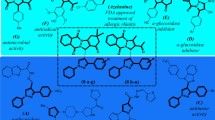
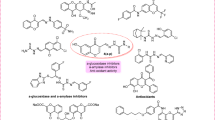
Abstract
Syzygium aromaticum (L.) Merr. & L.M. Perry is a known spice with a high phytochemical content that can be explored in drug discovery. We investigated the in vitro enzyme inhibitory activities of a flavonoid-rich extract of S. aromaticum (FRESA) against type II diabetes (T2D) and Alzheimer’s disease (AD) and identified its anti-T2D and anti-AD phytochemicals via computational prediction. The in vitro enzyme inhibitory activities of a flavonoid-rich extract of Syzygium aromaticum were evaluated via standard protocols following flavonoid-enriched extraction procedures. High-performance liquid chromatography (HPLC) was employed to characterize the constituent bioactive flavonoids. Molecular docking of eight phytochemicals was performed via AutoDock Vina in PyRx 0.8, which identified apigenin, myricetin, and quercetin as hit compounds with high binding affinities and multitarget activities against α-amylase, α-glucosidase, acetylcholinesterase (AChE), butyrylcholinesterase (BChE), and monoamine oxidase (MAO). Molecular dynamics simulations (100 ns) were conducted via GROMACS 2019.2, and binding free energy calculations were performed via the MM-GBSA approach to validate the stability and interaction integrity of the hit phytochemicals. FRESA (IC50 = 961.943 ± 21.031 μg/mL) exhibited moderate activity against α-amylase compared with that of acarbose (IC50 = 27.104 ± 0.270 μg/mL). Compared with acarbose (IC50 = 17.389 ± 0.436 μg/mL), FRESA had appreciable activity against α-glucosidase (IC50 = 562.045 ± 6.714 μg/mL). FRESA demonstrated significant (p < 0.0001) inhibition of acetylcholinesterase (IC50 = 26.911 ± 0.058 µg/mL), surpassed galantamine (IC50 = 27.950 ± 0.122 µg/mL), and moderately inhibited butyrylcholinesterase (IC50 = 28.168 ± 0.702 µg/mL) to galantamine (IC50 = 23.126 ± 0.683 µg/mL). FRESA also significantly suppressed monoamine oxidase activity in Fe2⁺-induced brain damage in a concentration-dependent manner. HPLC–DAD analysis identified apigenin, caffeic acid, ferulic acid, gallic acid, kaempferol, myricetin, quercetin, and syringic acid as major constituents. Molecular docking revealed apigenin, myricetin, and quercetin as top-ranked multitarget inhibitors, exhibiting strong binding affinities (− 9.0 to − 10.2 kcal/mol) comparable to those of reference inhibitors across α-amylase, α-glucosidase, AChE, BChE, and MAO. Molecular dynamics simulations and MM-GBSA confirmed the binding strength of the hit phytoconstituents in the active pockets of α-amylase, α-glucosidase, AChE, BChE, and MAO, with multitargeting inhibitory activities supporting the in vitro and ex vivo enzyme activities. ADMET profiling indicated favorable drug likeness for apigenin, whereas myricetin and quercetin displayed acceptable pharmacokinetic properties with minimal violations. Our findings provide scientific validation of the anti-T2D and anti-AD properties of S. aromaticum and identify apigenin, myricetin, and quercetin, which could be used for the development of inhibitors of α-amylase, α-glucosidase, AChE, BChE, and MAO as dual therapies to combat T2D and AD. Additional in vivo validation is recommended to ensure a thorough assessment in the present research.
Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Shi, Q., Liu, S., Fonseca, V. A., Thethi, T. K. & Shi, L. Effect of metformin on neurodegenerative disease among elderly adult US veterans with type 2 diabetes mellitus. BMJ Open 9, e024954 (2019).
Ojo, O. A. et al. Elucidating the interactions of compounds identified from Aframomum melegueta seeds as promising candidates for the management of diabetes mellitus: A computational approach. Inf. Med. Unlocked 26, 100720–100720 (2021).
Zarotti, N. et al. Psychological interventions for people with Parkinson’s disease in the early 2020s: Where do we stand?. Psychol. Psychother. Theory Res. Pract. 94, 760–797 (2020).
Kullenberg, H., Wibom, M., Kumlin, M., Nyström, T. & Svedberg, M. Associations between the use of metformin and behavioral and psychological symptoms in patients with Alzheimer’s disease, and type 2 diabetes mellitus—a register-based study. Curr. Alzheimer Res. 20, 109–119 (2023).
Nwidu, L., Elmorsy, E., Aprioku, J., Siminialayi, I. & Carter, W. In vitro anti-cholinesterase and antioxidant activity of extracts of Moringa oleifera plants from Rivers State, Niger Delta, Nigeria. Medicines 5, 71 (2018).
Hampel, H. et al. The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain J. Neurol. 41(7), 1917–1933 (2018).
Jellinger, K. A. & Korczyn, A. D. Are dementia with Lewy bodies and Parkinson’s disease dementia the same disease?. BMC Med. 16(1), 34 (2018).
Ćorković, I., Gašo-Sokač, D., Pichler, A., Šimunović, J. & Kopjar, M. Dietary polyphenols as natural inhibitors of α-amylase and α-glucosidase. Life 12(11), 1692. https://doi.org/10.3390/life12111692 (2022).
Casey, D. A., Antimisiaris, D. & O’Brien, J. Drugs for Alzheimer’s disease: Are they effective?. P T 35, 208–211 (2010).
Ojo, A. B. et al. Syzygium aromaticum (L.) Merr. & LM Perry mitigates iron-mediated oxidative brain injury via in vitro, ex vivo, and in-silico approaches. J. Mol. Struct. https://doi.org/10.1016/j.molstruc.2022.133675 (2022).
El-Saber Batiha, G. et al. Syzygium aromaticum L. (Myrtaceae): Traditional uses, bioactive chemical constituents, pharmacological and toxicological activities. Biomolecules 10, 202 (2020).
Jan, M. S. et al. Design, synthesis, in vitro, in vivo and in-silico studies of pyrrolidine-2,5-dione derivatives as multitarget anti-inflammatory agents. Eur. J. Med. Chem. 186, 111863 (2020).
Koh, T., Murakami, Y., Tanaka, S., Machino, M. & Sakagami, H. Re-evaluation of anti-inflammatory potential of eugenol in IL-1beta-stimulated gingival fibroblast and pulp cells. In Vivo 27, 269–273 (2013).
Nassar, M. et al. Chemical constituents of clove (Syzygium aromaticum, Fam. Myrtaceae) and their antioxidant activity. Latinoam. Quim. 35, 47 (2007).
Bouchentouf, S., Said, G., Noureddine, M., Hocine, A. & Angelika, B. A. A note study on antidiabetic effect of main molecules contained in clove using molecular modeling interactions with DPP-4 enzyme. Int. J. Comput. Theor. Chem. 5, 9–13 (2017).
Shukri, R., Mohamed, S. & Mustapha, N. M. Cloves protect the heart, liver and lens of diabetic rats. Food Chem. 122, 1116–1121 (2010).
Ahmad, S., Latif, A. & Qasmi, I. A. Effect of 50% ethanolic extract of Syzygium aromaticum (L.) Merr & Perry. (Clove) on the sexual behavior of normal male rats. BMC Complement. Altern. Med. 4, 17 (2004).
Issac, A., Gopakumar, G., Kuttan, R., Maliakel, B. & Krishnakumar, I. M. Safety and anti-ulcerogenic activity of a novel polyphenol-rich extract of clove buds (Syzygium aromaticum L.). Food Funct. 6, 842–852 (2015).
Agbaje, E. O. Gastrointestinal effects of Syzygium aromaticum (Myrtaceae) in animal models. Nig. Q. J. Hosp. Med. 18, 137–141 (2008).
Agbaje, E. O., Adeneye, A. A. & Daramola, A. O. Biochemical and toxicological studies of aqueous extract of Syzygium aromaticum (Myrtaceae) in rodents. Afr. J. Trad. CAM 6(3), 241–254 (2009).
Santin, J. R. et al. Gastroprotective activity of essential oil of the Syzygium aromaticum and its major component eugenol in different animal models. Naunyn Schmiedebergs Arch Pharmacol. 383(2), 149–158 (2011).
Adefegha, S. A. & Oboh, G. Inhibition of key enzymes linked to type 2 diabetes and sodium nitroprusside-induced lipid peroxidation in rat pancreas by water extractable phytochemicals from some tropical spices. Pharm. Biol. 50(7), 857–865 (2012).
Aisha, A. F. et al. Evaluation of antiangiogenic, cytotoxic and antioxidant effects of Syzygium aromaticum L. extracts. Asian J. Bio Sci. 4, 282–290 (2011).
Etukudo, E. M. et al. Exploring the neuroprotective potentials of flavonoid metabolites in Syzygium aromaticum: A review with in-silico insight to therapeutic potential. J. Exp. Pharmacol. 17, 587–611. https://doi.org/10.2147/JEP.S536765 (2025).
Amir Rawa, M. S., Mazlan, M. K. N., Ahmad, R., Nogawa, T. & Wahab, H. A. Roles of Syzygium in anti-cholinesterase, anti-diabetic, anti-inflammatory, and antioxidant: From Alzheimer’s perspective. Plants 11, 1476 (2022).
Zhan, F. et al. Toward understanding the interaction of β-lactoglobulin with capsaicin: Multispectroscopic, thermodynamic, molecular docking and molecular dynamics simulation approaches. Food Hydrocoll. 105, 105767–105767 (2020).
Ojo, O. A. et al. Exploring beetroot (Beta vulgaris L.) for diabetes mellitus and Alzheimer’s disease dual therapy: In vitro and computational studies. RSC Adv. 14, 19362–19380. https://doi.org/10.1039/d4ra03638g (2024).
Ojo, O. A. et al. Antidiabetic activity of avocado seeds (Persea americana Mill.) in diabetic rats via activation of PI3K/AKT signaling pathway. Sci. Rep. 12, 2919 (2022).
Bursal, E. et al. Phytochemical content, antioxidant activity, and enzyme inhibition effect ofSalvia eriophora Boiss. & Kotschy against acetylcholinesterase, α-amylase, butyrylcholinesterase, and α-glycosidase enzymes. J. Food Biochem. 43, e12776 (2019).
Erukainure, O. L. et al. Buddleja saligna Willd (Loganiaceae) inhibits angiotensin-converting enzyme activity in oxidative cardiopathy with concomitant modulation of nucleotide hydrolyzing enzymatic activities and dysregulated lipid metabolic pathways. J. Ethnopharmacol. 248, 112358 (2020).
Green, A. L. & Haughton, T. M. A. Colorimetric method for the estimation of monoamine oxidase. Biochem. J. 78, 172–176 (1961).
Morris, G. M. et al. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 30, 2785–2791 (2009).
O’Boyle, N. M. et al. Open Babel: An open chemical toolbox. J. Cheminform. https://doi.org/10.1186/1758-2946-3-33 (2011).
Trott, O. & Olson, A. J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31(2), 455–461 (2010).
Abraham, M. J. et al. GROMACS: High performance molecular simulations through multilevel parallelism from laptops to supercomputers. SoftwareX https://doi.org/10.1016/j.softx.2015.06.001 (2015).
Bekker, H. et al. Gromacs-a parallel computer for molecular-dynamics simulations, in Proceedings of the 4th International Conference on Computational Physics, (PC 92) (1993).
Oostenbrink, C., Villa, A., Mark, A. E. & Van Gunsteren, W. F. A biomolecular force field based on the free enthalpy of hydration and solvation: The GROMOS force-field parameter sets 53A5 and 53A6. J. Comput. Chem. 25(13), 1656–1676 (2004).
Lee, J. et al. CHARMM-GUI input generator for NAMD, GROMACS, AMBER, OpenMM, and CHARMM/OpenMM simulations using the CHARMM36 additive force field. J. Chem. Theory Comput. 12, 405–413 (2015).
Lee, J. et al. CHARMM-GUI supports the Amber force fields. J. Chem. Phys. 153, 035103 (2020).
Ogunyemi, O. M. et al. Identification of promising multitargeting inhibitors of obesity from Vernonia amygdalina through computational analysis. Mol. Divers. https://doi.org/10.1007/s11030-022-10397-6 (2022).
Gyebi, G. A., Ogunyemi, O. M., Ibrahim, I., Afolabi, S. O. & Adebayo, J. O. Dual targeting of cytokine storm and viral replication in COVID-19 by plant-derived steroidal pregnanes: An in-silico perspective. Comput. Biol. Med. 134, 104406 (2021).
Ogunyemi, O. M. et al. Dietary stigmastane-type saponins as promising dual-target directed inhibitors of SARS-CoV-2 proteases: A structure-based screening. RSC Adv. 11, 33380–33398 (2021).
Valdés-Tresanco, M. S., Valdés-Tresanco, M. E., Valiente, P. A. & Moreno, E. gmx_MMPBSA: A new tool to perform end-state free energy calculations with GROMACS. J. Chem. Theory Comput. 17, 6281–6291 (2021).
Miller, B. R. et al. MMPBSA.py: An efficient program for end-state free energy calculations. J. Chem. Theory Comput. 8, 3314–3321 (2012).
Bhat, B. A. et al. Natural therapeutics in aid of treating Alzheimer’s disease: A green gateway toward ending quest for treating neurological disorders. Front. Neurosci. 16, 1–23 (2022).
Salehi, P., Asghari, B., Esmaeili, M. A., Dehghan, H. & Ghazi, I. α-Glucosidase and α-amylase inhibitory effect and antioxidant activity of ten plant extracts traditionally used in Iran for diabetes. J. Med. Plants Res. 7, 257–266 (2013).
Khathi, A., Serumula, M. R., Myburg, R. B., Van Heerden, F. R. & Musabayane, C. T. Effects of Syzygium aromaticum-derived triterpenes on postprandial blood glucose in streptozotocin-induced diabetic rats following carbohydrate challenge. PLoS ONE 8, e81632 (2013).
Mwakalukwa, R., Amen, Y., Nagata, M. & Shimizu, K. Postprandial hyperglycemia lowering effect of the isolated compounds from olive mill wastes—An inhibitory activity and kinetics studies on α-Glucosidase and α-Amylase enzymes. ACS Omega 5, 20070–20079 (2020).
Liu, J.-Q. et al. Apigenin ameliorates lupus nephritis by inhibiting SAT3 signaling in CD8+T cells. Food. Funct. https://doi.org/10.1039/D4FO02773F (2024).
Samani, S. L. et al. Beneficial effects of apigenin on ovarian histological changes and angiogenesis gene expression in rat model of polycystic ovary syndrome. Naunyn-Schmiedeberg’s Arch. Pharmacol. https://doi.org/10.1007/s00210-024-03414-y (2024).
Nazdar, N., Imani, A., Abtahi Froushani, S. M., Farzaneh, M. & Sarvi Moghanlou, K. Antioxidative properties, phenolic compounds, and in vitro protective efficacy of multiherbal hydroalcoholic extracts of ginger, turmeric, and thyme against the toxicity of aflatoxin B1 on mouse macrophage RAW264.7 cell line. Food Sci. Nutr. 00, 1–17. https://doi.org/10.1002/fsn3.4257 (2024).
Egbuna, C. et al. Bioactive compounds effective against type 2 diabetes mellitus: A systematic review. Curr. Top. Med. Chem. 21(12), 1067–1095 (2021).
Nor, I., Wirasutisna, K. R., Hartati, R. & Insanu, M. The α-glucosidase inhibitory activity of avicularin and 4-O-methyl gallic acid isolated from Syzygium myrtifolium leaves. Saudi. Pharm. J. 31, 101677 (2023).
Mukherjee, PulokK.., Dalai, ManojK.., Bhadra, S., Chaudhary, SushilK.. & Bandyopadhyay, A. Anti-cholinesterase activity of the standardized extract of Syzygium aromaticum L.. Pharmacogn. Mag. 10, 276 (2014).
Moreira, N. C., Lima, J. E., Marchiori, M. F., Carvalho, I. & Sakamoto-Hojo, E. T. Neuroprotective effects of cholinesterase inhibitors: Current scenario in therapies for Alzheimer’s disease and future perspectives. J. Alzheimer’s Dis. Rep. 6(1), 177–193 (2022).
Chen, Z. R., Huang, J. B., Yang, S. L. & Hong, F. F. Role of cholinergic signaling in Alzheimer’s disease. Molecules 27(6), 1816 (2022).
El-Maati, M. F. A., Mahgoub, S. A., Labib, S. M., Al-Gaby, A. M. A. & Ramadan, M. F. Phenolic extracts of clove (Syzygium aromaticum) with novel antioxidant and antibacterial activities. Eur. J. Integr. Med. 8, 494–504 (2016).
Minic, J., Chatonnet, A., Krejci, E. & Molgó, J. Butyrylcholinesterase and acetylcholinesterase activity and quantal transmitter release at normal and acetylcholinesterase knockout mouse neuromuscular junctions. Br. J. Pharmacol. 138, 177–187 (2003).
Bouyahya, A. et al. Anti-inflammatory and analgesic properties of Moroccan medicinal plants: Phytochemistry, in vitro and in vivo investigations, mechanism insights, clinical evidence and perspectives. J. Pharm. Anal. 12, 35–57 (2021).
Sharma, H., Kim, D. Y., Shim, K. H., Sharma, N. & An, S. A. Multi-targeting neuroprotective effects of Syzygium aromaticum bud extracts and their key HPLC-identified compounds against neurodegenerative diseases.. Int. J. Mol. Sci. 24, 8148–8148 (2023).
Li, N., Yang, J., Wang, C., Wu, L. & Liu, Y. Screening bifunctional flavonoids of anti-cholinesterase and anti-glucosidase by in vitro and in silico studies: Quercetin, kaempferol and myricetin. Food Biosci. 51, 102312 (2023).
Rybak, M. & Wojdyło, A. Inhibition of α-Amylase, α-Glucosidase, pancreatic lipase, 15-Lipooxygenase and acetylcholinesterase modulated by polyphenolic compounds, organic acids, and carbohydrates of Prunus domestica fruit. Antioxidants 12(7), 1380 (2023).
Cheung, J. et al. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J. Med. Chem. 55(22), 10282–10286 (2012).
Gyebi, G. A. et al. Cholinergic inhibition and antioxidant potential of Gongronema latifolium benth leaf in neurodegeneration: Experimental and in silico study. Cell Biochem. Biophys. 83(1), 1–23 (2025).
Daina, A., Michielin, O. & Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 7, 42717 (2017).
Cacace, F., Menci, M., Papi, M. & Piemonte, V. In-silico prediction of oral drug bioavailability: A multiboluses approach. Med. Eng. Phys. 98, 140–150. https://doi.org/10.1016/j.medengphy.2021.11.004 (2021).
Park, K. S., Chong, Y. & Kim, M. K. Myricetin: Biological activity related to human health. Appl. Biol. Chem. 59, 259–269. https://doi.org/10.1007/s13765-016-0150-2 (2016).
Jablonsky, M., Haz, A., Burcova, Z., Kreps, F. & Jablonsky, J. Pharmacokinetic properties of biomass-extracted substances isolated by green solvents. Biores. 14(3), 6294–6303 (2019).
Liu, L., Barber, E., Kellow, N. J. & Williamson, G. Improving quercetin bioavailability: A systematic review and meta-analysis of human intervention studies. Food Chem. 477, 143630. https://doi.org/10.1016/j.foodchem.2025.143630 (2025).
Veber, D. F. et al. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 45, 2615–2623 (2002).
Lipinski, C. A. Drug-like properties and the causes of poor solubility and poor permeability. J. Pharmacol. Toxicol. Methods. 44, 235–249 (2000).
Funding
This study did not receive any funding whatsoever.
Author information
Authors and Affiliations
Contributions
OAO conceptualized and designed the study; MI, GAG, and OAO wrote the first draft; SD and TD performed the experiment; ABO, OAO, MI, AKO, BEO, BOA, and GAG analyzed and interpreted the data; and OAO supervised the experiment. MI, GAG, ABO, AKO, BEO, BOA, and OAO reviewed the final draft of the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The Institutional Research Animal Ethical Committee approved (BUI/BCH/2024/0002) the study protocols and research conducted in accordance with the ARRIVE (Animal Research: Reporting In Vivo Experiments) guidelines. All the experimental methods were performed in accordance with the relevant protocols, guidelines and regulations approved by the Bowen University Research Ethics Committee (BUREC).
Informed consent
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ojo, O.A., Gyebi, G.A., Iyobhebhe, M. et al. Deciphering the mechanisms underlying the dual-target inhibition of carbohydrate-digesting and neurodegenerative enzymes by Syzygium aromaticum (L.) Merr. & L.M. via molecular docking and dynamics simulations. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45482-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45482-5