Abstract
Major depressive disorder is highly prevalent and disabling, and many patients do not respond to standard therapies, resulting in treatment-resistant depression (TRD). Progress in TRD requires preclinical models with poor response to conventional treatments. The Wistar-Kyoto rat exposed to chronic mild stress (WKY/CMS) offers a robust paradigm: it shows limited responsiveness to standard antidepressants yet remains sensitive to interventions effective in TRD (esketamine, ECT), making it well-suited to probe TRD mechanisms and evaluate candidate therapies. To extend the characterization of this model—previously unstudied with quantitative 1H-MRS or DTI—we compared WKY/CMS rats with non-depressed controls, quantifying brain metabolism by 1H-MRS and assessing structural alterations with T2-weighted MRI and 30-direction DTI. In WKY/CMS rats, 1H-MRS revealed reduced glutamate, glutamine, and taurine, and increased myo-inositol in the prefrontal cortex (PFC), along with decreases in glutamine, choline-containing compounds, and macromolecular signals at 0.9 and 1.4 ppm in the hippocampus (Hip). DTI revealed increased mean diffusivity in the PFC and Hip of WKY/CMS rats—consistent with demyelination and/or axonal loss—and reduced fractional anisotropy in the Hip, suggesting compromised white-matter integrity. Overall, the WKY/CMS profile reflects depression- and stress-related metabolic and microstructural changes, supporting translational studies and testing of interventions relevant to treatment resistance (e.g. rTMS, psychedelics).
Similar content being viewed by others
Introduction
Major depressive disorder (MDD) affects millions of people worldwide and remains difficult to treat, with many patients developing treatment-resistant depression (TRD) when conventional therapies prove inadequate. However, most preclinical models are sensitive to antidepressants, limiting their translational value for TRD and highlighting the need for models with poor response to conventional therapies. Depression models can be generated through environmental, pharmacological, or genetic manipulations. Among environmental paradigms, chronic social defeat stress (CSDS), chronic restraint stress (CRS), and chronic mild stress (CMS) are widely used, each producing depressive-like phenotypes reversible by chronic antidepressant treatment, including selective serotonin reuptake inhibitors (SSRIs, e.g. fluoxetine, citalopram), tricyclic antidepressants (TCAs, e.g. imipramine), and serotonin–norepinephrine reuptake inhibitors (SNRIs, e.g. venlafaxine)1,2,3,4,5. Pharmacological models, such as chronic administration of corticosterone or adrenocorticotropic hormone (ACTH), also induce depression-like phenotypes that respond to several classes of antidepressants, including SSRIs, TCAs, and melatonergic agents, as well as to adjunctive interventions such as prebiotic compounds6,7,8,9. Surgical or genetic models, such as olfactory bulbectomy or transgenic lines with targeted gene deletions, likewise show antidepressant-sensitive behavioural and neurochemical alterations10,11. While these paradigms have proven valuable for studying pathophysiological mechanisms, their sensitivity to multiple classes of antidepressants limits their translational relevance for modelling TRD.
In this context, a particularly relevant example is the Wistar-Kyoto (WKY) rat strain, which was originally developed as a normotensive control group for studies involving the spontaneously hypertensive rat (SHR)12, but was later shown to exhibit a depression-like phenotype comparable to that observed in patients, leading to its current recognition as a well-established animal model of endogenous depression13,14.
The biological validity of the WKY model is supported by convergent endocrine, neurochemical, and synaptic alterations that parallel core pathophysiological features of human MDD.
WKY rats exhibit hormonal, behavioural, and physiological characteristics resembling MDD patients’ abnormalities15, such as heightened stress reactivity, altered emotional behaviour, anhedonia, helplessness, anxiety-like traits, cognitive deficits, social behaviour deficits, monoaminergic abnormalities, reduced synaptic plasticity (investigated by means of electrophysiological techniques, e.g. evoked potential recordings, LTP, LTD), and functional (fMRI) as well as structural brain alterations16. The latter were identified either through imaging studies, for example manually using the MIPAV software17, or using the Cavalieri method on fixed brain sections18.
The chronic mild stress (CMS) paradigm, the most widely used and validated rodent model of depression1, consists of exposing animals to a series of mild and unpredictable stressors over several weeks, resulting in behavioural, biochemical, and physiological alterations resembling depressive symptomatology19,20. Applying this paradigm to a strain with an inherent depressive-like phenotype, such as the WKY rat, provides a unique opportunity to investigate the interaction between environmental stress and genetic vulnerability. Reflecting their baseline hypersensitivity to stress, WKY rats display heightened physiological responses and impaired adaptation to repeated stress exposure, including exaggerated ACTH responses and greater stress-ulcer severity compared with Wistar controls15,21,22,23. Under CMS, they develop behavioural changes similar to those observed in Wistar rats, but unlike Wistars, these alterations are not adequately reversed by conventional antidepressants such as imipramine, citalopram, or venlafaxine24. More generally, the WKY strain exhibits poor responsiveness to both acute and chronic treatment with traditional antidepressants, including tricyclics (e.g. imipramine, desipramine) and SSRIs (e.g. fluoxetine, citalopram)16,25,26. In clinical practice, patients with TRD likewise often require non-monoaminergic or neuromodulatory interventions—such as ketamine/esketamine, repetitive transcranial magnetic stimulation (rTMS, including accelerated high-frequency protocols), electroconvulsive therapy (ECT), or deep brain stimulation (DBS) of fronto-limbic targets—which can exert antidepressant effects in individuals who have failed multiple adequate trials of standard antidepressants27,28,29,30,31,32,33. Consistently, WKY rats show poor response to conventional antidepressants but retain sensitivity to subacute ketamine, rTMS, ECT, and vmPFC DBS24,26,34,35,36,37, reinforcing the translational relevance of this model for investigating TRD-related mechanisms and testing novel therapeutic strategies.
WKY rats exhibit endocrine dysregulation involving both the hypothalamic–pituitary–thyroid (HPT) and hypothalamic–pituitary–adrenal (HPA) axes. Elevated basal plasma TSH and T3 levels have been reported, paralleling findings in TRD patients23,38. In addition, WKY rats show heightened and prolonged stress-induced endocrine responses, impaired glucocorticoid-mediated negative feedback, and adrenal hypertrophy13,39,40,41,42,43, consistent with a chronically activated or disinhibited HPA axis44.
Several laboratories and a substantial amount of research have shown that, compared to control rats, the WKY strain displays depressive-like behaviours and passive coping strategies across a variety of behavioural tests (for a review see13,14,16). These include increased anhedonia, helplessness, neophobia, anxiety, and impairments in social interactions, aggression, and social play, reflecting social avoidance16.
Numerous alterations in monoaminergic pathways have been reported in WKY rats compared to control strains, paralleling findings in clinical depression. Reduced levels of norepinephrine (NE), serotonin (5-HT), and dopamine (DA) have been observed across several brain regions, including the Hip, PFC, nucleus accumbens (NAc), and basolateral amygdala (BLA), together with receptor and transporter abnormalities45. In particular, WKY rats show marked changes in the distribution and density of dopamine transporters (DAT) and DA receptors (D1, D2, D3) within the mesolimbic system, leading to attenuated tissue DA levels in Hip, PFC, NAc shell, and BLA. Such alterations are thought to contribute to depressive-like behaviours, including anhedonia, social withdrawal, and cognitive impairments16,45,46,47,48.
WKY rats show either comparable or reduced basal serum BDNF levels relative to Wistar controls49 together with significantly decreased BDNF and TrkB expression in Hip and in the PFC50. They also display markedly attenuated adult neurogenesis in the dentate gyrus (DG), while neurogenesis in the subventricular zone (SVZ) remains unaffected51. According to the neurotrophic hypothesis, reduced BDNF and impaired neurogenesis may weaken glucocorticoid-mediated negative feedback on the HPA axis and compromise stress resilience, thereby contributing to depression52. Consistently, patients with MDD exhibit downregulation of BDNF and TrkB at both protein and transcript levels53,54.
Beyond functional alterations, biochemical evidence also points to marked neurometabolic changes in WKY rats. Nagasawa et al. (2012) reported that brain amino acid metabolism in WKY rats differs substantially from that of Wistar controls, with particularly pronounced reductions in serine and cystathionine and concurrent abnormalities in dopamine metabolism. In a subsequent study, Nagasawa et al. (2015) used high-performance liquid chromatography (HPLC) to measure plasma and brain free amino acid concentrations and found widespread alterations across multiple regions—including prefrontal cortex, striatum, thalamus, hypothalamus, hippocampus, cerebellum, and brainstem—with several excitatory and inhibitory amino acids reduced in WKY rats and partially normalized by chronic imipramine treatment55. Together, these findings support the view that altered amino acid and monoamine metabolism contribute to the depressive-like phenotype and antidepressant sensitivity of the WKY strain, underscoring the relevance of probing neurometabolic changes in this model using in vivo 1H-MRS.
Although the WKY/CMS rat model has not yet been investigated using quantitative magnetic resonance spectroscopy (MRS) or diffusion tensor imaging (DTI), several fMRI studies have examined neural activity and functional connectivity (FC). For example, Li and co-authors (2022) investigated the amplitude of low-frequency fluctuation (ALFF) values in WKY rats vs. Wistar controls56. In that study, ALFF, an rs-fMRI metric indexing the amplitude of spontaneous low-frequency BOLD oscillations, was used as a proxy of regional intrinsic activity rather than a direct measure of synaptic plasticity57,58. Control Wistar rats showed a time-dependent increase in ALFF within the NAc, whereas WKY rats did not, in a region that is considered highly plastic within the reward circuitry59. On this basis, the authors interpreted the blunted ALFF increase in WKY rats as reduced functional plasticity of NAc activity, in line with human intervention studies in which longitudinal changes in ALFF have been used as an indirect marker of experience-dependent brain plasticity60,61. Overall, the ALFF abnormalities observed in the dorsal striatum and hippocampus of WKY rats parallel rs-fMRI findings of altered spontaneous activity in hippocampal and thalamo–striatal regions in patients with MDD62, and are consistent with structural hippocampal alterations reported in first-episode MDD63, supporting the translational relevance of this functional phenotype.
Resting-state fMRI has also revealed regional and large-scale network alterations in WKY rats. Compared with Wistar controls, WKY rats show globally reduced FC and lower network resilience, segregation, efficiency, and integration, together with reduced FC involving the medial PFC, anterior cingulate, sensorimotor and parietal cortices56. These patterns closely resemble fronto–limbic and associative network disturbances reported in patients with MDD64,65. Graph-theoretical hubness analysis further indicated that the somatosensory cortex, a functional hub in control rats, loses its hub-like properties in WKY rats, paralleling reports linking somatosensory cortex alterations to emotional dysregulation and MDD66,67. Together with the ALFF findings described above, these rs-fMRI results point to widespread functional network disruption in WKY rats that mirrors fronto-limbic and sensory disturbances reported in patients.
Finally, voxel-based morphometry (VBM) in WKY rats compared with Wistar controls showed atrophy in the vHip, right caudate putamen, right lateral septum, cerebellar vermis, and cerebellar nuclei68. These patterns partially overlap with atrophic regions reported in MDD patients69,70, supporting the translational relevance of the WKY model.
Here, we compared a group of adult male WKY rats exposed to CMS for 4 weeks (hereafter referred to as WKY/CMS) with a corresponding group of undisturbed Wistar rats (hereafter referred to as W/CTRL), assessing brain metabolism in the PFC and Hip with quantitative MRS and investigating structural alterations using T2-weighted MRI and 30-gradient direction DTI. Based on prior 1H-MRS findings across rodent models of depression/stress, we expected region-specific alterations in metabolites related to glutamatergic metabolism and glial/inflammatory processes (e.g. glutamine, Gln; glutamate, Glu; myo-inositol, Ins), with several reports describing reduced Glx/Gln and increased Ins particularly in prefrontal and hippocampal regions, albeit with variability across models and acquisition/analysis protocols.
Results
Brain morphological differences in WKY/CMS vs. W/CTRL rats
Anatomical images revealed a shorter, more ellipsoid-shaped brain in WKY/CMS rats compared to W/CTRL. This difference, particularly evident in the sagittal view (see Fig. 1), was consistently observed across all subjects (see Supplementary Fig. 1 for representative axial and sagittal images from each animal).
Comparison between W/CTRL (yellow, upper row) and WKY/CMS (green, lower row) rat brains by using T2W MRI (TR/TE = 2500/33 ms, one average, one repetition, matrix = 256 × 256, slice thickness = 1 mm, in coronal (15 slices, FOV = 30 × 25 mm2), axial (27 slices, FOV = 25 × 25 mm2) and sagittal (19 slices, FOV = 30 × 25 mm2) orientations).
Differences were also observed in ventricular size, as well as in the shape and thickness of the PFC (Fig. 2). Specifically, the thickness of the cingulate cortex (averaged between hemispheres) at –1.30 mm from bregma was significantly reduced in the WKY/CMS rats (1.39 ± 0.07 mm) compared to W/CTRL (1.49 ± 0.06 mm; p = 0.003). Similarly, the average Hip thickness at –3.60 mm from bregma was lower in WKY/CMS rats (1.82 ± 0.03 mm) than in W/CTRL (1.90 ± 0.05 mm; p < 0.01).
Thickness of cingulate cortex and Hip in W/CTRL (yellow) vs. WKY/CMS (green) by using high resolution T2W MRI (TR/TE = 3463/33 ms, matrix 192 × 192, FOV = 25 × 25mm2, thickness = 0.6 mm, 1 average, axial orientation). Upper row: cingulate cortex; lower row Hip. The purple double-headed arrows denote the regions where thickness was measured.
Metabolic alterations in the PFC and Hip of WKY/CMS rats
A total of 11 metabolites, including macromolecular and lipid signals, were quantified. Specifically, to identify metabolic alterations associated with the MDD/TRD model, the metabolic profiles of the PFC and Hip in W/CTRL and WKY/CMS rats were subjected to univariate non-parametric test and multivariate analysis. Representative 1H MR spectra of the PFC and Hip of W/CTRL (blue line) and WKY/CMS rats (red line) are shown in Fig. 3a and d. Regarding the MM14 and Lip13 signals, applying a 20% exclusion criterion would have led to the removal of many subjects from both univariate and multivariate analyses. Among subjects with CRLB < 20%, no differences were observed in the Lip13 signal, which is typically very low in the brain. Conversely, we detected a reduction in the MM14 signal in WK/CMS rats, comparable to the decrease observed in the combined MM14 + Lip13 signal. Therefore, we decided to replace MM14 with the composite MM14 + Lip13 signal—obtained by summing the two resonances—for subsequent analyses, considering that its decrease is primarily driven by the reduction in MM14.
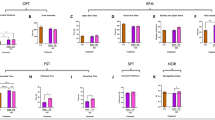
Comparison between W/CTRL and WKY/CMS rats using quantitative1H-MRS univariate analysis. (a) Representative water-suppressed 1H-MRS spectra acquired using the s-LASER sequence (TR/TE = 4000/20.6 ms, nt = 256) from the PFC of a W/CTRK rat (blue line) and a WKY/CMS rat (red line). Red and green arrows indicate metabolites with increased or decreased signal intensity, respectively, in the MDD/TRD model. (b) Heatmap showing significantly altered metabolites in WKY/CMS compared to W/CTRL, based on FDR-corrected Wilcoxon rank-sum test. Corresponding p values are reported in Table 1. (c) Metabolite concentrations (in mM) in the PFC measured by in vivo 1H-MRS in W/CTRL and WKY/CMS rats (mean ± SE; *p < 0.05, **p < 0.01). (d), (e), and (f) show the corresponding representative spectra, heatmap, and metabolite concentrations obtained in the Hip. The mean ± SD values of all metabolites, macromolecular, and lipid signals that met the inclusion criterion (see Methods – Statistical analysis) are reported in Supplementary Table 2. The mean ± SD of the quality metrics for the spectra (FWHM and SNR) and for the individual metabolites and macromolecular signals (CRLBs) included in the analysis in the PFC and Hip are reported in Supplementary Table 3, as recommended by the consensus group on Minimum Reporting Standards for in vivo Magnetic Resonance Spectroscopy (MRSinMRS)71.
No significant differences were observed between the T2 values of WKY/CMS and W/CTRL rats in either region Supplementary Table 1.
Quantitative univariate analyses based on Wilcoxon rank-sum test corrected with FDR for multiple comparison showed, in the WKY/CMS group, a significant decrease in Glu, Gln, taurine (Tau) and an increase in Ins in the PFC (Fig. 3b and c). In the Hip, the same analyses revealed decreases in Gln, choline-containing compounds (tCho), and macromolecular signals resonating at 0.9 and 1.4 ppm (MM09 and MM14) (Fig. 3e and f). The p values of these metabolites are reported in Table 1.
Both univariate and multivariate analyses were conducted to evaluate not only single metabolite variations but also the combined metabolic pattern distinguishing the two experimental groups.
PLS-DA multivariate analysis revealed a clear separation between W/CTRL and WKY/CMS groups, with high predictive performance based on cross-validation (Q2 = 0.84 for the PFC and 0.69 for the Hip, further details can be found in Supplementary Table 4). In the PFC, metabolites with a variable importance in the projection (VIP) score > 1 that contributed significantly to the group separation included Gln, Glu, Tau, and total N-acetylaspartate (tNAA, i.e., NAA + NAAG), as shown in Fig. 4a, b and c. In the Hip, metabolites with VIP > 1 included Gln, MM09 and MM14, as illustrated in Fig. 4d, e, and f. The metabolites identified as significantly different in the univariate analysis were also detected by the multivariate approach, indicating correlated changes and a joint contribution to group discrimination. The tCho signal in the Hip, although showing the largest individual decrease in the univariate analysis, contributed less prominently to the multivariate model and therefore did not emerge among the main discriminators in the PLS-DA loading plots.
Quantitative 1H-MRS-based multivariate analysis comparing W/CTRL and WKY/CMS rats. (a) PLS-DA score plot, (b) corresponding VIP scores, and (c) loading plot for the PFC. (d), (e), and (f) show the respective score plot, VIP scores, and loading plot for the Hip.
Structural alterations in the PFC, Hip and corpus callosum of WKY/CMS rats
Quantitative univariate analyses based on Wilcoxon rank-sum test corrected with FDR for multiple comparison showed significant increase in mean diffusivity (MD) in PFC and Hip (Fig. 5a) and decreased fractional anisotropy (FA) in Hip (Fig. 5b).
Comparison of W/CTRL and WKY/CMS rat brains using DTI. Mean diffusivity (MD) was increased in both the PFC and Hip, while fractional anisotropy (FA) was reduced in the Hip in WKY/CMS compared to W/CTRL rats (p < 0.05).
Discussion
The WKY/CMS model combines the genetic vulnerability of the WKY strain with the additional burden of CMS, providing a robust paradigm for the study of depression. WKY rats display a broad range of alterations—endocrine dysregulation, behavioural abnormalities consistent with depressive- and anxiety-like phenotypes, monoaminergic dysfunction, and reduced neurotrophic support—which mirror several pathophysiological hallmarks of MDD. Importantly, these animals show poor responsiveness to conventional antidepressants, while retaining sensitivity to innovative therapeutic approaches such as ketamine, ECT and DBS. This pharmacological profile strengthens the value of the WKY/CMS model for investigating mechanisms underlying treatment-resistant depression (TRD) and for testing novel interventions.
Building on this framework, we sought to expand the characterization of the WKY/CMS model by applying a multimodal MRI protocol. While previous investigations have primarily relied on fMRI to assess neural activity and FC, quantitative MRS and DTI had not yet been employed in this model. To address this gap, we compared WKY/CMS rats with W/CTRL counterparts, analysing brain metabolism in the PFC and Hip through quantitative MRS, and assessing structural alterations via T2-weighted MRI and 30-gradient direction DTI.
Brain morphological differences
While total brain volume appears preserved, notable differences were detected in frontal cortical thickness (cingulate cortex) and Hip, accompanied by a clear enlargement of the ventricles. These morphological differences were consistently observed across all subjects, as illustrated in Supplementary Fig. 1. These findings align with observations in depressed patients: Zhao and co-authors (2014) reported grey matter reductions in the prefrontal-limbic network in a cohort of 400 medication-free patients with MDD compared to 424 healthy controls72. In 2019, Klok and co-authors showed significant volume reductions in frontal regions of MDD patients, and distinguished TRD patients by means of specific alterations in the putamen and parietal white matter tracts. More recently, Kang and co-authors (2024) found that cortical thickness reductions in the corticolimbic circuit of MDD patients were correlated with neuroinflammatory markers in serum73. Additionally, a study on depressed adolescent patients reported volume decreases in several brain regions—including the cingulate cortex—with greater reductions associated with longer durations since MDD onset74.The Hip was also investigated in depressed adolescent patients, and abnormalities were found in the CA regions and DG, subfields primarily associated with memory impairments75.
Our findings extend the work of Yoshii and co-authors43, who applied voxel-based morphometry to compare WKY rats and Wistar rats, both housed in solitary conditions as a mild stress paradigm. As social isolation in Wistar rats is known to induce a drug-responsive depressive phenotype, their study addressed differences between two depression-related models. In contrast, by exposing WKY rats to CMS and using pair-housed Wistar rats as non-depressed controls, we further demonstrate that the WKY/CMS condition reproduces several brain morphological differences resembling those observed in depressed patients.
Metabolic alterations
MRS is widely employed to investigate neurochemical alterations in depression. Both multivariate and univariate analyses revealed significant alterations in several metabolites within the PFC and Hip of the WKY/CMS model of MDD/TRD.
MRS studies in MDD patients reveal pronounced metabolic abnormalities, emphasizing the role of glutamatergic and GABAergic dysfunction. Moriguchi and co-authors conducted a meta-analysis of 1H-MRS studies examining Glu levels and found significantly decreased levels of glutamatergic metabolites in the medial frontal cortex in patients with depression compared with controls76. Consistent with these clinical data, we likewise observed reduced Glu in the PFC of WKY/CMS rats (an MDD/TRD model). 1H-MRS quantifies regional metabolite concentrations (tissue pools) and does not directly measure synaptic glutamate transmission. However, alterations in Glu- and Gln-related measures are frequently reported across depression/stress studies and may reflect disruptions in glutamatergic metabolism and neuronal–glial coupling. Accordingly, we observed reduced Gln levels in the PFC, consistent with a previous study in Sprague–Dawley rats exposed to the CMS paradigm, which likewise reported decreased Gln in both the PFC and Hip77. Similar reductions in Gln have also been reported across several rodent models of depression/stress (e.g., in the NAc following chronic restraint stress78,79).
A meta-analysis of clinical 1H-MRS studies by Schür and co-authors (2016) provided evidence for lower GABA levels, reflecting impaired inhibitory neurotransmission, in depressed (but not remitted) MDD patients compared with healthy controls80. However, a recent review summarising preclinical 1H-MRS findings reported that GABA results across MDD rodent models are inconsistent79. In our study, GABA could not be reliably quantified in most WKY/CMS rats, as LCModel estimates it with CRLB that exceeded the threshold for inclusion. Therefore, we could not assess potential group differences in GABA levels. Future studies employing GABA-edited MRS sequences will be required to confirm whether GABA alterations are present in this model.
We did not detect group differences in tNAA in either the PFC or hippocampus. This is not inconsistent with the broader MRS literature in rodent depression/stress models, where NAA alterations appear variable and region-/model-dependent: Pavlova & Ruda-Kucerova79 summarize reports of both increased NAA in some cortical regions and decreased NAA in hippocampal and other regions across different paradigms. Given that decreases in NAA are often interpreted as reflecting neuronal dysfunction whereas the basis for NAA increases remains less clear79, the absence of tNAA changes in our regions of interest may reflect this heterogeneity across models, brain regions, and experimental conditions.
Ins is often interpreted in 1H-MRS studies as a glia-enriched metabolite, although it does not provide direct evidence of gliosis81,82,83. It plays key roles in osmoregulation, cell signalling (as a precursor of phosphatidylinositol), membrane turnover, and second-messenger systems. Elevated Ins levels have been reported in 1H-MRS studies, including an increase in the PFC of patients diagnosed with bipolar depression84. Such increases are generally considered consistent with glial or neuroinflammatory involvement, possibly reflecting altered astrocyte–neuron interactions or dysregulated osmotic and membrane processes85. Higher Ins levels may therefore serve as a potential biomarker for specific subtypes of depression, particularly those involving neuroinflammatory mechanisms. Another study reported increased Ins levels in the PFC of migraine patients with comorbid MDD86. This elevation may result from increased uptake or retention of Ins within the cellular or extracellular matrix, or from disrupted receptor coupling with secondary messenger systems. Reactive gliosis has also been proposed as one possible mechanism underlying elevated Ins levels, especially in elderly patients with depression87, although this remains speculative and would require histological confirmation. Finally, the increase in Ins observed in the PFC of the WKY/CMS group is consistent with a previous study in Sprague–Dawley rats using the forced swim test (FST) model, which reported elevated Ins in the left dorsolateral PFC88, although our PFC voxel encompassed the prefrontal cortex without hemispheric subdivision.
Taurine is one of the most abundant free amino acids in the central nervous system. Although it is not incorporated into proteins, taurine plays multiple critical roles, including inhibitory neurotransmission, neuromodulation, osmotic regulation, membrane stabilization, antioxidant defence, detoxification, and calcium homeostasis. It also modulates GABAergic and glycinergic activity. In our study, taurine levels were reduced, consistent with previous findings of decreased taurine in the PFC of a rat model of depression89. Taurine supplementation has been shown to exert anxiolytic-like and antidepressant-like effects in a CMS-induced depression mouse model by acting as an inhibitory neuromodulator through GABA and glycine receptors90. Through this mechanism, taurine may help counteract glutamate-related neuronal stress and overactivation of the HPA axis—processes proposed to occur in depression based on post-mortem evidence of astroglial and glutamatergic abnormalities91,9293. Although direct evidence of frank excitotoxicity in depression remains limited, these post-mortem findings are consistent with an increased risk of excitotoxic stress. In a chronic-stress mouse model, treatment with a mixture of creatine and taurine restored stress-induced reductions in monoamine levels and downregulated stress hormone and pro-inflammatory cytokine expression, suggesting a potential neuroprotective and anti-inflammatory action of taurine in stress-related conditions90. Furthermore, recent studies and reviews have highlighted taurine’s broader neuroprotective, antioxidant, and anti-inflammatory roles, including modulation of calcium homeostasis and astrocytic function94,95. Taken together, these findings support the view that taurine may contribute to maintaining neural and glial homeostasis and protecting brain circuits disrupted in mood disorders.
Total choline (tCho) represents the combined 1H-MRS signal from choline-containing compounds, primarily free choline, phosphocholine, and glycerophosphocholine, which play essential roles in membrane synthesis and turnover, cell signalling, and osmoregulation. In adolescent patients diagnosed with depression, significantly lower levels of N-acetylaspartate and choline have been reported in prefrontal white matter regions96. In our study, the reduction of tCho observed in the Hip may reflect altered membrane phospholipid metabolism, suggesting reduced membrane turnover or impaired intracellular signalling. However, findings on tCho in depression are heterogeneous. Several MRS studies have reported increases or no change in tCho levels, while decreases have been occasionally observed in specific subgroups or brain regions, such as in bipolar or late-life depression97,98. Meta-analytic reviews also highlight substantial variability across studies, reflecting methodological differences and population heterogeneity99. Our results may therefore represent model-specific alterations in phospholipid and membrane dynamics rather than a consistent biomarker of depression. The role of macromolecular (MM) signals in MDD remains poorly understood, as they are often treated as background components in MRS quantification rather than as primary outcomes. In our study, we observed reduced macromolecular signals at 0.9 ppm (MM09) and 1.4 ppm (MM14) in the Hip of WKY/CMS rats. This may reflect model-specific neurochemical features, as WKY rats have not previously been characterized by MRS.
Consistent with expert recommendations on MM assignments, the MM09 peak is thought to arise primarily from cytosolic macromolecules, including polypeptides and proteins enriched in branched-chain amino acids (BCAAs) such as leucine, isoleucine, and valine70. These BCAAs donate amino groups (–NH₂) in brain metabolism through transamination reactions catalysed by branched-chain amino acid aminotransferase (BCAT), transferring amino groups to α-ketoglutarate to form Glu and thereby contributing to the glutamate–glutamine cycle that regulates excitatory neurotransmission. In parallel, peripheral studies have reported significantly lower circulating BCAA levels in patients with MDD, with inverse associations with symptom severity71. Although our hippocampal reductions in MM09 and MM14 cannot be interpreted as direct in-vivo readouts of brain BCAA concentrations, the directionality is compatible with a diminished contribution from BCAA-rich macromolecular components. We therefore hypothesize that BCAA-related metabolic pathways may be altered in this model. This interpretation remains tentative, as MM signals are composite and peripheral BCAA levels do not necessarily mirror brain concentrations; further studies will be needed to test this link.
In addition to the MM09 component, we also observed a reduction in the MM14 signal, which may reflect decreased contributions from threonine-containing macromolecules The observed reduction in the MM14 signal may reflect decreased contributions from threonine-containing macromolecules. Threonine is an essential amino acid obtained through dietary intake and serves in the brain as a precursor for glycine and serine—two amino acids crucial for neurotransmission, synaptic plasticity, and metabolic support of neurons and glia. It also contributes to the synthesis of structural proteins and myelin, which are vital for maintaining neural integrity. Reduced threonine availability could therefore indicate altered amino acid metabolism and protein turnover. Moreover, threonine metabolism is functionally linked to the activity of glycogen synthase kinase-3β (GSK3β), a serine/threonine kinase implicated in mood regulation, neuroinflammation, and cognitive impairment. Interestingly, hyperactivation of GSK3β has been reported in a rat model of chronic mild stress, suggesting a potential connection between altered threonine-related pathways and depressive phenotypes100. Given that macromolecular signals are composite and their cellular assignments remain uncertain, the observed MM14 reduction should be interpreted with caution. To our knowledge, no prior in-vivo MRS study has directly linked MM14 alterations to depression. Methodological and human in-vivo studies indicate that MM peaks can vary due to modelling choices and non-disease factors such as age or tissue composition101,102,103,104. Accordingly, our working hypothesis that reduced MM14 may indicate altered contributions from specific amino acid-containing macromolecules remains preliminary and warrants targeted validation through combined MRS and biochemical analyses.
Structural alterations
In our study, DTI—which assesses white matter integrity by characterizing directional water diffusion along axonal pathways—identified microstructural abnormalities that were not detected with conventional structural MRI.
Several studies have demonstrated that TRD patients exhibit alterations in key DTI metrics. Main findings include: (i) reduced FA, suggesting compromised white matter integrity—often in tracts connecting the PFC, anterior cingulate cortex, and limbic structures; (ii) increased MD, reflecting potential neuroinflammation, demyelination, or reduced axonal density; and (iii) altered axial (AD) and radial (RD) diffusivity, which can provide additional insights into axonal damage vs. myelin disruption. These alterations have been most consistently observed in white matter tracts within the fronto-limbic network and may underlie impaired emotion regulation and cognitive dysfunction commonly reported in TRD patients105,106,107 and in WKY rats13,14,16.
The increase in MD observed in the PFC and the Hip in the WKY/CMS group may reflect underlying neuroinflammation, demyelination, or reduced axonal density. In parallel, the reduction in FA detected in the Hip suggests compromised white-matter integrity. Overall, these DTI-based alterations in both mean diffusivity (MD) and fractional anisotropy (FA) identified in the MDD/TRD model support the presence of microstructural brain abnormalities, in line with those consistently reported in patients with TRD96,105,106,107,108.
Limitations
Our cortical thickness analysis focused on the cingulate cortex and did not extend to prefrontal areas. In the WKY/CMS model we observed a strain-specific distortion of brain shape (shorter anteroposterior length and wider mediolateral extent), which made the delineation and registration of prefrontal ROIs unreliable and increased the risk of bias due to misregistration and partial-volume effects. For this reason, we refrained from reporting PFC thickness estimates and limited inference to the cingulate cortex and Hip. These constraints should not be interpreted as evidence of absent prefrontal alterations. Future studies should employ whole-brain morphometric approaches, such as voxel-based morphometry, to robustly detect prefrontal structural abnormalities74.
Although LCModel estimates the concentration of all metabolites, some resonances partially overlap in spectra acquired at 7 T. This overlap can introduce covariance and increase uncertainty, particularly in hippocampal voxels. To minimise this, we applied a 20% CRLB threshold and report group-level CRLBs in Supplementary Table 3. Adopting a more stringent CRLB threshold (e.g. < 15%) may represent a more conservative criterion. However, we believe that including metabolites with CRLB values < 20% remains informative. In addition, long scans, such as those adopted in our MRS protocol, could further benefit from phase and/or frequency corrections. Despite this technical limitation of our setup, we consider the group comparisons robust because spectra were acquired under identical experimental conditions and quality metrics are transparently reported.
Another technical limitation concerns the possibility that the PFC voxel, although carefully positioned to avoid ventricular spaces, may include a small fraction of CSF within the interhemispheric fissure. In principle, uncorrected CSF inclusion could lead to a partial underestimation of absolute metabolite concentrations. However, a systematic CSF-related dilution would be expected to affect all metabolites proportionally, which is not consistent with the heterogeneous pattern observed in the present study. Therefore, while this factor should be acknowledged, it is unlikely to materially affect the interpretation of the group comparisons.
A further limitation concerns the experimental design. We did not include unstressed WKY rats or stressed Wistar controls, which would help disentangle strain-related from stress-induced neurobiological differences and clarify whether the observed alterations are specific to the treatment-resistant phenotype. However, our study followed the validated WKY/CMS model for treatment-resistant depression, which combines genetic and environmental vulnerability, and was designed to minimize animal use in accordance with ethical and 3Rs principles. Future studies including these additional groups will be valuable to further refine this interpretation.
Conclusions
Our multimodal MRI findings indicate widespread metabolic and structural alterations in WKY rats exposed to CMS. By combining structural MRI, DTI and quantitative MRS, we identified microstructural and neurochemical features associated with the depressive-like phenotype. Notably, this study extends the characterization of the model to quantitative MRS and DTI—previously unexplored in this context—and establishes imaging readouts that can be used as translationally relevant quantitative endpoints in future mechanistic and intervention studies.
The WKY/CMS model captures core features of stress susceptibility and impaired responsivity that are relevant to treatment resistance, and may therefore provide a useful platform for investigating novel therapeutic approaches, including neuromodulation (rTMS) and psychedelic-based interventions. Against this backdrop, the prevailing reliance on antidepressant-sensitive models may limit predictive value for difficult-to-treat populations75. Accordingly, evaluating candidate interventions in models with TRD-relevant features such as WKY/CMS, and monitoring changes with quantitative MRS/DTI readouts, may help strengthen translational alignment and inform the development of more targeted treatments.
Methods
Animals and experimental design
Animals were housed in a temperature-controlled room (21–23 °C, humidity 40–50%), on a 12-h reversed light–dark cycle (lights on at 8:00 p.m.). Breeders were provided by Charles River Laboratories (Lecco, Italy). Pregnant dams were singly housed in cages (48.2 × 26.7 × 21.0 cm; Tecniplast, Varese, Italy) with sawdust bedding, ad libitum access to tap water and food (breeding diet 1314; Altromin, Lage, Germany) and environmental enrichment (cardboard tubes, 80 × 150 × 2 mm; Caipet, Foggia, Italy).
After weaning (on average 1 and 2 subjects from each W and WKY litter respectively), 12 male Wistar (W) rats and 12 male Wistar-Kyoto (WKY) were housed in pairs in cages (42.5 × 26.6 × 18.5 cm; Tecniplast, Varese, Italy) with sawdust bedding and environmental enrichment (poplar sticks, 100 × 20 × 20 mm; Caipet, Foggia-IT). Experimental animals had ad libitum access to tap water and food (maintenance diet 4RF21; Mucedola, Milan, Italy), unless otherwise specified.
The study design followed the validated Wistar-Kyoto + chronic mild stress (WKY/CMS) framework, a translational model of treatment-resistant depression (TRD) that combines genetic vulnerability and environmental stress exposure. The WKY/CMS group thus represents a treatment-resistant depressive phenotype, whereas the non-stressed Wistar (W/CTRL) group serves as the healthy control reference. This targeted comparison was selected to model the TRD-versus-healthy control contrast, consistent with the translational scope of the study, while adhering to the 3Rs principle (Reduction, Refinement, Replacement).
CMS paradigm
At 9 weeks of age, WKY were singly housed whilst W rats were left in pairs. At 10 weeks of age, WKY rats (bodyweight 272 g) were exposed to a CMS paradigm for 4 consecutive weeks19,25,26,109,110 whereas W rats (body weight 454 g) were left undisturbed, in pairs, in their home-cages (W/CTRL).
The paradigm consisted in the application of seven different mild stressors (cage tilting at 45°, food deprivation, water deprivation, pair housing with non-siblings changing partner every time, soiled bedding with 400 ml of water for 1.6 L of sawdust, alternating light and darkness every 2 h, stroboscopic light with 120–150 flashes/min), each applied for a period of 10 to 14 h. Each week included 3 periods per week of stress relief consisting in leaving the animals undisturbed in their cages for 10–14 h. Stressors were applied continuously (throughout the day and night), and randomly (changing the weekly sequence to make them more unpredictable for the animals). All animals were weighed weekly.
WKY/CMS and W/CTRL rats underwent MRI at 14 weeks of age.
Magnetic resonance imaging (MRI)
We studied structural alteration by using high resolution T2-weighted turboRARE MRI and axial multi-slices diffusion-weighted images (DTI) in the PFC, Hip and corpus callosum (WKY/CMS: n = 12; W/CTRL: n = 12). MRI experiments were performed using a preclinical 7.0 T system (Pharmascan 70/16 US, Bruker). In addition, we investigated changes in brain metabolism through in vivo quantitative MRS in the PFC and Hip (WKY/CMS: n = 8; W/CTRL: n = 8).
Rats were anesthetized with inhaled isoflurane (2.5% in O2, 2 L/min for induction, 2.0% in O2, 1 L/m for maintenance) and positioned on an animal cradle with mouth and ear bars for head fixation during the experiments. Body temperature was maintained constant via an integrated heating system. An MRI-compatible pulse oximeter (MouseOx, Starr Life Sciences Corp), was placed on the right posterior leg for continuous monitoring of the heart rate, breath rate, oxygen saturation (pO2) and pulse distension (a surrogate parameter for blood pressure) during the MRI session.
After tuning and shimming adjustments, T2-weighted scans were performed in the three orientations (axial, sagittal, coronal) to visualize the geometry (repetition time/echo time (TR/TE) = 2500/33 ms, matrix = 256 × 256, field of view (FOV) = 25 × 25 mm2, slice thickness = 1 mm, multi-contiguous slices in order to cover the entire brain).
Anatomical MRI
Anatomic alterations were investigated by using a T2-weighted MRI (TR/TE = 3463/33 ms, matrix 192 × 192, FOV = 25 × 25 mm2, thickness = 0.6 mm, 1 average). Thickness measurements were restricted to two regions—the cingulate cortex and Hip—where anatomical boundaries could be reliably identified based on MRI contrast. Because only these predefined ROIs were quantitatively analysed, no additional correction for multiple comparisons was applied.
Magnetic resonance spectroscopy (MRS)
We adopted a quantitative protocol which includes a semi-LASER sequence, unsuppressed T2-corrected water signal as reference, assuming a water content of 80%111 and LCModel fitting analysis112,113. The semi-LASER sequence was chosen to minimize the chemical shift displacement errors114. The repetition time/echo time (TR/TE) = 4000/20.6 ms of the metabolite (water suppressed) spectrum were chosen to minimize the loss of signal due to T1 and T2 relaxation times of the water and metabolites; 256 averages per spectrum were acquired to have a signal-to-noise higher than 10. The unsuppressed water signal was acquired with the same sequence, the same voxel and parameters except for TR = 15,000ms and 1 single scan.
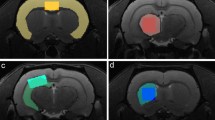
Magnetic resonance spectra were collected from 2 key regions implicated in MDD: the PFC and the Hip, as shown in Fig. 6. Because of the differences in the shape of cortical and subcortical brain areas between WKY and W strains, voxel sizes were slightly adjusted to cover the same functional regions (see details in the figure legend). Multi-slice multi-echo T2-weighted MRI (TR/TE = 2476/7.5 (× 14) ms, matrix 128 × 128, FOV = 25 × 25 mm2, 2 averages) were acquired to measure water T2 for metabolite quantification.
Positioning of the voxels for MRS. Coronal, axial, and sagittal T2W MRI (TR/TE = 2500/33 ms) indicating the voxel positioned in the PFC (upper row; voxel size 6 × 3 × 1.8 = 32.4 mm3 and 6 × 2.85 × 1.8 = 30.8 mm3 in W/CTRL and WKY/CMS brains respectively) and in the right Hip (lower row; voxel size 3.8 × 2.5 × 1.8 = 17.1 mm3 and 3.8 × 2.37 × 1.71 = 15.4 mm3 in W/CTRL and WKY/CMS brains respectively).
The basis set used for spectral analysis by the LCModel fitting routine included all metabolites, lipids, and macromolecules typically present in the standard brain basis set. Specifically, it comprised alanine (Ala), aspartate (Asp), creatine (Cr), phosphocreatine (PCr), γ-aminobutyric acid (GABA), glucose (Glc), glutamine (Gln), glutamate (Glu), glycerophosphocholine (GPC), phosphocholine (PCho), glutathione (GSH), myo-inositol (Ins), lactate (Lac), N-acetylaspartate (NAA), N-acetylaspartylglutamate (NAAG), scyllo-inositol, taurine (Tau), lipid signals at 1.3 ppm (Lip13a and Lip13b), lipid signal at 0.9 ppm (Lip09), macromolecule signal at 0.9 ppm (MM09), lipid signal at 2.0 ppm (Lip20), and macromolecule signals at 1.2 ppm (MM12), 1.4 ppm (MM14), 1.7 ppm (MM17), and 2.0 ppm (MM20). For metabolites with strongly overlapping signals, such as PCho and GPC or NAA and NAAG, LCModel also provided their summed concentrations (denoted as tCho and tNAA). We used the PRESS basis set (appropriate for spin echo sequences such as semi LASER) with TE = 20 ms.
MRS data were acquired from a subset of animals (n = 8 per group) according to the original experimental design, which included separate imaging sessions for structural and metabolic measurements. The reduced sample size for MRS reflects the longer acquisition time and complexity of this protocol, while still ensuring sufficient statistical power for quantitative analyses.
Diffusion tensor imaging (DTI)
DTI provides information about the microarchitecture of white and grey matter, by quantifying MD and FA. Structural differences between the two animal models were studied by axial multi-slices DTI obtained using a 4-segment echo-planar encoding sequence with following parameters: TR/TE = 2000/18.8 ms, matrix = 128 × 128, FOV = 25 × 25 mm2, slice thickness = 1 mm, one average.
Stejskal-Tanner diffusion gradients were applied along 30 spatial directions (b-value = 0, 1200 s/mm2, diffusion gradient pulse duration (δ) = 2.5 ms and diffusion gradient separation (Δ) 8.4 ms). From tensor analysis (Paravision 360, Bruker), we extrapolated FA and MD in three brain regions (PFC, Hip and corpus callosum), employing manual delineation.
Statistical analysis
Metabolites, macromolecules, and lipid signals that did not meet the predefined inclusion criteria were excluded. Specifically, Ala, Asp, GABA, Scyllo-Ins, and all lipid and macromolecular signals—except for MM14 + Lip13, MM20, and MM09—did not satisfy the inclusion criteria. The macromolecule at 2.1 ppm was excluded due to its poor reliability, as the strong overlap with other resonances compromised robust quantification.
Statistical analysis was performed using GraphPad Prism® 10 (version 10.3.1, GraphPad Software Inc., San Diego, CA, USA) and Metaboanalyst version 6.0115 for metabolite analyses. In particular, MRS matrix was pareto-scaled (mean-centred and divided by the square root of the standard deviation of each variable) and analysed using univariate methods (Wilcoxon rank-sum test corrected for multiple comparison by false discovery rate [FDR] analyses) and multivariate analysis (PLS-DA). FDR correction was performed using the Benjamini–Hochberg (BH) procedure as implemented in MetaboAnalyst. For the BH False Discovery Rate correction, the total number of variables (m) was set to 11, corresponding to all metabolites and macromolecular signals included in the univariate analyses (those with CRLB ≤ 20%, except for Cr and GSH in the Hip of WKY/CMS rats, CRLB ≤ 22%, and Gln in the Hip of WKY/CMS rats, CRLB ≤ 25%). Group-level CRLBs (mean ± SD) for all metabolites and macromolecules signals in each region are reported in Supplementary Table 3. GABA was excluded from both regions because its CRLB exceeded 20% in all spectra. For group comparisons, Wilcoxon rank-sum (Mann–Whitney U) tests were performed using MetaboAnalyst. Non-parametric tests were selected due to the small sample size and because the data did not meet the assumption of normal distribution. The resulting test statistic is reported as V, corresponding to the standard Wilcoxon rank-sum statistic. A five-fold internal cross-validation was used to validate the performance of the PLS-DA models, determining the number of principal components (PC) and assessing the model’s accuracy (Acc), sum of squares (R2) and predictive ability (Q2) of the model. For data interpretation, PLS-DA loading plots were generated, and relevant resonances identified with VIP greater than 1.0 were considered. Each data point corresponds to an individual rat from each experimental group. For parameters derived from DTI and cortical and hippocampal thickness we adopt a Wilcoxon rank-sum test. Statistical significance was set at p < 0.05. Data are presented as mean ± SE.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Willner, P. The chronic mild stress (CMS) model of depression: History, evaluation and usage. Neurobiol. Stress 6, 78–93 (2017).
Hollis, F. & Kabbaj, M. Social defeat as an animal model for depression. ILAR J. 55, 221–232 (2014).
Rygula, R. et al. Citalopram counteracts depressive-like symptoms evoked by chronic social stress in rats. Behav. Pharmacol. 17, 19–29 (2006).
Vialou, V. et al. DeltaFosB in brain reward circuits mediates resilience to stress and antidepressant responses. Nat Neurosci 13, 745–752 (2010).
Venzala, E., García-García, A. L., Elizalde, N., Delagrange, P. & Tordera, R. M. Chronic social defeat stress model: Behavioral features, antidepressant action, and interaction with biological risk factors. Psychopharmacology 224, 313–325 (2012).
Iijima, M., Ito, A., Kurosu, S. & Chaki, S. Pharmacological characterization of repeated corticosterone injection-induced depression model in rats. Brain Res. 1359, 75–80 (2010).
Rainer, Q. et al. Beneficial behavioural and neurogenic effects of agomelatine in a model of depression/anxiety. Int. J. Neuropsychopharmacol. 15, 321–335 (2012).
Sallie, F. N. et al. Neurobehavioral and molecular changes in a rodent model of ACTH-induced HPA axis dysfunction. Brain Res. https://doi.org/10.1016/j.brainres.2024.148913 (2024).
Zoratto, F. et al. Consumption of the prebiotic-rich chicory taproot contrasts the cognitive and motivational consequences of chronic corticosterone exposure and modulates gut microbiota composition in mice. Biol. Psychiatry Glob. Open Sci. https://doi.org/10.1016/j.bpsgos.2025.100520 (2025).
Wang, D. et al. Behavioural and neurochemical features of olfactory bulbectomized rats resembling depression with comorbid anxiety. Behav. Brain Res. 178, 262–273 (2007).
Ho, Y. J., Chen, K. H., Tai, M. Y. & Tsai, Y. F. MK-801 suppresses muricidal behavior but not locomotion in olfactory bulbectomized rats: Involvement of NMDA receptors. Pharmacol. Biochem. Behav. 77, 641–646 (2004).
Okamoto, K. & Aoki, K. Development of a strain of spontaneously hypertensive rats. Jpn. Circ. J. 27, 282–293 (1963).
Nam, H., Clinton, S. M., Jackson, N. L. & Kerman, I. A. Learned helplessness and social avoidance in the Wistar-Kyoto rat. Front. Behav. Neurosci. 8, 109 (2014).
Part~, W. P. Open field, learned helplessness, conditioned defensive burying, and forced-swim lests in WKY rats. Physiol. Behav. 55(33), 433–439 (1994).
Will, C. C., Aird, F. & Redei, E. E. Selectively bred Wistar-Kyoto rats: An animal model of depression and hyper-responsiveness to antidepressants. Mol. Psychiatry 8, 925–932 (2003).
Aleksandrova, L. R., Wang, Y. T. & Phillips, A. G. Evaluation of the Wistar-Kyoto rat model of depression and the role of synaptic plasticity in depression and antidepressant response. Neurosci. Biobehav. Rev. 105, 1–23 (2019).
Gormley, S., Rouine, J., McIntosh, A., Kerskens, C. & Harkin, A. Glial fibrillary acidic protein (GFAP) immunoreactivity correlates with cortical perfusion parameters determined by bolus tracking arterial spin labelling (bt-ASL) magnetic resonance (MR) imaging in the Wistar Kyoto rat. Physiol. Behav. 160, 66–79 (2016).
Cominski, T. P., Jiao, X., Catuzzi, J. E., Stewart, A. L. & Pang, K. C. H. The role of the hippocampus in avoidance learning and anxiety vulnerability. Front. Behav. Neurosci. 8, 273 (2014).
Papp, M. & Willner, P. Models of affective illness: Chronic mild stress in the rat. Curr. Protoc. https://doi.org/10.1002/cpz1.712 (2023).
Strekalova, T. et al. Chronic mild stress paradigm as a rat model of depression: Facts, artifacts, and future perspectives. Psychopharmacology 239, 663–693 (2022).
Paré, W. P. & Redei, E. Depressive behavior and stress ulcer in Wistar Kyoto rats. J. Physiol. (Paris) 87, 229–238 (1993).
Redei, E., Pare, W. P., Aird, F. & Kluczynski, J. Strain differences in hypothalamic-pituitary-adrenal activity and stress ulcer. Am. J. Physiol. https://doi.org/10.1152/ajpregu.1994.266.2.R353 (1994).
Solberg, L. C., Olson, S. L., Turek, F. W. & Redei, E. Altered Hormone Levels and Circadian Rhythm of Activity in the WKY Rat, a Putative Animal Model of Depression Experimental Protocol. vol. 47 www.ajpregu.org.
Loizeau, V. et al. Behavioural characteristics and sex differences of a treatment-resistant depression model: Chronic mild stress in the Wistar-Kyoto rat. Behav. Brain Res. https://doi.org/10.1016/j.bbr.2023.114712 (2024).
Willner, P. & Belzung, C. Treatment-resistant depression: Are animal models of depression fit for purpose?. Psychopharmacology 232, 3473–3495 (2015).
Willner, P. et al. Validation of chronic mild stress in the Wistar-Kyoto rat as an animal model of treatment-resistant depression. Behav. Pharmacol. 30, 239–250 (2019).
Hashimoto, K. Rapid-acting antidepressant ketamine, its metabolites and other candidates: A historical overview and future perspective. Psychiatry Clin. Neurosci. 73, 613–627 (2019).
Berlim, M. T., Van Den Eynde, F., Tovar-Perdomo, S. & Daskalakis, Z. J. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: A systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol. Med. 44, 225–239 (2014).
Pettorruso, M. et al. Comparing fast-acting interventions for treatment-resistant depression: An explorative study of accelerated HF-rTMS versus intranasal esketamine. Brain Stimul 16, 1041–1043 (2023).
Weiner, R. D. & Reti, I. M. Key updates in the clinical application of electroconvulsive therapy. Int. Rev. Psychiatry 29, 54–62 (2017).
Mayberg, H. S. et al. Deep brain stimulation for treatment-resistant depression. Neuron 45, 651–660 (2005).
Holtzheimer, P. E. et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: a multisite, randomised, sham-controlled trial. Lancet Psychiatry 4, 839–849 (2017).
Bergfeld, I. O. et al. Deep brain stimulation of the ventral anterior limb of the internal capsule for treatment-resistant depression: A randomized clinical trial. JAMA Psychiatry 73, 456–464 (2016).
Falowski, S. M. et al. An evaluation of neuroplasticity and behavior after deep brain stimulation of the nucleus accumbens in an animal model of depression. Neurosurgery 69, 1281–1290 (2011).
Kyeremanteng, C. et al. Effects of electroconvulsive seizures on depression-related behavior, memory and neurochemical changes in Wistar and Wistar-Kyoto rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 54, 170–178 (2014).
Tizabi, Y., Bhatti, B. H., Manaye, K. F., Das, J. R. & Akinfiresoye, L. Antidepressant-like effects of low ketamine dose is associated with increased hippocampal AMPA/NMDA receptor density ratio in female Wistar-Kyoto rats. Neuroscience 213, 72–80 (2012).
Cerri, C. et al. Timing matters: Inverted U-shaped efficacy of dose distribution in translational neuromodulation for treatment-resistant depression. Brain Stimul. 19, 102976 (2025).
Redei, E. E., Solberg, L. C. & Kluczynski, J. M. Paradoxical hormonal and behavioral responses to hypothyroid and hyperthyroid states in the Wistar-Kyoto rat. Neuropsychopharmacology https://doi.org/10.1016/S0893-133X(00)00229-3 (2001).
Ne Durand, M. et al. Strain-dependent neurochemical and neuroendocrine effects of Desipramine, but not Fluoxetine or Imipramine, in Spontaneously Hypertensive and Wistar-Kyoto rats. Neuropharmacology https://doi.org/10.1016/S0028-3908(00)00088-5 (2000).
Cusick, J. A., Wellman, C. L. & Demas, G. E. Maternal stress and the maternal microbiome have sex-specific effects on offspring development and aggressive behavior in Siberian hamsters (Phodopus sungorus). Horm. Behav. https://doi.org/10.1016/j.yhbeh.2022.105146 (2022).
Rittenhouse, P. A., López-Rubalcava, C., Stanwood, G. D. & Lucki, I. Amplified behavioral and endocrine responses to forced swim stress in the Wistar-Kyoto rat. Psychoneuroendocrinology https://doi.org/10.1016/S0306-4530(01)00052-X (2002).
De La Garza, R. & Mahoney, J. J. A distinct neurochemical profile in WKY rats at baseline and in response to acute stress: Implications for animal models of anxiety and depression. Brain Res. 1021, 209–218 (2004).
Malkesman, O. & Weller, A. Two different putative genetic animal models of childhood depression-a review. Prog. Neurobiol. 88, 153–169 (2009).
Villanueva, R. Neurobiology of major depressive disorder. Neural Plast. https://doi.org/10.1155/2013/873278 (2013).
Scholl, J. L., Renner, K. J., Forster, G. L. & Tejani-Butt, S. Central monoamine levels differ between rat strains used in studies of depressive behavior. Brain Res. 1355, 41–51 (2010).
Jiao, X., Paré, W. P. & Tejani-Butt, S. Strain differences in the distribution of dopamine transporter sites in rat brain. Prog. Neuropsychopharmacol. Biol. Psychiatry 27, 913–919 (2003).
Jiao, X., Paré, W. P. & Tejani-Butt, S. M. Antidepressant drug induced alterations in binding to central dopamine transporter sites in the Wistar Kyoto rat strain. Prog. Neuropsychopharmacol. Biol. Psychiatry 30, 30–41 (2006).
Novick, A., Yaroslavsky, I. & Tejani-Butt, S. Strain differences in the expression of dopamine D1 receptors in Wistar-Kyoto (WKY) and Wistar rats. Life Sci. 83, 74–78 (2008).
Guan, J. et al. Early life stress increases brain glutamate and induces neurobehavioral manifestations in rats. ACS Chem. Neurosci. 11, 4169–4178 (2020).
Kaadt, E., Hedemann, N. K., Damgaard, C. K., Müller, H. K. & Elfving, B. From microRNA to protein, linking the neurotrophic hypothesis of depression to the Wistar-Kyoto rat. Neurosci. Appl. https://doi.org/10.1016/j.nsa.2023.101131 (2023).
Kin, K. et al. Hippocampal neurogenesis of Wistar Kyoto rats is congenitally impaired and correlated with stress resistance. Behav. Brain Res. 329, 148–156 (2017).
Filatova, E. V., Shadrina, M. I. & Slominsky, P. A. Major depression: One brain, one disease, one set of intertwined processes. Cells 10, 1283 (2021).
Karege, F. et al. Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res. https://doi.org/10.1016/s0165-1781(02)00005-7 (2002).
Pandey, G. N. et al. Brain-derived neurotrophic factor gene and protein expression in pediatric and adult depressed subjects. Prog. Neuropsychopharmacol. Biol. Psychiatry 34, 645–651 (2010).
Nagasawa, M., Otsuka, T., Yasuo, S. & Furuse, M. Chronic imipramine treatment differentially alters the brain and plasma amino acid metabolism in Wistar and Wistar Kyoto rats. Eur. J. Pharmacol. 762, 127–135 (2015).
Li, Q. et al. Partial resistance to citalopram in a Wistar-Kyoto rat model of depression: An evaluation using resting-state functional MRI and graph analysis. J. Psychiatr. Res. 151, 242–251 (2022).
Zou, Q. H. et al. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: Fractional ALFF. J Neurosci Methods 172, 137–141 (2008).
Di, X. et al. The influence of the amplitude of low-frequency fluctuations on resting-state functional connectivity. Front. Hum. Neurosci. 7, 118 (2013).
Dingess, P. M. et al. Structural and functional plasticity within the nucleus accumbens and prefrontal cortex associated with time-dependent increases in food cue-seeking behavior. Neuropsychopharmacology 42, 2354–2364 (2017).
Yin, S. et al. Intervention-induced enhancement in intrinsic brain activity in healthy older adults. Sci. Rep. https://doi.org/10.1038/srep07309 (2014).
Tao, J. et al. Tai Chi Chuan and Baduanjin mind-body training changes resting-state low-frequency fluctuations in the frontal lobe of older adults: A resting-state fMRI study. Front. Hum. Neurosci. 11, 1–10 (2017).
Lan, M. J. et al. Resting-state amplitude of low-frequency fluctuation is associated with suicidal ideation. Depress. Anxiety 36, 433–441 (2019).
Frodl, T. et al. Hippocampal changes in patients with a first episode of major depression. Am. J. Psychiatry https://doi.org/10.1176/appi.ajp.159.7.1112 (2002).
Tura, A. & Goya-Maldonado, R. Brain connectivity in major depressive disorder: A precision component of treatment modalities?. Transl. Psychiatry 13, 196 (2023).
Hou, X. et al. Neural activity in adults with major depressive disorder differs from that in healthy individuals: A resting-state functional magnetic resonance imaging study. Front. Psychiatry https://doi.org/10.3389/fpsyt.2022.1028518 (2022).
Tomasi, D. & Volkow, N. D. Functional connectivity hubs in the human brain. Neuroimage 57, 908–917 (2011).
Kropf, E., Syan, S. K., Minuzzi, L. & Frey, B. N. From anatomy to function: The role of the somatosensory cortex in emotional regulation. Braz. J. Psychiatry 41, 261–269 (2019).
Yoshii, T. et al. Validation of Wistar-Kyoto rats kept in solitary housing as an animal model for depression using voxel-based morphometry. Sci. Rep. https://doi.org/10.1038/s41598-024-53103-2 (2024).
Wise, T. et al. Common and distinct patterns of grey-matter volume alteration in major depression and bipolar disorder: Evidence from voxel-based meta-analysis. Mol. Psychiatry 22, 1455–1463 (2017).
Arnone, D. et al. Computational meta-analysis of statistical parametric maps in major depression. Hum. Brain Mapp. 37, 1393–1404 (2016).
Lin, A. et al. Minimum reporting standards for in vivo magnetic resonance spectroscopy (MRSinMRS): Experts’ consensus recommendations. NMR Biomed. 34(5), e4484 (2021).
Zhao, Y. J. et al. Brain grey matter abnormalities in medication-free patients with major depressive disorder: A meta-analysis. Psychol. Med. 44, 2927–2937 (2014).
Kang, Y. et al. The effect of inflammation markers on cortical thinning in major depressive disorder: A possible mediator of depression and cortical changes. J. Affect. Disord. 348, 229–237 (2024).
Chen, J. et al. Causal structural covariance network identifies progressive gray matter atrophy in adolescents with major depressive disorder. Psychol Med 55, e249 (2012).
Zhang, Y. et al. Revealing complexity: segmentation of hippocampal subfields in adolescents with major depressive disorder reveals specific links to cognitive dysfunctions. Eur. Psychiatry 68(1), e5 (2025).
Moriguchi, S. et al. Glutamatergic neurometabolite levels in major depressive disorder: A systematic review and meta-analysis of proton magnetic resonance spectroscopy studies. Mol. Psychiatry 24, 952–964 (2019).
Hemanth Kumar, B. S., Mishra, S. K., Rana, P., Singh, S. & Khushu, S. Neurodegenerative evidences during early onset of depression in CMS rats as detected by proton magnetic resonance spectroscopy at 7 T. Behav. Brain Res. 232, 53–59 (2012).
Cherix, A. et al. Metabolic signature in nucleus accumbens for anti-depressant-like effects of acetyl-L-carnitine. eLife 9, e50631 (2020).
Pavlova, I. & Ruda-Kucerova, J. Brain metabolic derangements examined using 1H MRS and their (in)consistency among different rodent models of depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 127, 110808 (2023).
Schür, R. R. et al. Brain GABA levels across psychiatric disorders: A systematic literature review and meta-analysis of (1) H-MRS studies. Hum. Brain Mapp. 37, 3337–3352 (2016).
Lebel, C. et al. Prepartum and postpartum maternal depressive symptoms are related to children’s brain structure in preschool. Biol. Psychiatry 80, 859–868 (2016).
Harris, A. D. & MacMillan, E. L. MRS in neuroinflammation. In Advances in Magnetic Resonance Technology and Applications Vol. Vol. 9 (eds Laule, C. & D., P. J.) 79–116 (Academic Press, 2023).
Jung, C. et al. Magnetic resonance imaging of neuroinflammation in chronic pain: A role for astrogliosis?. Pain 161, 1555–1564 (2020).
Patel, N. C., Cecil, K. M., Strakowski, S. M., Adler, C. M. & DelBello, M. P. Neurochemical alterations in adolescent bipolar depression: A proton magnetic resonance spectroscopy pilot study of the prefrontal cortex. J. Child Adolesc. Psychopharmacol. 18, 623–627 (2008).
Drago, T. et al. A comprehensive regional neurochemical theory in depression: A protocol for the systematic review and meta-analysis of 1H-MRS studies in major depressive disorder. Syst. Rev. 7(1), 158 (2018).
Lirng, J. F. et al. Increased myo-inositol level in dorsolateral prefrontal cortex in migraine patients with major depression. Cephalalgia 35, 702–709 (2015).
Kumar, A. et al. Frontal white matter biochemical abnormalities in late-life major depression detected with proton magnetic resonance spectroscopy. Am. J. Psychiatry https://doi.org/10.1176/appi.ajp.159.4.630 (2002).
Kim, S. Y. et al. Reversal of myo-inositol metabolic level in the left dorsolateral prefrontal cortex of rats exposed to forced swimming test following desipramine treatment: An in vivo localized (1)H-MRS study at 4.7 T. Magn. Reson. Imaging 28, 1461–1467 (2010).
Li, C. X. et al. Cerebral metabolic changes in a depression-like rat model of chronic forced swimming studied by ex vivo high resolution 1H magnetic resonance spectroscopy. Neurochem. Res. 33, 2342–2349 (2008).
Kim, S., Hong, K. B., Kim, S., Suh, H. J. & Jo, K. Creatine and taurine mixtures alleviate depressive-like behaviour in Drosophila melanogaster and mice via regulating Akt and ERK/BDNF pathways. Sci. Rep. 10(1), 11370 (2020).
Rajkowska, G. & Stockmeier, C. Astrocyte pathology in major depressive disorder: Insights from human postmortem brain tissue. Curr. Drug Targets. 14, 1225–1236 (2013).
Mechawar, N. & Savitz, J. Neuropathology of mood disorders: Do we see the stigmata of inflammation?. Transl. Psychiatry. https://doi.org/10.1038/tp.2016.212 (2016).
Gibbons, A. S., Brooks, L., Scarr, E. & Dean, B. AMPA receptor expression is increased post-mortem samples of the anterior cingulate from subjects with major depressive disorder. J. Affect. Disord. 136, 1232–1237 (2012).
Ramírez-Guerrero, S. et al. Taurine and astrocytes: A homeostatic and neuroprotective relationship. Front. Mol. Neurosci. https://doi.org/10.3389/fnmol.2022.937789 (2022).
Zhu, Y. et al. Taurine Alleviates Chronic Social Defeat Stress-Induced Depression by Protecting Cortical Neurons from Dendritic Spine Loss. Cell Mol Neurobiol 43, 827–840 (2023).
Zhou, H., Gao, Y., Dong, T., Wang, J. & Liu, Z. F. Magnetic resonance spectroscopy studies in children and adolescents with depression: A systematic review and meta-analysis. J. Affect. Disord. 389, 119747 (2025).
Rae, C. D. A guide to the metabolic pathways and function of metabolites observed in human brain 1H magnetic resonance spectra. Neurochem. Res. 39, 1–36 (2014).
Kumar, A. et al. Frontal white matter biochemical abnormalities in late-life major depression detected with proton magnetic resonance spectroscopy. Am. J. Psychiatry. 159, 630–636 (2002).
Xie, X. et al. Altered neurometabolite levels in the brains of patients with depression: A systematic analysis of magnetic resonance spectroscopy studies. J. Affect. Disord. 328, 95–102 (2023).
Ebeid, M. A. et al. Cognitive effects of the GSK-3 inhibitor “lithium” in LPS/chronic mild stress rat model of depression: Hippocampal and cortical neuroinflammation and tauopathy. Neurotoxicology 83, 77–88 (2021).
Cudalbu, C. et al. Contribution of macromolecules to brain 1H MR spectra: Experts’ consensus recommendations. NMR Biomed. 34, e4393 (2021).
Simicic, D. et al. In vivo macromolecule signals in rat brain 1 H-MR spectra at 9.4T: Parametrization, spline baseline estimation, and T2 relaxation times. Magn. Reson. Med. 86, 2384–2401 (2021).
Birch, R., Peet, A. C., Dehghani, H. & Wilson, M. Influence of macromolecule baseline on 1 H MR spectroscopic imaging reproducibility. Magn. Reson. Med. 77, 34–43 (2017).
Genovese, G. et al. Age-related differences in macromolecular resonances observed in ultra-short-TE STEAM MR spectra at 7T. Magn. Reson. Med. 92, 4–14 (2024).
Guo, W. B. et al. Altered white matter integrity of forebrain in treatment-resistant depression: A diffusion tensor imaging study with tract-based spatial statistics. Prog. Neuropsychopharmacol. Biol. Psychiatry 38, 201–206 (2012).
White, J. D. et al. Early life stress causes sex-specific changes in adult fronto-limbic connectivity that differentially drive learning. Elife 9, 1–29 (2020).
Klok, M. P. C., van Eijndhoven, P., Argyelan, M., Schene, A. H. & Tendolkar, I. Structural brain characteristics in treatment-resistant depression: Review of magnetic resonance imaging studies. BJPsych Open. 5(4), 76 (2019).
Xu, Y. et al. Shared and distinct white matter alterations in major depression and bipolar disorder: A systematic review and meta-analysis. J. Integr. Neurosci. https://doi.org/10.31083/j.jin2309170 (2024).
Papp, M. Models of affective illness: Chronic mild stress in the rat. Curr. Protoc. Pharmacol. 5(1), 5–9 (2012).
Papp, M. et al. Rapid antidepressant effects of deep brain stimulation of the pre-frontal cortex in an animal model of treatment-resistant depression. J. Psychopharmacol. 32, 1133–1140 (2018).
Gottschalk, A., Scafidi, S. & Toung, T. J. K. Brain water as a function of age and weight in normal rats. PLoS One. https://doi.org/10.1371/journal.pone.0249384 (2021).
Canese, R. et al. Characterisation of in vivo ovarian cancer models by quantitative 1H magnetic resonance spectroscopy and diffusion-weighted imaging. NMR Biomed. 25, 632–642 (2012).
Provencher, S. W. In Vivo Proton NMR Spectra. (1993).
Öz, G. et al. Advanced single voxel 1H magnetic resonance spectroscopy techniques in humans: Experts’ consensus recommendations. NMR Biomed. 34, e4236 (2020).
Pang, Z. et al. MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 49, W388–W396 (2021).
Acknowledgements
The authors wish to thank Luigia Cancemi, Andrea Martinelli, Paolo Frassinito and Flavio Torriani for their valuable assistance with animal care.
Funding
This work was funded by the European Union – NextGenerationEU – within the Italian National Recovery and Resilience Plan – PNRR M6C2 – Investment 2.1 “Valorisation and strengthening of biomedical research in the National Health Service” (project code PNRR-MCNT2-2023–12377068 to FZ, RC, and LDR); and by the Italian Ministry of Health (Ricerca Finalizzata – Young Researchers, project code GR-2019–12370173 to MP, LDR, and FZ).
Author information
Authors and Affiliations
Contributions
FZ, RC, MP, and LDR conceived and supervised the study. RC and FZ designed the experiments. MB, and CC conducted CMS procedures and animal work; GP, VZ, and TS acquired and processed the MRI/DTI data and the 1 H-MRS data; RC performed the statistical analyses. MP, LDR, and FZ acquired funding. RC, MB, and CC drafted the manuscript; FZ, MP, and LDR provided critical revisions. All authors discussed the results, approved the final version, and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Approval for animal experiments
All procedures were approved by the Italian Ministry of Health (license n. 1150/2024-PR to FZ) and performed in accordance with the Directive 2010/63/EU on the protection of animals use for scientific purposes and Italian law (Legislative Decree 26/2014). This study complies with the ARRIVE guidelines for reporting animal research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gianmauro, P., Valentina, Z., Marta, B. et al. Neuroimaging insights from Wistar-Kyoto rats under chronic mild stress: morphological and metabolic brain correlates of treatment-resistant depression. Sci Rep 16, 10868 (2026). https://doi.org/10.1038/s41598-026-45121-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-45121-z