Abstract
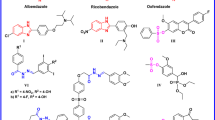
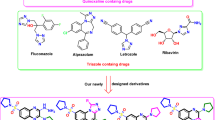
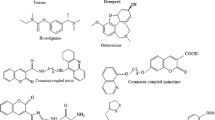
Isonicotinoyl hydrazone derivatives (1–20) were synthesized and evaluated for their inhibitory activities against myeloperoxidase (MPO) and acetylcholinesterase (AChE), antioxidant activity, metal ion chelation, regulation of reactive oxygen species, and cytoprotective activity, with the aim of identifying multi-target therapeutic candidates for Alzheimer’s disease. In this study, 9 exhibited a free radical scavenging activity of 93% and showed strong inhibitory effects against MPO and AChE, with inhibition rates of 90% and 73%, respectively. An in-depth structure–activity relationship analysis revealed that 9 possesses a favorable pharmacological balance between lipophilicity and electronic properties. Moreover, 9 effectively chelated Cu2⁺, Fe2⁺, Mg2⁺, and Zn2⁺ ions. It also reduced oxidative stress, significantly improved cell viability in neuronal damage models, and prevented cell death in a concentration-dependent manner. Furthermore, molecular docking studies demonstrated that 9 exhibits favorable binding affinities toward both target enzymes, providing a structural basis for its dual-target inhibitory activity and supporting the rationality of the structure–activity relationship analysis. Collectively, these results suggest that 9 represents a potential therapeutic candidate for the development of therapeutic agents for Alzheimer’s disease.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
Scheltens, P. et al. Alzheimer’s disease. Lancet 397, 1577–1590 (2021).
Avila, J. & Perry, G. A multilevel view of the development of Alzheimer’s disease. Neuroscience 457, 283–293 (2021).
Hane, F. T., Lee, B. Y. & Leonenko, Z. Recent progress in Alzheimer’s disease research, Part 1: pathology. J. Alzheimers Dis. 57, 1–28 (2017).
Knopman, D. S. et al. Alzheimer disease. Nat. Rev. Dis. Primers 10, 42 (2024).
Bai, R., Guo, J., Ye, X. Y., Xie, Y. & Xie, T. Oxidative stress: the core pathogenesis and mechanism of Alzheimer’s disease. Ageing Res. Rev. 77, 101619 (2022).
Sankar, J., Chauhan, A., Singh, R. & Mahajan, D. Isoniazid: historical development, metabolism associated toxicity and a perspective on its pharmacological improvement. Front. Pharmacol. 15, 1441147 (2024).
Frias, C. C. et al. Acylhydrazones derived from isonicotinic acid: synthesis, characterization, and evaluation against Alzheimer’s disease biomarkers. J. Mol. Struct. 1312, 138456 (2024).
Santos, D. C. et al. Acylhydrazones as isoniazid derivatives with multi-target profiles for the treatment of Alzheimer’s disease: radical scavenging, myeloperoxidase/acetylcholinesterase inhibition and biometal chelation. Bioorg. Med. Chem. 28, 115470 (2020).
Singh, A., Kukreti, R., Saso, L. & Kukreti, S. Oxidative stress: a key modulator in neurodegenerative diseases. Molecules 24, 1583 (2019).
Cipriano, A. et al. NADPH oxidases: from molecular mechanisms to current inhibitors in anticancer therapy. J. Med. Chem. 66, 7926–7955 (2023).
Birben, E., Sahiner, U. M., Sackesen, C., Erzurum, S. & Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 5, 9–19 (2012).
Rivera Antonio, A. M., Padilla Martínez, I. I., Torres-Ramos, M. A. & Rosales-Hernández, M. C. Myeloperoxidase as a therapeutic target for oxidative damage in Alzheimer’s disease. J. Enzyme Inhib. Med. Chem. 40, 2456282 (2025).
Liu, Z., Zhou, T., Ziegler, A. C., Dimitrion, P. & Zuo, L. Oxidative stress in neurodegenerative diseases: from molecular mechanisms to clinical applications. Oxid. Med. Cell. Longev. 2017, 2525967 (2017).
Bartolić, M., Matošević, A., Maraković, N. & Gašo-Sokač, D. Evaluation of hydrazone and N-acylhydrazone derivatives of vitamin B6 and pyridine-4-carbaldehyde as potential drugs against Alzheimer’s disease. J. Enzyme Inhib. Med. Chem. 39, 2431832 (2024).
Zou, D. et al. Latest advances in dual inhibitors of acetylcholinesterase and monoamine oxidase B against Alzheimer’s disease. J. Enzyme Inhib. Med. Chem. 38, 2270781 (2023).
Kondeva-Burdina, M., Krastev, K., Pencheva, T. & Angelova, V. T. Indole-based hydrazide-hydrazone and sulfonylhydrazone derivatives as MAO-B inhibitors with multitarget potential for neurodegenerative diseases. Pharmacia 72, 1–13 (2025).
Siddiqui, S. M., Salahuddin, A. & Azam, A. Synthesis, characterization and antiamoebic activity of some hydrazone and azole derivatives bearing pyridyl moiety as a promising heterocyclic scaffold. Eur. J. Med. Chem. 49, 411–416 (2012).
Judge, V. et al. Isonicotinic acid hydrazide derivatives: synthesis, antimicrobial activity, and QSAR studies. Med. Chem. Res. 21, 1451–1470 (2012).
Alegaon, S. G., Alagawadi, K. R. & Dadwe, D. H. Synthesis and antitubercular activity of novel 3,5-diaryl-4,5-dihydro-1H-pyrazole derivatives. Drug Res. (Stuttg.) 64, 553–558 (2014).
Perluigi, M., Di Domenico, F. & Butterfield, D. A. Oxidative damage in neurodegeneration: roles in the pathogenesis and progression of Alzheimer disease. Physiol. Rev. 104, 233–270 (2024).
Belkheiri, N. et al. Synthesis and antioxidant activity evaluation of a syringic hydrazones family. Eur. J. Med. Chem. 45, 3019–3026 (2010).
Uzor, N. E., McCullough, L. D. & Tsvetkov, A. S. Peroxisomal dysfunction in neurological diseases and brain aging. Front. Cell. Neurosci. 14, 44 (2020).
Yu, Y., Yu, S., Battaglia, G. & Tian, X. Amyloid-β in Alzheimer’s disease: structure, toxicity, distribution, treatment, and prospects. Int. J. Mol. Sci. 25, 10705 (2024).
Bitar, D. et al. Population-based analysis of invasive fungal infections, France. Emerg. Infect. Dis. 20, 1149–1155 (2014).
Pattison, D. I., Hawkins, C. L. & Davies, M. J. What are the plasma targets of the oxidant hypochlorous acid? A kinetic modeling approach. Free Radic. Biol. Med. 212, 345–358 (2024).
Baliyan, S. et al. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules 27, 1326 (2022).
Chen, W. et al. Therapeutic inhibition of MPO stabilizes pre-existing high risk atherosclerotic plaque. Redox Biol. 58, 102532 (2022).
Cabral, R. G., Viegas, G., Pacheco, R., Sousa, A. C. & Robalo, M. P. Sustainable synthesis, antiproliferative and acetylcholinesterase inhibition of 1,4- and 1,2-naphthoquinone derivatives. Molecules 28, 1232 (2023).
Dudev, T. & Lim, C. Metal binding affinity and selectivity in metalloproteins: insights from computational studies. Annu. Rev. Biophys. 37, 97–116 (2008).
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Contributions
ZW and KZ conceived and designed the study and supervised the overall research project. YC and ZL carried out the chemical synthesis and structural characterization of the compounds. BG and ZWg performed the biological activity assays, including enzyme inhibition and antioxidant experiments, and ZWg conducted the molecular docking studies. XM conducted the cell-based experiments and analyzed the cytoprotective activity data. ZW analyzed the data and drafted the manuscript. All authors discussed the results, contributed to data interpretation, and reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study did not involve any human participants or animals. Therefore, ethical approval was not required for this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wei, Z., Liu, Z., Chang, Y. et al. Synthesis, SAR, and multi-target evaluation of isonicotinoyl hydrazones as potent MPO/AChE inhibitors and metal chelators for Alzheimer’s disease. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45771-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45771-z