Abstract
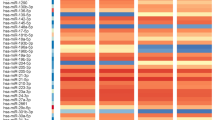
Oral mucosal abnormalities considered to have malignant potential may involve a large area of mucosa. Standard care histopathological investigation is invasive, limited to site selection and subject to variation in assessment between pathologists. A quantitative, minimally invasive, rapidly collected, oral mucosal site-specific assessment will assist in decision making and increase diagnostic precision. This study aimed to validate a workflow to analyse oral scrape derived cancer-associated microRNA analysis for mucosal site-specific assessment as a surrogate biomarker to histopathological diagnosis. Forty-one oral scrapes were collected from 33 patients undergoing investigation at the Royal Dental Hospital of Melbourne before mucosal biopsy. RNA from oral scrapes was used to investigate ten cancer-associated microRNAs. An algorithm for categorical high or low-risk based on histopathological diagnosis was developed. The novel risk stratification algorithm utilised two microRNAs and categorised all cases of carcinoma and severe dysplasia as high-risk and accurately distinguished all non-potentially malignant disorders as low-risk lesions. This study provides proof-of-concept that oral scrapes can be predictably collected in a clinical workflow to assess the expression of cancer-associated microRNA specific to an oral mucosal site with minimal invasiveness.
Similar content being viewed by others
Data availability
All processed data generated or analysed during this study are included in this published article. Raw data are available from the corresponding author on reasonable request.
References
Irani, S. Pre-Cancerous Lesions in the Oral and Maxillofacial Region: A Literature Review with Special Focus on Etiopathogenesis. Iran. J. Pathol. 11 (4), 303–322 (2016).
Warnakulasuriya, S. et al. Oral potentially malignant disorders: A consensus report from an international seminar on nomenclature and classification, convened by the WHO Collaborating Centre for Oral Cancer. Oral Dis. 27 (8), 1862–1880 (2021).
Ganesh, D. et al. Potentially Malignant Oral Disorders and Cancer Transformation. Anticancer Res. 38 (6), 3223–3229 (2018).
Iocca, O. et al. Potentially malignant disorders of the oral cavity and oral dysplasia: A systematic review and meta-analysis of malignant transformation rate by subtype. Head Neck. 42 (3), 539–555 (2020).
Farah, C., Balasubramaniam, R. & McCullough, M. J. Contemporary oral medicine (Springer, 2019).
Gupta, P. C. et al. Incidence rates of oral cancer and natural history of oral precancerous lesions in a 10-year follow-up study of Indian villagers. Commun. Dent. Oral Epidemiol. 8 (6), 283–333 (1980).
Holmstrup, P. et al. Long-term treatment outcome of oral premalignant lesions. Oral Oncol. 42 (5), 461–474 (2006).
Speight, P. M., Khurram, S. A. & Kujan, O. Oral potentially malignant disorders: risk of progression to malignancy. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 125 (6), 612–627 (2018).
Warnakulasuriya, S. et al. Factors predicting malignant transformation in oral potentially malignant disorders among patients accrued over a 10-year period in South East England. J. Oral Pathol. Med. 40 (9), 677–683 (2011).
Reboiras-López, M. D. et al. Comparison of the Cytobrush®, dermatological curette and oral CDx® brush test as methods for obtaining samples of RNA for molecular analysis of oral cytology. Cytopathology 23 (3), 192–197 (2012).
Navone, R. et al. Oral potentially malignant lesions: first-level micro-histological diagnosis from tissue fragments sampled in liquid-based diagnostic cytology. J. Oral Pathol. Med. 37 (6), 358–363 (2008).
Mehrotra, R. et al. Application of cytology and molecular biology in diagnosing premalignant or malignant oral lesions. Mol. Cancer. 5, 11 (2006).
Prasad, G. et al. The assessment of the robustness of microRNAs from oral cytological scrapings. J. Oral Pathol. Med. 46 (5), 359–364 (2017).
Chen, H. C. et al. Genetic variants in microRNA-146a (C > G) and microRNA-1269b (G > C) are associated with the decreased risk of oral premalignant lesions, oral cancer, and pharyngeal cancer. Arch. Oral Biol. 72, 21–32 (2016).
Li, M. et al. MicroRNAs: control and loss of control in human physiology and disease. World J. Surg. 33 (4), 667–684 (2009).
de Rooij, L. A. et al. The microRNA Lifecycle in Health and Cancer. Cancers (Basel), 14(23). (2022).
Hung, K. F. et al. MicroRNA-31 upregulation predicts increased risk of progression of oral potentially malignant disorder. Oral Oncol. 53, 42–47 (2016).
Yang, Y. et al. Progress risk assessment of oral premalignant lesions with saliva miRNA analysis. BMC Cancer. 13 (1), 129 (2013).
Yap, T. Oral cancer prediction using microRNA in oral swirls. (2018).
Prasad, G. et al. A panel of microRNAs can be used to determine oral squamous cell carcinoma. J. Oral Pathol. Med. 46 (10), 940–948 (2017).
Kaunein, N. et al. A systematic review of microRNA signatures associated with the progression of leukoplakia with and without epithelial dysplasia. Biomolecules, 11(12), (2021).
Prasad, G. Development of a molecular tool for oral squamous cell carcinoma risk assessment using microrna markers. (2014).
El-Naggar, A. K. et al. The fourth edition of the head and neck World Health Organization blue book: editors’ perspectives. Hum. Pathol. 66, 10–12 (2017).
Kaunein, N. et al. A Systematic Review of MicroRNA Signatures Associated with the Progression of Leukoplakia with and without Epithelial Dysplasia. Biomolecules 11 (12), 1879 (2021).
Cervigne, N. K. et al. Identification of a microRNA signature associated with progression of leukoplakia to oral carcinoma. Hum. Mol. Genet. 18 (24), 4818–4829 (2009).
Brito, J. A. et al. Relationship between microRNA expression levels and histopathological features of dysplasia in oral leukoplakia. J. Oral Pathol. Med. 43 (3), 211–216 (2014).
Philipone, E. et al. MicroRNAs-208b-3p, 204-5p, 129-2-3p and 3065-5p as predictive markers of oral leukoplakia that progress to cancer. Am. J. cancer Res. 6 (7), 1537–1546 (2016).
Harrandah, A. M. et al. MicroRNA-375 as a biomarker for malignant transformation in oral lesions. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology, 122(6): pp. 743–752.e1. (2016).
Lu, Y. C. et al. Combined determination of circulating miR-196a and miR-196b levels produces high sensitivity and specificity for early detection of oral cancer. Clin. Biochem. 48 (3), 115–121 (2015).
Kao, Y. Y. et al. The increase of oncogenic miRNA expression in tongue carcinogenesis of a mouse model. Oral Oncol. 51 (12), 1103–1112 (2015).
Yap, T. et al. Predicting the Presence of Oral Squamous Cell Carcinoma Using Commonly Dysregulated MicroRNA in Oral Swirls. Cancer Prev. Res. (Phila). 11 (8), 491–502 (2018).
Xie, F., Wang, J. & Zhang, B. RefFinder: a web-based tool for comprehensively analyzing and identifying reference genes. Funct. Integr. Genomics. 23 (2), 125 (2023).
Yan, B. et al. Downregulation of microRNA 99a in oral squamous cell carcinomas contributes to the growth and survival of oral cancer cells. Mol. Med. Rep. 6 (3), 675–681 (2012).
Farshbaf, A. et al. Clinical value and potential circulating of miR-99a as tumor suppressor biomarker in serum of oral squamous cell carcinoma and erosive atrophic lichen planus. Journal of Stomatology, Oral and Maxillofacial Surgery, 125(3, Supplement): p. 101806. (2024).
Sun, X. & Yan, H. MicroRNA-99a-5p suppresses cell proliferation, migration, and invasion by targeting isoprenylcysteine carboxylmethyltransferase in oral squamous cell carcinoma. J. Int. Med. Res. 49 (5), 300060520939031 (2021).
Chen, L. et al. Diagnostic and prognostic value of serum miR-99a expression in oral squamous cell carcinoma. Cancer Biomark. 23 (3), 333–339 (2018).
Cousseau, C. P. V. et al. miR-99a-5p as a biomarker for lymph node metastasis prediction in oral squamous cell carcinoma patients. Head Neck. 45 (10), 2489–2497 (2023).
Li, J. et al. MiR-21 indicates poor prognosis in tongue squamous cell carcinomas as an apoptosis inhibitor. Clin. Cancer Res. 15 (12), 3998–4008 (2009).
Hou, B. et al. Circulating microRNAs as novel prognosis biomarkers for head and neck squamous cell carcinoma. Cancer Biol. Ther. 16 (7), 1042–1046 (2015).
Iliopoulos, D. et al. STAT3 Activation of miR-21 and miR-181b-1 via PTEN and CYLD Are Part of the Epigenetic Switch Linking Inflammation to Cancer. Mol. Cell. 39 (4), 493–506 (2010).
Dioguardi, M. et al. MicroRNA-21 Expression as a Prognostic Biomarker in Oral Cancer: Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public. Health, 19(6). (2022).
Hedbäck, N. et al. MiR-21 expression in the tumor stroma of oral squamous cell carcinoma: an independent biomarker of disease free survival. PLoS One. 9 (4), e95193 (2014).
Jenike, A. E. & Halushka, M. K. miR-21: a non-specific biomarker of all maladies. Biomark. Res. 9 (1), 18 (2021).
Nevanpää, T. T. et al. Malignant transformation of oral epithelial dysplasia in Southwest Finland. Sci. Rep. 12 (1), 8261 (2022).
Dioguardi, M. et al. The Potential microRNA Prognostic Signature in HNSCCs: A Systematic Review. Non-Coding RNA. 9 (5), 54 (2023).
Yoon, A. J. et al. MicroRNA-based risk scoring system to identify early-stage oral squamous cell carcinoma patients at high-risk for cancer-specific mortality. Head Neck. 42 (8), 1699–1712 (2020).
Acknowledgements
This work was generously funded by the Australian Dental Research Foundation (ADRF), Early Career Research Grant (UoM), Australia and New Zealand Head and Neck Cancer Society Research Foundation (ANZHNCSRF), Medical Research Future Fund – BioMedTech Horizons (BMTH) program awarded in partnership with Optiscan Imaging. Nadia Kaunein was a recipient of an Australian Government Research Training Program Scholarship provided by the Australian Commonwealth Government and the University of Melbourne.
Funding
Australian Dental Research Foundation(ADRF), Early Career Research Grant(UoM), Australia-New Zealand Head and Neck Cancer Society Research Foundation(ANZHNCSRF), Medical Research Future Fund–BioMedTech Horizons(BMTH) program awarded with Optiscan-Imaging. NK was a recipient of Australian Government Research Training Program Scholarship.
Author information
Authors and Affiliations
Contributions
NK-Writing-original draft, data curation, formal analysis, investigation, methodology, project admin, visualization, writing review and editing.MMC- Inception, Experiment Design and Conduction, Funding, Manuscript Review, Supervision, Writing – review and editingCB- Supervision, Writing - review and editingNB- Methodology, Writing – review and editingIV- Methodology, Writing – review and editingCM- Writing – review and editingCA- Writing - review and editingTY- Inception, Experiment Design and Conduction, Funding, Manuscript Review, Writing – review and editing.
Corresponding author
Ethics declarations
competing interests
The authors declare no competing interests.
Ethics
The study was approved by the Human Research Ethics Committee of the University of Melbourne (2021-13213-18885-5) and the Royal Dental Hospital Melbourne (2021- 22004-22381-5). Informed written consent was obtained from all participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kaunein, N., McCullough, M., Butler, C. et al. Oral mucosal scrapes capture cancer associated microRNA expression consistent with histopathology. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45078-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45078-z