Abstract
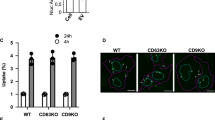
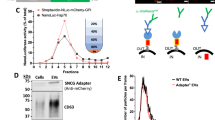
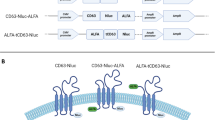
Extracellular vesicles (EVs), including exosomes, are widely believed to mediate intercellular communication by delivering molecular cargo into the cytosol of recipient cells. However, the efficiency and mechanisms of such cargo delivery remain unclear. In this study, we investigated whether EVs bearing the canonical tetraspanin EVs marker CD63 are capable of fusing with recipient cell membranes to release their contents. Using the highly sensitive NanoBiT split luciferase system, we tracked potential fusion events between HiBiT-tagged CD63-EVs and HEK293 cells expressing cytosolic or endosome-localized LgBiT. While HiBiT and LgBiT reconstitution was readily detected when EVs carried the viral fusion protein VSV-G, no luminescence was observed with HiBiT tagged CD63-EVs in absence of VSV-G, despite their uptake or binding by recipient cells. Similarly, EVs carrying HiBiT-HSP70 failed to show evidence of cargo release. These findings demonstrate that unmodified HiBiT-CD63-EVs do not fuse with the plasma or endosomal membranes of recipient cells, suggesting that EV-mediated cargo delivery via membrane fusion is inefficient or absent under standard culture conditions. Our results challenge the prevailing view of EV function in intercellular communication and underscore the need for re-evaluation of EV cargo transfer mechanisms.
Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Jeppesen, D. K. et al. Reassessment of exosome composition. Cell 177, 428–445 e418. https://doi.org/10.1016/j.cell.2019.02.029 (2019).
Joshi, B. S., de Beer, M. A., Giepmans, B. N. G. & Zuhorn, I. S. Endocytosis of Extracellular Vesicles and Release of Their Cargo from Endosomes. ACS nano. 14, 4444–4455. https://doi.org/10.1021/acsnano.9b10033 (2020).
Bonsergent, E. et al. Quantitative characterization of extracellular vesicle uptake and content delivery within mammalian cells. Nat. Commun. 12, 1864. https://doi.org/10.1038/s41467-021-22126-y (2021).
O’Brien, K., Ughetto, S., Mahjoum, S., Nair, A. V. & Breakefield, X. O. Uptake, functionality, and re-release of extracellular vesicle-encapsulated cargo. Cell Rep. 39, 110651. https://doi.org/10.1016/j.celrep.2022.110651 (2022).
Verweij, F. J. et al. Quantifying exosome secretion from single cells reveals a modulatory role for GPCR signaling. J. Cell. Biol. 217, 1129–1142. https://doi.org/10.1083/jcb.201703206 (2018).
Mathieu, M. et al. Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat. Commun. 12, 4389. https://doi.org/10.1038/s41467-021-24384-2 (2021).
Nakase, I. & Futaki, S. Combined treatment with a pH-sensitive fusogenic peptide and cationic lipids achieves enhanced cytosolic delivery of exosomes. Sci. Rep. 5, 10112. https://doi.org/10.1038/srep10112 (2015).
Chivet, M. et al. Exosomes secreted by cortical neurons upon glutamatergic synapse activation specifically interact with neurons. J. Extracell. vesicles. 3, 24722. https://doi.org/10.3402/jev.v3.24722 (2014).
Heusermann, W. et al. Exosomes surf on filopodia to enter cells at endocytic hot spots, traffic within endosomes, and are targeted to the ER. J. Cell. Biol. 213, 173–184. https://doi.org/10.1083/jcb.201506084 (2016).
Sasaki, M. et al. Development of a rapid and quantitative method for the analysis of viral entry and release using a NanoLuc luciferase complementation assay. Virus Res. 243, 69–74. https://doi.org/10.1016/j.virusres.2017.10.015 (2018).
Yamamoto, M. et al. Cell-cell and virus-cell fusion assay-based analyses of alanine insertion mutants in the distal alpha9 portion of the JRFL gp41 subunit from HIV-1. J. Biol. Chem. 294, 5677–5687. https://doi.org/10.1074/jbc.RA118.004579 (2019).
Sung, B. H. et al. A live cell reporter of exosome secretion and uptake reveals pathfinding behavior of migrating cells. Nat. Commun. 11, 2092. https://doi.org/10.1038/s41467-020-15747-2 (2020)
Hoare, B. L., Kocan, M., Bruell, S., Scott, D. J. & Bathgate, R. A. D. Using the novel HiBiT tag to label cell surface relaxin receptors for BRET proximity analysis. Pharmacol. Res. Perspect. 7, e00513. https://doi.org/10.1002/prp2.513 (2019).
Mathew, A., Bell, A. & Johnstone, R. M. Hsp-70 is closely associated with the transferrin receptor in exosomes from maturing reticulocytes. Biochem. J. 308(Pt 3), 823–830. https://doi.org/10.1042/bj3080823 (1995).
Zhang, J. et al. Genetically engineered extracellular vesicles harboring transmembrane scaffolds exhibit differences in their size, expression levels of specific surface markers and cell-uptake. Pharmaceutics. https://doi.org/10.3390/pharmaceutics14122564 (2022).
Votteler, J. et al. Designed proteins induce the formation of nanocage-containing extracellular vesicles. Nature 540, 292–295. https://doi.org/10.1038/nature20607 (2016).
Somiya, M. & Kuroda, S. Real-time luminescence assay for cytoplasmic cargo delivery of extracellular vesicles. Anal. Chem. 93, 5612–5620. https://doi.org/10.1021/acs.analchem.1c00339 (2021).
Perrin, P. et al. Retrofusion of intralumenal MVB membranes parallels viral infection and coexists with exosome release. Curr. Biol. 31, 3884–3893. https://doi.org/10.1016/j.cub.2021.06.022 (2021). e3884.
de Jong, O. G. et al. A CRISPR-Cas9-based reporter system for single-cell detection of extracellular vesicle-mediated functional transfer of RNA. Nat. Commun. 11, 1113. https://doi.org/10.1038/s41467-020-14977-8 (2020).
Castro-Cruz, M. et al. PDZ scaffolds regulate extracellular vesicle production, composition, and uptake. Proc. Natl. Acad. Sci. U S. A120, e2310914120. https://doi.org/10.1073/pnas.2310914120 (2023).
Acknowledgements
We thank Rose-Laure Revel-Goyet, Oleksandr Glushonkov, Jean-Philippe Kleman (Institut de Biologie Structurale, Grenoble) for the support and access to the Cell imaging Platform, Delphine Guilligay (Institut de Biologie Structurale, Grenoble) for TEM of the EVs.We thank B. Blot for helping with some of the cDNA constructs, M.O. Fauvarque, J. Fauré, S. Fraboulet for helpful discussions throughout this work. We also thank K. Sadoul for critically reading the manuscript.
Funding
We acknowledge support from the ‘Fondation pour la Recherche Medicale (FRM, Equipe FRM 2023, project EQU202303016333) and access to the platforms of the Grenoble Instruct-ERIC center (IBS and ISBG; UAR 3518 CNRS-CEA-UGA-EMBL) within the Grenoble Partnership for Structural Biology (PSB), with support from FRISBI (ANR-10-INBS-05-02) and GRAL, a project of the University Grenoble Alpes graduate school (EcolesUniversitaires de Recherche) CBH-EUR-GS (ANR-17-EURE-0003). A.A.S. was supported by funding from GRAL. IBS acknowledges integration into the Interdisciplinary Research Institute of Grenoble (IRIG, CEA) and financial support from CEA, CNRS and UGA. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
A.A.S. performed the EV purification and characterization (NTA), did most of NanoLuc assays, made DNA constructions, W.W. and R.S. funding acquisition, R.S. supervised the project and wrote the manuscript, C.C. performed all experiments with VSV-G-EVs, made statistical analysis and co-supervised the project. A.A.S. and C.C. writing original draft preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Askarian-Amiri, S., Weissenhorn, W., Sadoul, R. et al. Lack of evidence for cargo release of CD63-EVs into recipient cells. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45021-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45021-2