. H. A. Clowes Memorial Lecture. Cancer Res. 42 (12), 4875–4917 (1982).
Kensler, T. W. et al. Aflatoxin: a 50-year odyssey of mechanistic and translational toxicology. Toxicol. Sci. 120 (suppl_1), S28–S48 (2011).
Das, I. & Saha, T. Effect of garlic on lipid peroxidation and antioxidation enzymes in DMBA-induced skin carcinoma. Nutrition 25 (4), 459–471 (2009).
Calaf, G. M. et al. Protective role of curcumin in oxidative stress of breast cells. Oncol. Rep. 26 (4), 1029–1035 (2011).
Mohamed, H. K. et al. Anti-inflammatory, anti-apoptotic, and antioxidant roles of honey, royal jelly, and propolis in suppressing nephrotoxicity induced by doxorubicin in male albino rats. Antioxidants 11 (5), 1029 (2022).
Skrajnowska, D. et al. Copper and resveratrol attenuates serum catalase, glutathione peroxidase, and element values in rats with DMBA-induced mammary carcinogenesis. Biol. Trace Elem. Res. 156, 271–278 (2013).
Elmore, S. Apoptosis: a review of programmed cell death. Toxicol. Pathol. 35 (4), 495–516 (2007).
Chan, T. K. et al. Polycyclic aromatic hydrocarbons regulate the pigmentation pathway and induce DNA damage responses in keratinocytes, a process driven by systemic immunity. J. Dermatol. Sci. 104 (2), 83–94 (2021).
Ayala, A., Muñoz, M. F. & Argüelles, S. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy‐2‐nonenal. Oxidative Med. Cell. Longev. 2014 (1), 360438 (2014).
Bergmann, A. & Steller, H. Apoptosis, stem cells, and tissue regeneration. Sci. Signal. 3 (145), re8–re8 (2010).
Pavel, C. I. et al. Biological activities of royal jelly-review. Sci. Papers Anim. Sci. Biotechnologies. 44 (2), 108–108 (2011).
Khazaei, M., Ansarian, A. & Ghanbari, E. New findings on biological actions and clinical applications of royal jelly: a review. J. Diet. supplements. 15 (5), 757–775 (2018).
Vazhacharickal, P. J. A review on health benefits and biological action of honey, propolis and royal jelly. J. Med. Plants Stud. 9 (5), 1–13 (2021).
Aslan, A. et al. Royal jelly arranges apoptotic and oxidative stress pathways and reduces damage to liver tissues of rats by down-regulation of Bcl-2, GSK3 and NF-κB and up-regulation of caspase and Nrf-2 protein signalling pathways. Biomarkers 28 (2), 217–226 (2023).
Karadeniz, A. et al. Royal jelly modulates oxidative stress and apoptosis in liver and kidneys of rats treated with cisplatin. Oxidative Med. Cell. Longev. 2011 (1), 981793 (2011).
Beigom Hejazian, L., Hosseini, S. M. & Salehi, A. Neuroprotective Effects of Rosa damascena Extract against Aluminum Chloride-Induced Brain Damage in Rat Offspring. Neurol. Res. Int. 2023 (1), 5342849 (2023).
Shakib Khoob, M., Hosseini, S. M. & Kazemi, S. In vitro and in vivo antioxidant and anticancer potentials of royal jelly for dimethylhydrazine-induced colorectal cancer in Wistar rats. Oxidative Med. Cell. Longev. 2022 (1), 9506026 (2022).
Wang, X., Yuwen, T. & Yanqin, T. Mangiferin inhibits inflammation and cell proliferation, and activates proapoptotic events via NF-κB inhibition in DMBA-induced mammary carcinogenesis in rats. J. Environ. Pathol. Toxicol. Oncol., 40(2). (2021).
Wellington, D., Mikaelian, I. & Singer, L. Comparison of ketamine–xylazine and ketamine–dexmedetomidine anesthesia and intraperitoneal tolerance in rats. J. Am. Assoc. Lab. Anim. Sci. 52 (4), 481–487 (2013).
Percie du Sert, N. et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. J. Cereb. Blood Flow. Metabolism. 40 (9), 1769–1777 (2020).
Valaei, A. et al. Antioxidant and Anticancer Potentials of the Olive and Sesame Mixture against Dimethylhydrazine-Induced Colorectal Cancer in Wistar Rats. Biomed. Res. Int. 2022 (1), 5440773 (2022).
Weiss, S. L. & Deutschman, C. S. Elevated malondialdehyde levels in sepsis-something to’stress’ about? Crit. Care. 18, 1–2 (2014).
Krishnamurthy, H. K. et al. Oxidative stress: fundamentals and advances in quantification techniques. Front. Chem. 12, 1470458 (2024).
Pei, J. et al. Research progress of glutathione peroxidase family (GPX) in redoxidation. Front. Pharmacol. 14, 1147414 (2023).
Jafari, A. et al. Effect of exercise training on Bcl-2 and bax gene expression in the rat heart. Gene Cell. Tissue, 2(4). (2015).
Saaed, H. K., Mahmood, M. A. & Khoshnaw, N. Quantitative real time PCR analysis of apoptotic gene expression in chronic lymphocytic leukemia patients and their relationships with chemosensitivity. Appl. Cancer Res. 37, 1–7 (2017).
Salehi, A., Hosseini, S. M. & Kazemi, S. Antioxidant and Anticarcinogenic Potentials of Propolis for Dimethylhydrazine-Induced Colorectal Cancer in Wistar Rats Vol. 2022, p. 8497562 (BioMed Research International, 2022). 1.
Saxena, R. & Darnell, A. J. Proliferation in the normal gastrointestinal tract: an immunohistochemical study with Ki-67. J. Histotechnology. 35 (1), 22–26 (2012).
Salehi, A., Hosseini, S. M. & Kazemi, S. Propolis ameliorates renal, liver, and pancreatic lesions in Wistar rats (Biotechnology and Applied Biochemistry, 2024).
Rahimi, O. et al. Hepatorenal protective effects of hydroalcoholic extract of Solidago canadensis L. against paracetamol-induced toxicity in mice. J. Toxicol. 2022 (1), 9091605 (2022).
Beyaz, S. et al. Fullerene C60 protects against 7, 12-dimethylbenz [a] anthracene (DMBA) induced-pancreatic damage via NF-κB and Nrf-2/HO-1 axis in rats. Toxicol. Res. 12 (5), 954–963 (2023).
Stevanović, J. et al. Bee-inspired healing: apitherapy in veterinary medicine for maintenance and improvement animal health and well-being. Pharmaceuticals 17 (8), 1050 (2024).
Malkoç, M. et al. The effects of royal jelly on the oxidant-antioxidant system in rats with N-methyl-N-nitrosourea-induced breast cancer. Turkish J. Biochem. 43 (2), 176–183 (2018).
Baptista, B. G. et al. Royal jelly: a predictive, preventive and personalised strategy for novel treatment options in non-communicable diseases. EPMA J. 14 (3), 381–404 (2023).
Rizki, A. M. F. et al. Effect of royal jelly to deal with stress oxidative in preconception women: A literature review. Gac. Sanit. 35, S288–S290 (2021).
Sindi, A. A. Thymoquinone decreases oxidative DNA damage (8-OHdG) in DMBA treated female Sprague Dawley rats. (2013).
Yin, C. et al. Bax suppresses tumorigenesis and stimulates apoptosis in vivo. Nature 385 (6617), 637–640 (1997).
Knudson, C. M. et al. Bax accelerates tumorigenesis in p53-deficient mice. Cancer Res. 61 (2), 659–665 (2001).
Hamza, A. A. et al. Salvadora persica attenuates DMBA-induced mammary cancer through downregulation oxidative stress, estrogen receptor expression and proliferation and augmenting apoptosis Vol. 147, p. 112666 (Biomedicine & Pharmacotherapy, 2022).
Lin, Y. et al. The in vitro and in vivo wound-healing effects of royal jelly derived from Apis mellifera L. during blossom seasons of Castanea mollissima Bl. and Brassica napus L. in South China exhibited distinct patterns. BMC Complement. Med. Ther. 20, 1–13 (2020).
Balmain, A. & Yuspa, S. H. Milestones in skin carcinogenesis: the biology of multistage carcinogenesis. J. Invest. Dermatol. 134 (e1), E2–7 (2014).
Yapar, K. et al. Protective effect of royal jelly and green tea extracts effect against cisplatin-induced nephrotoxicity in mice: a comparative study. J. Med. Food. 12 (5), 1136–1142 (2009).
Funding
No funding source has impacted the objectivity or integrity of this work.
Author information
Authors and Affiliations
Contributions
Alireza Salehi and Fatemeh Yaghoobi: Conceptualization, Methodology, Software, Writing original draft preparation, and investigation. Seyed Mohammad Hosseini: Data curation and Visualization. Seyedeh-Sara Hashemi: Supervision, Software, Validation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
All experiments were conducted following the International Council for Laboratory Animal Science guidelines. Additionally, the ethical approval for the study was granted by the Research Ethics Committees of Shiraz University of Medical Science with the approval ID IR.SUMS.REC.1404.402.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Salehi, A., Yaghoobi, F., Hosseini, S.M. et al. Royal jelly attenuates DMBA induced preneoplastic lesions in rat mammary gland and Skin. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44968-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44968-6