Abstract
Multi-isotope analyses of extant and extinct animals provide key insights into dietary ecology. Here, we examined mineral-bound nitrogen isotopes alongside carbonate carbon and oxygen stable isotope ratios in tooth enamel from sympatric chimpanzees, other primates (yellow baboons, red-tailed, and red colobus monkeys), herbivores, omnivores, and a radicivore from the miombo woodlands of the Issa Valley, Tanzania. Chimpanzees occupy a distinct isotopic niche characterized by low nitrogen and carbon isotope values and enriched oxygen isotope values relative to other primates. This unique isotopic niche likely reflects their reliance on termites as a major source of dietary protein. Our data indicate that termites contribute substantially to the chimpanzee’s nitrogen intake (at least 50%), consistent with nitrogen isotope values of chimpanzees. These findings from woodland-dwelling primates demonstrate the power of multi-isotope approaches for reconstructing modern and past dietary ecology and highlight their potential for interpreting ancient diets, including those of hominins.
Similar content being viewed by others
Introduction
Stable isotope analysis provides an integrated record of dietary behavior over months to years and is a powerful tool for reconstructing feeding ecology, physiology, and environmental context in both modern and fossil animals. Because the behavior of extinct species cannot be directly observed, modern ecosystems offer essential reference frameworks for interpreting fossil isotope data and tracing evolutionary adaptations in diet and behavior1,2. Such analogues are particularly valuable for understanding the ecology of hominins, as some modern primates share deep evolutionary histories and key physiological and ecological traits with early hominins. Chimpanzees (Pan troglodytes) – and to a lesser extent some baboon taxa (Papio spp.) – approximate hominin body size (e.g., Sahelanthropus, Ardipithecus, Australopithecus)3,4, and chimpanzees exhibit complex foraging behaviors also attributed to hominins, including the ability to select and modify raw materials to produce tools for specific tasks such as termite fishing or nut cracking5. These behaviors provide insight into the cognitive and ecological foundations of tool use and diet in our lineage6,7,8.
Isotopic studies of modern ecosystems typically analyze biological materials, including body fluids, soft tissues, or collagen from bone or dentin9,10. However, these materials are rarely preserved over geologic timescales, making diagenetically robust tooth enamel a preferred archive for paleodietary reconstructions. While stable carbon (δ¹³Cenamel) and oxygen (δ¹⁸Oenamel) isotope values have been routinely measured in the inorganic fraction of enamel for decades, nitrogen isotope (δ¹⁵Nenamel) analysis has been far more challenging because enamel contains only trace amounts of nitrogen. This small nitrogen fraction has historically been difficult to extract and measure, limiting δ15Nenamel applications in fossil teeth despite enamel’s excellent preservation potential11,12. Recent methodological advances now allow δ¹⁵Nenamel to be measured reliably from enamel’s organic fraction13, greatly expanding the potential to link isotopic patterns in modern ecosystems to reconstructions of trophic ecology of the past14,15,16,17,18.
Nitrogen isotope ratios of body tissues are enriched relative to diet due to the excretion of 15N-depleted nitrogenous waste products, primarily ammonium, urea and uric acid, as well as feces and sweat2. This enrichment – typically 3 to 6‰ – has been quantified in experimental as well as ecological studies13,17,18,19,20 and permits reconstruction of trophic position from tissues and enamel.
Complementary, stable carbon isotopes in animal tissues are a well-established tool for reconstructing plant based-diets21. In African savanna ecosystems, which often contain a mosaic of plant types, δ13C values are primarily influenced by the proportion of C3 versus C4 plants consumed. Most dicots (such as trees and shrubs) use the C3 photosynthetic pathway, which results in δ13C values ranging from −22 to −37‰, with an average of −27‰22. Monocots (primarily grasses), which are well adapted to warm, arid environments, use the C4 photosynthetic pathway and typically have δ13C values ranging from −9 to −15‰, with an average of −12.5‰. A fractionation of ca. +14‰ between diet and enamel has been observed for large herbivores23,24 with a slightly smaller fractionation of +11.8‰ documented for chimpanzees25.
Oxygen isotopes primarily reflect the δ¹⁸O of body water, which is influenced by the isotopic composition of drinking, food, and metabolic water. These values vary with environmental factors such as temperature, rainfall, altitude, and evaporation, as well as species-specific drinking behavior. In seasonal environments, δ¹⁸Oenamel profiles can also record wet-dry cycles during tooth formation26,27,28.
By understanding how δ15Nenamel, δ13Cenamel, and δ18Oenamel values vary with diet, trophic position, water sources, and physiological processes, we can interpret the isotopic niches of different taxa within their ecological and environmental contexts, using modern primates as analogs for reconstructing the dietary behavior of extinct hominids29,30. To explore these relationships, we focused on the Issa Valley in western Tanzania, a savanna-woodland mosaic of open C4-dominated habitats interspersed with C3-rich riparian forest31(Fig. 1). Paleohabitat reconstructions suggest that such habitats resemble those inhabited by hominids during key periods in early human evolution32,33,34, making Issa an ideal modern analog for investigating diet-environment interactions. Reconstructing hominid paleodiets requires integrated isotopic data for consumers and their environment. Here, we analyze nitrogen, carbon, and oxygen isotopes derived from tooth enamel of sympatric wild eastern chimpanzees (Pan troglodytes schweinfurthii), yellow baboons (Papio cynocephalus), and other mammalian fauna from Issa to evaluate how enamel isotopes reflect dietary and ecological patterns across taxa. Chimpanzees in this community have been the focus of long-term research addressing key questions about their feeding ecology. Previous studies combined stable isotope analyses of chimpanzee soft tissues with δ15N data from locally collected plants35 to investigate trophic enrichment compared to their diet.
Location of Issa Valley (red star) in western Tanzania (a), location on the African continent (b), and vegetation zones31(c).
Primates play an important ecological role in ecosystems, often influencing plant dispersal and habitat structure. The eastern chimpanzee diet is highly seasonal: during the wet season, Issa chimpanzees rely heavily on ripe fruits, seeds, flowers and leaves from at least 69 identified plant species, primarily sourced from within the riparian forests; whereas in the dry season, they turn to unripe seed pods from miombo woodlands35. At Issa, as elsewhere, chimpanzees are largely frugivorous, but they also use flexible probe tools to fish for termites, particularly for Macrotermes subhyalinus36. Individual chimpanzees may engage in termite fishing for extended periods of time during the rainy season, when soldier termites are particularly active near the mound surface37,38,39. Termite consumption and termite fishing is a low-risk protein resource that is regularly consumed, compared to vertebrate meat, which is also protein rich, but eaten more rarely.
Issa chimpanzees also consume mammalian prey, both from opportunistic scavenging and active hunting40,41. Prior to habituation of the Issa community in 2018, evidence of meat consumption based on fecal analysis was limited, despite chimpanzees routinely encountering potential prey42. Since habituation, hunts of small- to mid-sized vertebrate prey have been observed, as well as a single case of carcass theft from a crowned eagle40. These behaviors suggest that, while meat is not a regular dietary staple at Issa, it is selectively and opportunistically incorporated into their diet. While these observations provide useful information about the Issa chimpanzee diet, isotopic data can complement and quantify dietary components in a multiyear record, even in retrospect.
Here we integrate multi-isotope data from enamel and ecological observations, demonstrating the utility of δ15Nenamel in combination with δ13Cenamel and δ18Oenamel for reconstructing and quantifying dietary behavior in extant primates. Our study aims to not only demonstrate that stable isotope analysis of primate tooth enamel offers a reliable proxy for reconstructing feeding behavior and trophic ecology in modern primates, but also quantify different dietary components, which are essential for the nitrogen budget. Our findings offer an important comparative dataset which can aid in interpretation of isotopic niches derived from the fossil tooth enamel of hominids and hominins.
Results
Inorganic δ13Cenamel and δ18Oenamel values of the carbonate (CO3) phase of enamel, and δ15Nenamel values of enamel-bound organic matter were determined for fauna with diverse dietary niches (i.e. grazers, browsers, omnivores, radicivores, primates) alongside sympatric yellow baboons and eastern chimpanzees from the Issa Valley, Tanzania (Table 1). We present δ13Cenamel, δ15Nenamel, and δ18Oenamel data for all analyzed specimens (n = 45), followed by biplots of δ13Cenamel/δ15Nenamel, δ18Oenamel/δ15Nenamel, and δ13Cenamel/δ18Oenamel.
Nitrogen isotope values
The δ15Nenamel values of all specimens ranged from 3.0 to 10.6‰ (Fig. 3a), with an overall median (\(\tilde{\text{x}}\)) of 4.8 ± 1.6‰ (H(3) = 21.25, p < 0.001). Median values were 4.6 ± 1.6‰ for primates, 5.9 ± 1.0‰ for browsers, 6.1 ± 2.0‰ for omnivores, 3.9 ± 0.9‰ for grazers, and 3.0‰ for the single radicivore (Fig. 2a). Primates had statistically significant lower δ15Nenamel values compared to omnivores (p = 0.001) and browsers (p = 0.005), but similar values to grazers (p = 0.258). The δ15Nenamel values among omnivores and browsers were similar (p = 0.359), whereas grazers showed lower ones (p ≤ 0.001).
Isotopic values of the Issa Valley fauna: (a) δ15Nenamel, (b) δ13Cenamel, (c) δ18Oenamel. Boxplots on the right show values grouped by ecological category, while those on the left display only the primate data, separated by taxon for better visualization. Boxplots show interquartile ranges with medians indicated as solid lines. Among primates, chimpanzees exhibit the lowest δ15Nenamel values, overlapping with grazing herbivores. Baboons have the highest median δ13Cenamel, consistent with regular C4 resource utilization in open woodlands. The largest variation in δ18Oenamel values occurs among forest-dwelling browsers. Note there is a break in the in δ13Cenamel axis between − 12 and − 1‰.
Biplots of (a) δ15Nenamel vs. δ13Cenamel, (b) δ15Nenamel vs. δ18Oenamel, and (c) δ18Oenamel vs. δ13Cenamel of Issa Valley fauna. Ellipses indicate 40% SEAC, convex hulls encompass full variation for each group (lines). Due to limited sample size (n < 5) for most primate taxa, SEAC was calculated only for baboons. Chimpanzees’ isotopic niche is presented as a convex hull only. Individual data points for red-tailed and red colobus monkeys are displayed but excluded from statistical analyses. Note that grazers are not shown in panels (a) and (c) due to distinct δ13Cenamel values; but extended plots including this group are provided in Supplementary Figure S2.
Primate data were further subdivided by genus to visualize potential isotopic similarities or differences between baboons and chimpanzees (see Fig. 3): δ15Nenamel values of chimpanzees (\(\tilde{\text{x}}\) = 3.7 ± 0.4‰) were significantly lower compared to baboons (\(\tilde{\text{x}}\) = 4.6 ± 0.4‰, p = 0.002), as well as the single analyzed red colobus (9.9‰) and red-tailed monkey (7.5‰).
Carbon isotope values
The δ13Cenamel values spanned the full isotopic range of C3 and C4 vegetation as expected for herbivores in African ecosystems, from −16.9 to 1.1‰ (H(3) = 17.69, p < 0.001). Fauna exhibits median values of −14.8 ± 0.4‰ for primates, −13.9 ± 0.9‰ for omnivores, −14.5 ± 0.9‰ for browsers, 0.1 ± 1‰ for grazers, and −15.1‰ for the porcupine (Fig. 2b). Primates had similar values to omnivores (p = 0.164) and browsers (p = 0.886) but had significantly lower δ13Cenamel values than grazers (p = < 0.001). Within the primate group, chimpanzees (\(\tilde{\text{x}}\) = −14.9 ± 0.1‰) and baboons (\(\tilde{\text{x}}\) = −14.3 ± 0.4‰) showed similar median values in δ13Cenamel, but baboons were more variable. The δ13Cenamel value of the red-tailed monkey fell within the range observed in chimpanzees whereas the red colobus monkey showed a slightly higher value, similar to those of baboons. Grazers were significantly higher in δ13Cenamel compared to any other group (p < 0.001); no significant difference was found between omnivores and browsers (p = 0.135).
Oxygen isotope values
The δ18Oenamel values of all specimens ranged from −4.9 to 5.3‰ (H(3) = 19.45, p < 0.001) with median values of −1.0 ± 0.9‰ for primates, −2.9 ± 1.4‰ for omnivores, −0.2 ± 1.8‰ for browsers, 2.6 ± 1.6‰ for grazers, and 0.1‰ for the porcupine (Fig. 2c). Primates had significantly lower δ18Oenamel values compared to grazers (p = 0.006) and higher ones than omnivores (p = 0.025) but overlapped with browsers (p = 0.237). Within the primates, chimpanzees had the highest δ18Oenamel values (\(\tilde{\text{x}}\) = −0.1 ± 0.5‰) followed by baboons (\(\tilde{\text{x}}\) = −1.3 ± 0.8‰), the red-tailed monkey (1.3‰), and the red colobus monkey (1.9‰). Browsers had significantly higher δ18Oenamel values compared to omnivores (p = 0.001), and were higher compared to both browsers (p = 0.049) and omnivores (p < 0.001).
Isotopic pairs and ecological niches
Isotopic niche overlap (%) and niche area (‰2) were calculated using paired isotope values to compare the different groups. A 40% standard ellipse overlap corrected for small sample size (SEAC) for δ13Cenamel/δ15Nenamel, δ18Oenamel/δ15Nenamel, and δ13Cenamel/δ18Oenamel was used in this study (Fig. 3; for details, see section Statistical Analysis; for data, including 95% SEAC calculations, see Table S2). For primates, SEAC could only be calculated for baboons due to small sample size (< 5) in the other taxa43. For chimpanzees, convex hulls are shown to visualize their ecological niche, and individual data points for red-tailed and red colobus monkeys are shown to highlight the ecological position for each primate species within the Issa food web.
δ13Cenamel/δ15Nenamel isotope niche (Fig. 3a): Baboons overlap with all other groups except the grazers and showed the largest overlap with omnivores (0.3‰2), accounting for 51.7% of the baboons’ SEAC, and 5.5% of the omnivores’ SEAC. This is followed by a < 0.1‰2 overlap with browsers, representing 1.3% of the primates’ SEAC and 0.3% of the browsers’ SEAC. Omnivores, in turn, overlapped with browsers by 1.6‰2, corresponding to 26.9 and 64.3% of their respective SEAC. Grazers, however, showed no overlap with any other group and occupied a distinct δ13Cenamel/δ15Nenamel isotopic niche.
δ18Oenamel/δ15Nenamel isotope niche (Fig. 3b): Baboons overlapped with no other groups. Omnivores, in turn, overlapped with browsers by 0.4‰2, corresponding to 6.7% and 4.1% of their respective SEAC. Grazers did not overlap with omnivores and baboons but showed an overlap of 0.2‰2 with browsers in δ18Oenamel/δ15Nenamel isotopic niche, accounting for 3.0 and 3.6% of their respective niche space.
δ13Cenamel/δ18Oenamel isotope niche (Fig. 3c): Baboons overlapped with all other groups except the grazers and overlap the most with browsers (1.1‰2), accounting for 69.4% of the primates’ SEAC and 15.5% of the browsers’ SEAC. This is followed by a 0.3‰2 overlap with omnivores, representing 45.3% of the baboons’ SEAC and 14.0% of the omnivores’ SEAC. Omnivores, in turn, overlap with browsers by 0.4‰2. corresponding to 9.1% and 6.6% of their respective SEAC. Grazers, however, show no overlap with any other group and occupy a distinct δ13Cenamel/δ18Oenamel isotopic niche.
Discussion
Chimpanzees had the lowest median δ15Nenamel value (3.7‰) of all analyzed primate species, which is significantly lower than those of sympatric baboons and comparable only to local herbivores grazing in the miombo woodland and grasslands. These low δ15Nenamel values agree with low hair δ15N data from Issa chimpanzees, which averaged 4.1 ± 0.4‰35, and thus fall within the 1σ range of the enamel values. Issa baboons also feed on woodland trees and spend a greater proportion of their time feeding on plants in miombo biomes compared to chimpanzees44,45,46. Given that the overall vegetation at Issa has an average δ15N value of 2.8‰ (n = 31; Fig. S4a)35, even a conservative enrichment factor between diet and tooth enamel of + 2.3‰ (from Leichliter et al.13) would yield expected chimpanzee δ15N values of approximately 5‰. The substantially lower δ15N value observed in Issa chimpanzees therefore cannot be explained by the plant component of their diet alone, indicating the inclusion of an additional dietary source with substantially lower δ15N than the local plants.
Analyses of whole bodies of M. subhyalinus soldiers and workers from 12 mounds at Issa revealed a low δ15N value (\(\tilde{\text{x}}\) = 0.3 ± 0.9‰; n = 47)37,47, which is on average 2.0‰ lower than plants commonly eaten by chimpanzees (Fig. 3a). By combining quantitative observational data with nitrogen content (%N) from these studies, we can estimate the contribution of termites to the chimpanzee protein budget. Termites’ high crude protein relative to plant-based resources indicated that their inclusion in the diet can account for a substantial proportion of dietary nitrogen, possibly explaining the low δ15Nenamel value observed in Issa chimpanzees (Table 2). Observational data document that chimpanzees spend only around 4% of their feeding time fishing termites48. However, despite the proportionally small amount of time devoted to termite fishing, the high N content and digestibility of the termites indicate that they make up ∼41% of the Issa chimpanzee’s protein intake (Table 2).
Our isotopic data can provide additional quantitative constrains on the relative importance of the different nitrogen sources for chimpanzees. For example, using a simple linear mixing model: δ15Nenamel = ftermites * δ15Ntermites + (1 - ftermites) * δ15Nplants + ε, where δ15Ntermites = 0.3‰, δ15Nplants = 2.3‰, and the trophic enrichment factor ε = 2.3‰, we can estimate the contribution of termites to dietary nitrogen. To reach the observed δ15Nenamel value of 3.7‰ in chimpanzees, termites would need to contribute approximately 50% of the chimpanzees’ nitrogen intake. For larger enrichment factors, as have been documented for enamel in experimental animals13, the contribution of termites to dietary nitrogen would need to be even higher. For example, considering a +3.3‰ enrichment, termites would need to account for nearly 95% of dietary nitrogen. If vertebrate meat is also consumed, which would result in enrichment in 15N, the contribution of termites to the overall nitrogen budget would need to be extremely high to account for the low δ15Nenamel values observed in Issa chimpanzees.
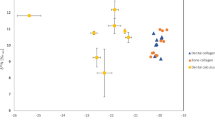
These calculations demonstrate that the inclusion of δ¹⁵N-depleted, nitrogen-rich termites is essential to explain the low δ¹⁵Nenamel values observed in Issa chimpanzees. The high contribution of termites to their protein budget may reflect the greater bioavailability of protein relative to plants, and/or small uncertainties in the direct observational estimates of the proportion of termites in the diet. Based on our conservative enrichment factor of 2.3‰ and the median published δ15N values of termites at Issa (Fig. 3a)47, we estimate that an exclusive termite-feeder would have an δ15Nenamel value of ≈ 2.6 ± 0.9‰. For comparison, we included δ15Nenamel data from two aardwolf specimens (Proteles cristatus), an obligate termite-feeder, which are with values of 2.5 and 3.2‰ consistent with this estimated isotope value (Fig. S1, Table S5). As the specimens originated from East London (South Africa) and thus a different ecosystem, this comparison should be interpreted with caution.
Interestingly, when comparing the combined isotopic niche (δ13Cenamel/δ15Nenamel, δ15Nenamel/δ18Oenamel, δ13Cenamel/δ18Oenamel), chimpanzees are clearly separated from the other primates and only overlap with browsing herbivores in the δ13Cenamel/δ18Oenamel biplot.
Chimpanzees are close to the crested porcupine in the three isotopic dimensions. However, porcupines are obligative radicivores that dig for roots and underground storage organs, a behavior usually not observed in chimpanzees. It is important to note that a study published in 200749, more than a decade prior to the habituation of the Issa community studied here, reported indirect signs of chimpanzees in the same area digging for underground storage organs. However, since habituation, this behavior has neither been directly observed nor has it appeared in tens of thousands of hours of footage from over 70 camera traps across the study area. The previously reported digging holes were therefore most likely attributable to other animals, to human activity in the area, or potentially a unique behavior of a neighboring chimpanzee community. This observation strongly supports our argument for a substantial contribution of termites to the N intake of chimpanzees.
Isotope data thus provide meaningful insights into the nutritional importance of food items that constitute a small fraction of the foraging budget, such as protein-rich insects, but whose intake is difficult to quantify through direct observation. Given that termite exploitation in chimpanzees across Africa is associated with tool-use techniques50, δ15Nenamel analysis may also shed light on early hominin tool use. As for chimpanzees, termites may have supplied early hominins with a protein rich resource comparable to meat, but one that is more easily acquired with lower risk51.
While termites are the primary invertebrates consumed by Issa chimpanzees, baboons consume a wider variety of invertebrates, which together account for approximately 10% of their observed feeding time36 (Table 2). Although isotope data for these invertebrates are not yet available from Issa, data from Serengeti National Park, ca. 700 km NE of Issa in northern Tanzania, showed δ15N values ranging from 3.2 to 12.9‰53, thus generally much higher than those of Macrotermes. These invertebrate values typically fall within or even above the δ15N range of the overall vegetation at Issa. This pattern is also reflected in the high δ¹⁵Nenamel values of the mongoose (Bdeogale crassicauda), red-tailed monkey (Cercopithecus ascanius), and civet (Nandinia binotata), all of which consume a high proportion of insects and other invertebrates, while termites appear to form only a minor component of their diet40,42,48,54.
Additionally, baboons consumed different foods, including large amounts of grasses and roots, which are rare or absent in the chimpanzees’ diet. The δ15N value of grass (3.4‰) at Issa falls close to the mean value of the general vegetation (3.0‰). δ15N data for roots, seedpods, and mushrooms are not yet available. Consumption of vertebrates and invertebrates (excluding termites) generally results in elevated δ15Nenamel values, thus the relatively low δ15Nenamel value observed in Papio remain unexpected36,55. Seeds and seedpods also contribute substantially (26%) to the diet, with baboons primarily feeding on miombo woodland (Brachystegia and Julbernardia) species, showing low δ15N values in leaves and fruits compared to the overall vegetation35,36. Roots, another important dietary component for baboons, contribute about 18% to the diet at Issa, based on observed feeding time36. Although δ15N values of these plant components at Issa are not yet available, data from Kruger National Park (South Africa) show that roots generally have lower δ15N values compared to other plant organs across both C3 and C4 plants22. Supporting this, the δ15Nenamel value of the crested porcupine which feeds primarily on these roots and underground storage organs55 are low. Together, these observations suggest that the consumption of subterranean plant tissues can contribute to the low δ15Nenamel values observed in baboons. Future analyses of stable isotopes in roots and other plant components, in combination with behavioral data, could more directly identify the specific dietary resources responsible for these depleted δ¹⁵N signatures. However, while both chimpanzees and baboons are feeding on substantial amounts of low δ15N items they are separated by about 0.9‰ in our dataset. δ15N analysis of mushrooms at Issa may help explain observed differences in δ15N values between baboons and chimpanzees as they are consumed by both primate taxa but in different quantities52,56. Mushrooms, the fruiting bodies of fungi, are high in crude protein and are increasingly recommended as an alternative protein source to meat in human populations globally57. Mushrooms have highly variable isotopic compositions, with δ15N values ranging globally from − 7.1 to 21.8‰ depending on species52. Mushrooms, are a common food source for primates at Issa and contribute up ca. 10% to the baboons diet year-round, with a peak intake of up to 30% during the wet season36. In contrast, mushrooms make up only ca. 2% of diet of chimpanzees and red-tailed monkeys. Moreover, baboons consume a greater variety of fungal species compared to chimpanzees36, including a variety of large edible ectomycorrhizal fungi, such as different species of Cantharellus, Lactifluus and Russula36. Thus, mushroom consumption may substantially contribute to the mineral-bound nitrogen in the enamel of baboons. Isotopic analyses of a variety of fungal fruiting bodies collected at Issa Valley are currently underway and will help clarify whether the consumption of these resources contributes to the higher δ15Nenamel values observed in baboons compared to chimpanzees at this site, or if the observed values are just a result of substantially lower termite consumption. Mushrooms may have played an important role in primate diets, both as a nutritional resource and as a source of (self-) medication52,58. Integrating mycophagy into the dietary ecology of extant primates will help illuminate the role of this third, and often understudied, food source.
Given our small sample size for red-tailed (n = 1) and red colobus (n = 1) monkeys, we cannot make broad generalizations about their dietary behavior. Nevertheless, we can propose factors that may account for the observed variation in isotope ratios. Both individuals exhibited higher δ15Nenamel values than the larger primates in this study (Fig. 2a). While both chimpanzees and red-tailed monkeys feed on forest fruits and leaves in similar amounts59, red-tailed monkeys at Issa have been observed to hunt and consume birds60, as well as a variable amount of invertebrates, which could contribute to the higher observed δ15Nenamel values in this taxon. Red colobus monkeys occupy the high canopy and are mainly folivorous, supplementing their diet with seeds, fruit, bark, and invertebrates61. While the smaller primates at Issa differ from chimpanzees and baboons in δ15Nenamel, we do not see similar differences in δ13Cenamel or δ18Oenamel, possibly pointing to a similar use of plant and water resources.
Regarding intra-fauna comparison from the Issa Valley, we observed isotope niche differentiation between browsers, grazers, and omnivores. Primates exhibited no overlap with grazers in any of the isotope biplots (Fig. 3), suggesting that they primarily foraged in the C3 dominated woodland habitats, thereby avoiding direct competition with grazers.
Conclusion
This study focused on the mosaic savanna-woodland landscape in the Issa valley, an environment broadly similar to those in which early hominins may have evolved. We used chimpanzees and baboons as modern analogues to investigate how isotope data (δ13Cenamel, δ15Nenamel, δ18Oenamel) reflect the ecological position of these primates. We integrated field observations and isotopic data, providing important behavioral context not available for fossil hominids. We found that Issa’s primates prefer C3 resources and occupy an isotopic niche that is distinct from that of grazers, overlapping primarily with browsers and, to a lesser extent, with omnivores.
Our isotope data further reveal a distinct isotopic niche for chimpanzees in both δ¹³Cenamel/δ¹⁵Nenamel and δ¹⁵Nenamel/δ¹⁸Oenamel space, indicating a dietary composition that differs markedly from that of the other primates or any other faunal groups. Chimpanzees consume dietary resources relatively low in δ15N, and observational data are essential to identify the specific dietary components. In all three isotope systems, chimpanzees cluster closely to the porcupine – a specialized radicivore – but the feeding behavior of the two taxa differs, as wild chimpanzees at Issa (or elsewhere) are not known to routinely dig for and eat roots or underground storage organs. Instead, their low δ¹⁵Nenamel values likely reflect a substantial contribution of termites to their nitrogen intake. Our estimates suggest that termites provided at least 50% of the Issa chimpanzees’ nitrogen requirements, making termite consumption the primary factor shaping their unique isotopic signature.
In contrary to the chimpanzees, the consumption of roots, seeds and seedpods could play an important role in driving baboons’ relatively low δ¹⁵Nenamel values, all items that are regularly consumed by baboons. However, specific seeds or seedpod data for miombo species are not available at Issa and we were limited to fruit and leave data, which show overall low δ15N values for our calculations. Also roots have not yet been analyzed for δ15N at Issa, but evidence from the δ15Nenamel values of the analyzed porcupine, as well as δ15N data of roots from South Africa22 suggest them as low in δ15N. In addition, the frequent consumption of mushrooms36 may contribute to the relatively higher δ15Nenamel values in baboons compared to chimpanzees. All dietary components would benefit of further isotope investigation to enable a more coherent breakdown of the contribution of individual dietary items to the nitrogen budget.
Additional research could include serial sampling of the same tooth, to evaluate seasonal variability in diet, particularity with respect to the consumption of nutrient-rich food resources termites in chimpanzees and mushrooms in baboons. Our findings from bulk enamel sampling provide a robust foundation for such work. Additionally, non-traditional isotope systems (e.g., calcium, strontium, zinc) are increasingly used to complement traditional stable isotopes analysis in reconstructing dietary variability and resource use. A forthcoming complementary study using the same specimens and enamel powders will examine these non-traditional isotopes to further investigate inter- and intra-species dietary variation at Issa.
Finally, our study analyzed only late-forming molars, which reflect diet after weaning. A comparison of early- and late- mineralizing teeth, using both traditional and non-traditional isotopic approaches, could provide valuable insight into nursing, weaning, and early-life dietary transitions in primates and early hominins.
We conclude that from our isotope analyses that termites, seeds, seedpods and roots are dietary items, that can contribute to the low δ15Nenamel values observed in primates. For baboons, observational data suggest that seeds and seedpods of miombo legumes and root consumption is the one of the primary factors shaping their isotopic niche, and they appear higher compared to our chimpanzees because of the frequent consumption of mushrooms. In contrast, the distinct isotopic niche of chimpanzees – well separated from primates – probably reflect the consumption of termites, an energy-rich dietary component that makes a substantial contribution to the overall protein budget. The inclusion of invertebrates may have supplied essential protein comparable to those from mammalian meat, potentially meeting the elevated energy demands associated with encephalization and contributing to the evolutionary success of the hominin lineage.
Materials and methods
The Issa Valley
The Issa Valley – located ca. 90 km east of Lake Tanganyika in western Tanzania – offers an ideal setting for environmental and dietary reconstructions due to its remote location (the nearest town, Uvinza, is about 70 km to the NW) and modest anthropogenic influence. For isotopic studies, such conditions are advantageous, as artificial fertilizers result in an δ15N-enriched groundwater and soil, potentially confounding natural signals62.
Situated at 1550 m above sea level and part of the Greater Mahale Ecosystem, the area encompasses a mosaic vegetation, including miombo woodland (characterized by the genera Brachystegia and Julbernardia), seasonally inundated grasslands, as well as riverine and thicket forests31. The relative abundance of plants in the surrounding areas are described by Moore63 and plant isotope data suggest that the savanna grasses at Issa include C4 species, while the riparian vegetation is dominated by C3 plants35 (Fig. S4). The region experiences two distinct seasons: a wet season from November to April, and a dry season from May to October, with dry months defined as those with less than 100 mm rainfall. Environmental data collected between 2009 and 2015 showed that the Issa Valley receives annual rainfall ranging from 930 to 1650 mm, and daily temperatures of the region range from 11 to 38°C54,55.
Issa is home to eight different primate species64, some of which (i.e. eastern chimpanzees, yellow baboons, and red-tailed monkeys (Cercopithecus ascanius)) have been the focus of continuous research in the Issa Valley since 2008, with group and focal follows of habituated individuals of baboons and red-tailed monkeys since 201254, and chimpanzees since 201848. Other diurnal primate species at Issa include blue monkeys (Cercopithecus mitis), ashy red colobus monkeys (Piliocolobus tephrosceles), and vervet monkeys (Cholorocebus pygerythrus)64. This diversity in primate species in a modern ecosystem with modest human influence resembles environmental conditions that some early hominins might have experienced in eastern Africa during the Plio-Pleistocene65,66.
Materials
Tooth enamel from skeletal remains, primary isolated skulls, of 45 individuals representing 18 mammalian species, was sampled for isotopic analysis in July 2022 (Table 1). These specimens were opportunistically collected from naturally deceased individuals encountered between 2012 and 2021.
Given the focus on primate dietary reconstruction, we consider primates separate from the group of omnivores and visualize the different primate species individually in our data. The dataset includes a total of 15 non-human primates: four chimpanzees, nine baboons, one ashy red colobus monkey, and a red-tailed monkey. Sympatric fauna included for comparative purposes comprised eight omnivores (i.e. suids, a civet and mongooses), one radicivore (porcupine), as well as 15 browsers and six grazing bovids (Table 1).
We sampled from each specimen a small chip of tooth enamel. All samples were mechanically cleaned of surface contaminants, rinsed in ultrapure acetone, and air dried in a covered test tube before shipment to the Max Planck Institute for Chemistry (MPIC, Mainz, Germany). Here, if necessary, remaining dentin was carefully removed with a handheld Dremel multitool using a diamond drill tip at low speed (max. 2500 rpm). The enamel flakes were then crushed with an agate mortar and pestle, each sample yielding 15 to 77 mg of enamel powder. A small aliquot (< 0.2 mg) of this homogenized powder was used for δ13Cenamel and δ18Oenamel analysis, and another (< 10 mg) for δ15Nenamel analysis. All analyses were performed at the MPIC. Additionally, a split of ca. 10 mg was sent to collaborators at Ecole normale superieure de Lyon (Lyon, France) for complimentary calcium, magnesium, stable strontium and zinc isotope analyses of the same specimens.
Methods
δ15Nenamel analysis using the oxidation denitrification method
Mineral-bound δ15Nenamel was measured using the oxidation-denitrification method at the Organic Isotope Geochemistry Laboratory, MPIC, Germany following the protocols of Leichliter13. The method was originally introduced for marine archive analysis67,68and is routinely applied to measure δ15N in modern and fossil materials including corals, foraminifera, diatoms, and teeth. The method was first applied for analyzing δ15Nenamel by Leichliter13 using rodent teeth from a controlled feeding experiment, and then subsequently used to validate diet and trophic behavior reconstructions of wild mammals in natural African ecosystems17,18. Further studies of the δ15Nenamel values of fossil teeth showed that enamel is highly resistant to alteration under experimentally altered conditions in a laboratory study69, reconstructs diet in Pleistocene southeast Asian fossil assemblages15 and Pliocene southern African Australopithecus sp.14.
The powdered enamel samples were prepared for analysis by first removing potential metal oxides through a reductive cleaning with a dithionite solution in centrifuge tubes. Samples were transferred into glass vials and exogenous, non-mineral-bound organic matter was then removed using a potassium persulfate oxidation reagent (POR)70. The apatite matrix was subsequently demineralized with hydrochloric acid to free the mineral-bound organic N, followed by a second oxidation step using POR to convert liberated N to nitrate. The resulting nitrate was then converted to nitrous oxide (N2O) by the denitrifying bacteria Pseudomonas chlororaphis, which are grown, cultured, and harvested at MPIC following the protocols of Sigman et al. and Weigand et al.68,71.
δ15Nenamel measurements of the bacterially produced N2O were carried out using a custom-built multi-valve system designed to aggregate and separate N2O from residual CO2 in the gas flow using a series of water and CO2 traps coupled with a Thermo Fisher Scientific MAT 253-Plus gas-chromatography mass spectrometry after Sigman et al. and Weigand et al.68,71. Measurements were performed at a target load of 5 nmol for optimal precision, typically using 4.4 to 8.5 mg of untreated enamel. During chemical pretreatment, 10 to 55% of initial material mass was lost due to reagent reactions and handling, resulting in 2.4 to 6.9 mg pre-treated enamel powder. To validate measurement precision, out of the 45 specimens of our dataset, we measured 24 enamel samples in duplicates resulting in 69 individuals across three analytical batches.
To monitor instrumental and methodological precision, we included four in-house (see Table S1 for details) and four international standards. The in-house standards include two powdered enamel samples of Proboscideans (the modern elephant “AG-Lox” and fossil mammoth “Mammy”, respectively), and two modern coral powders (“CF-1” and “PO2”), which were each measured in triplicate in each analytical batch, resulting in a precision of 0.2‰ across all batches. Additionally, triplicates of the international standards USGS-65 (glycine) and USGS-40 (L-glutamic acid) were included during the oxidation step, as well as IAEA-NO3 (potassium nitrate) and USGS-34 (nitrate) during the bacteria injection step (at least 3 replicas in different concentrations). Inter-batch precision (± 1σ) for δ15N for USGS-40 (n = 9) was < 0.2‰ and for USGS-65 (n = 9) 0.1‰, respectively. Furthermore, we introduce 15 blank vials per batch during the demineralization to record the fraction of the blank and its δ15N value (< 0.3 nmol/ml; range − 39 to 3‰). The average blank contribution was ca. 1%. Moreover, two bacteria blank vials are analyzed in each batch. For details see Leichliter et al.13.
δ13Cenamel and δ18Oenamel analyses using the cold-trap method
For δ13Cenamel and δ18Oenamel analyses, a small aliquot (70–120 µg) of untreated enamel powder was analyzed in the inorganic gas isotope geochemistry lab at the MPIC using the “cold trap method”72. A Thermo Scientific Gasbench II continuous flow interface unit with a pneumatically activated liquid N2 trap coupled with a Thermo Scientific Delta-V mass spectrometer was used. Weighted sample powder was automatically flushed with 5.0 grade helium (He) for 90 minutes before being acidified with > 99% H3PO4 at 70 °C in 12 ml Labco exetainer vials for another 90 minutes. The He carrier gas flushes the CO2 sample gas into the liquid N2 trap where it is cryogenically focused for 6 to 7 minutes. The accumulated sample gas is transported into the spectrometer resulting in one single sample peak. Two international standards (IAEA-603 and NBS-120c) and three in-house standards (Carrara marble “VICS” and the fossil tooth enamel standard “Mammy” and “Ag-Lox”, the same material used for δ15Nenamel measurements) are used to calculate analytical precision72. For internal precisions refer to table S4.
Statistical analysis
Statistical analysis was conducted in R (version 4.3.1. R Core Team. 2024). Non-parametric statistics were used, as normality could not be assumed due to the small sample size of many groups. Statistical significance between isotopic groups was calculated using a Kruskal-Wallis Test followed by post-hoc Dunn`s test with a significance level of p = 0.05, Spearman’s rank correlation coefficient (rs) is given for correlations. Outliers were identified using Tukey’s Fence (k = 1.5), which were then excluded for statistical significance test and typically median values (\(\tilde{\text{x}}\)) are stated for each group. p-values and H-statistics are reported in the main text while z-scores are reported in Table S3.
Furthermore, multivariate isotope comparisons were performed using the Stable Isotopes Bayesian Ellipses (SIBER) package43 and the statistical code after Turner73. For all groups (baboons, omnivores, browsers, grazers) isotopic niche analysis was performed for δ15Nenamel, δ13Cenamel, δ18Oenamel with estimated standard ellipses areas corrected for sample size (SEAC). SEAC values for 40% ellipses were calculated and the proportional niche overlap (%) is reported in the text. Additionally, calculations for 40% and 95% ellipses including proportional and total overlap (‰2) were calculated and are reported in table S2. Additionally centroid distance between groups was calculated using Layman Metrics74 and a Hotelling T2 test was performed.
Data availability
All data are available in the main text or the supplementary material.
References
Bocherens, H. in in The Evolution of Hominin Diets: Integrating Approaches to the Study of Palaeolithic Subsistence. 241–250 (eds Hublin, J. J. & Richards, M. P.) (Springer Netherlands, 2009).
DeNiro, M. J. & Epstein, S. Influence of diet on the distribution of nitrogen isotopes in animals. Geochim. Cosmochim. Acta. 45, 341–351. https://doi.org/10.1016/0016-7037(81)90244-1 (1981).
Lucas, P. W., Constantino, P. J. & Wood, B. A. Inferences regarding the diet of extinct hominins: structural and functional trends in dental and mandibular morphology within the hominin clade. J. Anat. 212, 486–500. https://doi.org/10.1111/j.1469-7580.2008.00877.x (2008).
Wilson, M. L. Insights into human evolution from 60 years of research on chimpanzees at Gombe. Evol. Hum. Sci. https://doi.org/10.1017/ehs.2021.2 (2021).
McGrew, W. C. Chimpanzee technology. Science 328, 579–580 (2010).
Haslam, M. On the tool use behavior of the bonobo-chimpanzee last common ancestor, and the origins of hominine stone tool use. Am. J. Primatol. 76, 910–918. https://doi.org/10.1002/ajp.22284 (2014).
Boesch, C. & Boesch, H. Tool use and tool making in wild chimpanzees. Folia Primatol (Basel) 54, 86–99. https://doi.org/10.1159/000156428 (1990).
Lawick-Goodall, J. V., Lawick, H. V. & Packer, C. Tool-use in free-living baboons in the Gombe National Park, Tanzania. Nature 241, 212–213. https://doi.org/10.1038/241212a0 (1973).
Ambrose, S. H. Stable carbon and nitrogen isotope analysis of human and animal diet in Africa. J. Hum. Evol. 15, 707–731. https://doi.org/10.1016/S0047-2484(86)80006-9 (1986).
Lee-Thorp, J. A., Sealy, J. C. & van der Merwe, N. J. Stable carbon isotope ratio differences between bone collagen and bone apatite, and their relationship to diet. J. Archaeol. Sci. 16, 585–599. https://doi.org/10.1016/0305-4403(89)90024-1 (1989).
Robinson, C., Kirkham, J., Brookes, S. J., Bonass, W. A. & Shore, R. C. The chemistry of enamel development. Int. J. Dev. Biol. 39, 145–152. https://doi.org/10.1387/IJDB.7626401 (1995).
Savory, A. & Brudevold, F. The distribution of nitrogen in human enamel. J. Dent. Res. 38, 436–442. https://doi.org/10.1177/00220345590380030301 (1959).
Leichliter, J. N. et al. Nitrogen isotopes in tooth enamel record diet and trophic level enrichment: Results from a controlled feeding experiment. Chem. Geol. 563, 120047. https://doi.org/10.1016/j.chemgeo.2020.120047 (2021).
Lüdecke, T. et al. Australopithecus at Sterkfontein did not consume substantial mammalian meat. Science 387, 309–314. https://doi.org/10.1126/science.adq7315 (2025).
Bourgon, N. et al. Faunal persistence and ecological flexibility in Pleistocene Southeast Asia revealed through multi-isotope analysis. Sci. Adv. 11, eadu3642. https://doi.org/10.1126/sciadv.adu3642 (2025).
Vink, M. et al. Eemian climate seasonality reconstructed from intra-tooth oxygen, carbon, and nitrogen stable isotopes in equid enamel from Neumark-Nord 2, Germany. Quatern. Sci. Rev. 373, 109724. https://doi.org/10.1016/j.quascirev.2025.109724 (2026).
Leichliter, J. N. et al. Tooth enamel nitrogen isotope composition records trophic position: A tool for reconstructing food webs. Commun. Biol. 6, 373. https://doi.org/10.1038/s42003-023-04744-y (2023).
Lüdecke, T. et al. Carbon, nitrogen, and oxygen stable isotopes in modern tooth enamel: A case study from Gorongosa National Park, central Mozambique. Front. Ecol. Evol. (2022). https://doi.org/10.3389/fevo.2022.958032
Bocherens, H. & Drucker, D. Trophic level isotopic enrichment of carbon and nitrogen in bone collagen: Case studies from recent and ancient terrestrial ecosystems. Int. J. Osteoarchaeol. 13, 46–53. https://doi.org/10.1002/oa.662 (2003).
Schoeninger, M. J. & DeNiro, M. J. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochim. Cosmochim. Acta 48, 625–639. https://doi.org/10.1016/0016-7037(84)90091-7 (1984).
Kohn, M. J. Carbon isotope compositions of terrestrial C3 plants as indicators of (paleo)ecology and (paleo)climate. Proc. Natl. Acad. Sci. U. S. A. 107, 19691. https://doi.org/10.1073/pnas.1004933107 (2010).
Codron, J. et al. Taxonomic, anatomical, and spatio-temporal variations in the stable carbon and nitrogen isotopic compositions of plants from an African savanna. J. Archaeol. Sci. 32, 1757–1772. https://doi.org/10.1016/j.jas.2005.06.006 (2005).
Cerling, T. E. et al. Global vegetation change through the Miocene/Pliocene boundary. Nature 389, 153–158. https://doi.org/10.1038/38229 (1997).
Passey, B. H. et al. Carbon isotope fractionation between diet, breath CO2, and bioapatite in different mammals. J. Archaeol. Sci. 32, 1459–1470. https://doi.org/10.1016/j.jas.2005.03.015 (2005).
Malone, M. A., MacLatchy, L. M., Mitani, J. C., Kityo, R. & Kingston, J. D. A chimpanzee enamel-diet δ(13)C enrichment factor and a refined enamel sampling strategy: Implications for dietary reconstructions. J. Hum. Evol. 159, 103062. https://doi.org/10.1016/j.jhevol.2021.103062 (2021).
Green, D. R. et al. Fine-scaled climate variation in equatorial Africa revealed by modern and fossil primate teeth. Proc. Natl. Acad. Sci. U. S. A. 119, e2123366119. https://doi.org/10.1073/pnas.2123366119 (2022).
Smith, T. M. et al. Primate teeth are good proxies for understanding past water inputs and seasonality. Geochim. Cosmochim. Acta 402, 200–216. https://doi.org/10.1016/j.gca.2025.05.033 (2025).
Kohn, M. J., Schoeninger, M. J. & Valley, J. W. Herbivore tooth oxygen isotope compositions: Effects of diet and physiology. Geochim. Cosmochim. Acta 60, 3889–3896. https://doi.org/10.1016/0016-7037(96)00248-7 (1996).
Oelze, V. M. et al. Comparative isotope ecology of African great apes. J. Hum. Evol. 101, 1–16. https://doi.org/10.1016/j.jhevol.2016.08.007 (2016).
King, G. E. Baboon perspectives on the ecology and behavior of early human ancestors. Proc. Natl. Acad. Sci. U. S. A. 119, e2116182119. https://doi.org/10.1073/pnas.2116182119 (2022).
Drummond-Clarke, R. C. et al. Wild chimpanzee behavior suggests that a savanna-mosaic habitat did not support the emergence of hominin terrestrial bipedalism. Sci. Adv. 8, eadd9752. https://doi.org/10.1126/sciadv.add9752 (2022).
Su, D. F. Early hominin paleoenvironments and habitat heterogeneity. Annu. Rev. Anthropol. 53, 21–35. https://doi.org/10.1146/annurev-anthro-041222-102712 (2024).
Lüdecke, T. et al. Dietary versatility of Early Pleistocene hominins. Proc. Natl. Acad. Sci. U. S. A. 115, 13330–13335. https://doi.org/10.1073/pnas.1809439115 (2018).
Cerling, T. E. et al. Stable isotope-based diet reconstructions of Turkana Basin hominins. Proc. Natl. Acad. Sci. USA 110, 10501–10506. https://doi.org/10.1073/pnas.1222568110 (2013).
van Casteren, A. et al. Food mechanical properties and isotopic signatures in forest versus savannah dwelling eastern chimpanzees. Commun. Biol. 1, 109. https://doi.org/10.1038/s42003-018-0115-6 (2018).
Schulze, T. A. et al. Mycophagy in primates of the Issa Valley, Tanzania. Ecol. Evol. 15, e72000. https://doi.org/10.1002/ece3.72000 (2025).
Phillips, S., Piel, A. K., Stewart, F. A. & Oelze, V. M. A chimpanzee’s time to feast: Seasonality of Macrotermes flight hole activity and alate dispersal flights detected by termite-fishing experiments and camera traps in the Issa Valley, Tanzania. Front. Ecol. Evol. https://doi.org/10.3389/fevo.2023.1289433 (2023).
Phillips, S. et al. Wild chimpanzee termite mound inspections converge with the onset of rain. Sci. Rep. 15, 13247. https://doi.org/10.1038/s41598-025-90382-9 (2025).
Stewart, F. A. & Piel, A. K. Termite fishing by wild chimpanzees: New data from Ugalla, western Tanzania. Primates 55, 35–40. https://doi.org/10.1007/s10329-013-0362-6 (2014).
Baker, S. A., Stewart, F. A. & Piel, A. K. A case of suspected chimpanzee scavenging in the Issa Valley, Tanzania. Primates 65, 41–48. https://doi.org/10.1007/s10329-023-01099-0 (2024).
Ramirez Amaya, S., McLester, E., Stewart, F. & Piel, A. Savanna chimpanzees (Pan troglodytes schweinfurthii) consume and share blue duiker (Philantomba monticola) meat in the Issa Valley, Ugalla, western Tanzania. PANA 22, 17–21. https://doi.org/10.5134/203111 (2015).
Piel, A. K. et al. The diet of open-habitat chimpanzees (Pan troglodytes schweinfurthii) in the Issa Valley, western Tanzania. J. Hum. Evol. 112, 57–69. https://doi.org/10.1016/j.jhevol.2017.08.016 (2017).
Jackson, A., Inger, R., Parnell, A. & Bearhop, S. Comparing isotopic niche width among and within communities: SIBER – Stable isotope Bayesian ellipses in R. J. Anim. Ecol. 80, 595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x (2011).
Norton, G. W., Rhine, R. J., Wynn, G. W. & Wynn, R. D. Baboon diet: A five-year study of stability and variability in the plant feeding and habitat of the yellow baboons (Papio cynocephalus) of Mikumi National Park, Tanzania. Folia Primatol (Basel) 48, 78–120. https://doi.org/10.1159/000156287 (1987).
Post, D. G. Feeding behavior of yellow baboons (Papio cynocephalusin) the Amboseli National Park, Kenya. Int. J. Primatol. 3, 403–430. https://doi.org/10.1007/BF02693741 (1982).
Drummond-Clarke, R. C., Reuben, S. C., Stewart, F. A., Piel, A. K. & Kivell, T. L. Foraging strategy and tree structure as drivers of arboreality and suspensory behaviour in savannah-dwelling chimpanzees. Front. Ecol. Evol. https://doi.org/10.3389/fevo.2025.1561078 (2025).
Phillips, S. et al. Limited evidence of C4 plant consumption in mound building Macrotermes termites from savanna woodland chimpanzee sites. PLoS One 16, e0244685. https://doi.org/10.1371/journal.pone.0244685 (2021).
Giuliano, C. Chimpanzee (Pan troglodytes schweinfurthii) behavioral responses to resource scarcity in the savanna-woodland environment of Issa valley, Tanzania — Study of feeding, ranging, and grouping patterns (Liverpool John Moores University, 2022).
Hernandez-Aguilar, R. A., Moore, J. & Pickering, T. R. Savanna chimpanzees use tools to harvest the underground storage organs of plants. Proc. Natl. Acad. Sci. U. S. A. 104, 19210–19213. https://doi.org/10.1073/pnas.0707929104 (2007).
Boesch, C. et al. Chimpanzee ethnography reveals unexpected cultural diversity. Nat. Hum. Behav. 4, 910–916. https://doi.org/10.1038/s41562-020-0890-1 (2020).
Lesnik, J. J. Termites in the hominin diet: A meta-analysis of termite genera, species and castes as a dietary supplement for South African robust australopithecines. J. Hum. Evol. 71, 94–104. https://doi.org/10.1016/j.jhevol.2013.07.015 (2014).
O’Regan, H. J., Lamb, A. L. & Wilkinson, D. M. The missing mushrooms: Searching for fungi in ancient human dietary analysis. J. Archaeol. Sci. 75, 139–143. https://doi.org/10.1016/j.jas.2016.09.009 (2016).
DeVisser, S. N., Freymann, B. P. & Schnyder, H. Trophic interactions among invertebrates in termitaria in the African savanna: A stable isotope approach. Ecol. Entomol. 33, 758–764. https://doi.org/10.1111/j.1365-2311.2008.01029.x (2008).
Piel, A. K., Bonnin, N., Ramirez Amaya, S., Wondra, E. & Stewart, F. A. Chimpanzees and their mammalian sympatriates in the Issa Valley, Tanzania. Afr. J. Ecol. 57, 31–40. https://doi.org/10.1111/aje.12570 (2019).
D’Ammando, G. et al. Ecological drivers of habitat use by meso mammals in a Miombo ecosystem in the Issa Valley, Tanzania. Front. Ecol. Evol. https://doi.org/10.3389/fevo.2022.773568 (2022).
Jaouen, K. et al. Exceptionally high δ15N values in collagen single amino acids confirm Neandertals as high-trophic level carnivores. Proc. Natl. Acad. Sci. U. S. A. 116, 4928–4933. https://doi.org/10.1073/pnas.1814087116 (2019).
Ayimbila, F. & Keawsompong, S. Nutritional quality and biological application of mushroom protein as a novel protein alternative. Curr. Nutr. Rep. 12, 290–307. https://doi.org/10.1007/s13668-023-00468-x (2023).
Weyrich, L. S. et al. Neanderthal behaviour, diet, and disease inferred from ancient DNA in dental calculus. Nature 544, 357–361. https://doi.org/10.1038/nature21674 (2017).
Tweheyo, M. & Obua, J. Feeding habits of chimpanzees (Pan troglodytes), red-tail monkeys (Cercopithecus ascanius schmidti) and blue monkeys (Cercopithecus mitis stuhlmanii) on figs in Budongo Forest Reserve, Uganda. Afr. J. Ecol. 39, 133–139. https://doi.org/10.1046/j.1365-2028.2001.00290.x (2001).
Lile, C., McLester, E., Stewart, F. & Piel, A. Red-tailed monkeys (Cercopithecus ascanius) prey upon and mob birds in the Issa Valley, Western Tanzania. Primates https://doi.org/10.1007/s10329-020-00834-1 (2020).
Kibaja, M. J. et al. On the move: Activity budget and ranging ecology of endangered Ashy red colobus monkeys (Piliocolobus tephrosceles) in a savanna woodland habitat. Glob. Ecol. Conserv. 43, e02440. https://doi.org/10.1016/j.gecco.2023.e02440 (2023).
Yoo, S. H. et al. Temporal Variations in Isotope Ratios and Concentrations of Nitrate-nitrogen in Groundwater as Affected by Chemical Fertilizer and Livestock Manure. J. Appl. Biol. Chem. 42, 186–190 (1999).
Moore, J. Plants of the Tongwe East Forest Reserve (Ugalla), Tanzania. Tropics 3, 333–340. https://doi.org/10.3759/tropics.3.333 (1994).
McLester, E., Pintea, L., Stewart, F. A. & Piel, A. K. Cercopithecine and colobine abundance across protected and unprotected land in the Greater Mahale Ecosystem, Western Tanzania. Int. J. Primatol. 40, 687–705. https://doi.org/10.1007/s10764-019-00118-6 (2019).
Wynn, J. G. Paleosols, stable carbon isotopes, and paleoenvironmental interpretation of Kanapoi, Northern Kenya. J. Hum. Evol. 39, 411–432. https://doi.org/10.1006/jhev.2000.0431 (2000).
White, T. D. et al. Asa Issie, Aramis and the origin of Australopithecus. Nature 440, 883–889. https://doi.org/10.1038/nature04629 (2006).
Ren, H. et al. Foraminiferal isotope evidence of reduced nitrogen fixation in the Ice Age Atlantic Ocean. Science 323, 244–248. https://doi.org/10.1126/science.1165787 (2009).
Sigman, D. M. et al. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 73, 4145–4153. https://doi.org/10.1021/ac010088e (2001).
Martinez-Garcia, A. et al. Laboratory assessment of the impact of chemical oxidation, mineral dissolution, and heating on the nitrogen isotopic composition of fossil-bound organic matter. ESS Open. Archive. (2022). https://doi.org/10.1002/essoar.10510728.2
Moretti, S. et al. Analytical improvements and assessment of long-term performance of the oxidation–denitrifier method. Rapid Commun. Mass Spectrom. 38, e9650. https://doi.org/10.1002/rcm.9650 (2024).
Weigand, M. A., Foriel, J., Barnett, B., Oleynik, S. & Sigman, D. M. Updates to instrumentation and protocols for isotopic analysis of nitrate by the denitrifier method. Rapid Commun. Mass Spectrom. 30, 1365–1383. https://doi.org/10.1002/rcm.7570 (2016).
Vonhof, H. et al. High‐precision stable isotope analysis of <5 μg CaCO3 samples by continuous‐flow mass spectrometry. Rapid Commun. Mass Spectrom. 34, e8878. https://doi.org/10.1002/rcm.8878 (2020).
Turner, T. F., Collyer, M. L. & Krabbenhoft, T. J. A general hypothesis-testing framework for stable isotope ratios in ecological studies. Ecology 91, 2227–2233. https://doi.org/10.1890/09-1454.1 (2010).
Layman, C. A., Arrington, D. A., Montaña, C. G. & Post, D. M. Can stable isotope ratios provide for community-wide measures of trophic structure?. Ecology 88, 42–48. https://doi.org/10.1890/0012-9658(2007)88[42:csirpf]2.0.co;2 (2007).
Acknowledgements
We express our gratitude to the Tanzania Commission for Science and Technology (COSTECH) and to the Tanzania Wildlife Research Institute (TAWIRI) for permission to conduct field work and sampling. The skeletal specimens analyzed are curated at the Greater Mahale Ecosystem Research and Conservation project base camp and samples were exported under permits TWRI/RS/307/49, CITES DE-E-05453/22, and CITES DE-E-04674/23. We thank F. Rubach, M. Schmitt, M. Vink, J. Storsberg, and B. Hinnenberg (Max Planck Institute for Chemistry, Mainz, Germany) for technical support, as well as T. Kaiser (Leibniz Institute for the Analysis of Biodiversity Change, Hamburg, Germany) for access to the two aardwolf specimens. We also thank Maciej Krajcarz and two anonymous reviewers who provided valuable critiques for this manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was funded by the Deutsche Forschungsgemeinschaft (DFG) Emmy Noether Fellowship LU 2199/2 − 2 to TL and the Max Planck Society to TL, AMG, HV, GHH. SB acknowledges funding by the Max Planck Graduate Center (MPGC). VMO was funded by the University of California at Santa Cruz. Long-term research at Issa is supported by the UCSD/Salk Center for Academic Research and Training in Anthropogeny (CARTA) and the Department of Human Origins, Max Planck Institute of Evolutionary Anthropology, Leipzig, Germany.
Author information
Authors and Affiliations
Contributions
VO, AP and TL conceptualized the project. VO selected and sampled specimens. SB performed the δ15Nenamel measurements in the lab of AMG. SB performed δ13Cenamel and δ18Oenamel measurements in the lab of HV. SB, VO, FS, AMG and TL discussed the interpretation of the results. SB and TL conducted statistical analyses. SB, VO and TL wrote the original draft, and all other contributing authors provided input to the final manuscript at different stages of the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brömme, S., Oelze, V.M., Martínez-García, A. et al. Multi-isotope analysis reconstructs termite feeding in chimpanzees. Sci Rep 16, 14026 (2026). https://doi.org/10.1038/s41598-026-45049-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-45049-4