Abstract
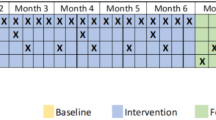
Minimally processed dietary interventions have been associated with reductions in depressive symptoms; however, the effort required to plan, shop for, and prepare minimally processed foods may limit adherence. Providing convenient access to minimally processed meals through commercial delivery services may reduce these barriers and potentially enhance intervention impact. This pilot randomized study evaluated the feasibility, acceptability, and preliminary effectiveness of this novel approach through a two-week dietary intervention among adults with moderate to moderately severe depressive symptoms. After one week of baseline observation, participants (N = 31) received nutritional guidance on how to eat a more minimally processed diet for two weeks, which was either fully facilitated by commercial meal delivery (n = 20), or self-implemented (e.g., nutritional guidance; n = 11). Dietary quality (assessed via Diet ID) and depressive symptoms (assessed via PHQ-8) were evaluated through a combination of surveys administered remotely and during in-person laboratory visits at intake, pre-intervention, and post-intervention. Both approaches exhibited high feasibility, producing significant pre–post improvements in dietary quality across the total sample (d = 1.93). Pre-post reductions in depressive symptoms were observed in the meal delivery condition (d = 1.62), but not the nutritional guidance condition (d = .54); these between-group differences should be interpreted cautiously given the pilot design and limited statistical power. Within-person analyses for the total sample indicated that greater improvements in dietary quality were associated with larger reductions in depressive symptoms, with dietary quality explaining 17% of the variance in depressive symptoms (semi-partial R2 = .17). These findings provide insight into the potential of minimally processed dietary interventions for improving dietary quality and mental health and lay the groundwork for larger trials to assess longer-term effectiveness and sustainability.
Similar content being viewed by others
Funding
We would like to acknowledge the Frances and Kenneth Eisenberg Endowment Fund and the University of Michigan Eisenberg Family Depression Center for their support in providing research funding that made this project possible. REDCap at the University of Michigan is supported by the Michigan Institute for Clinical and Health Research (MICHR), which is funded by the National Center for Advancing Translational Sciences (NCATS) of the National Institutes of Health under Award Number UM1TR004404. In addition to direct funding of this project, A. Gearhardt, C. Furman, J. Zhang, I. Worth, J. Henderson, and K. Sonneville are supported by the National Institute on Drug Abuse under Award Number R01DA055027. J. Lee is supported by the Elizabeth Weiser Caswell Diabetes Institute at the University of Michigan and the National Institute of Diabetes and Digestive and Kidney Diseases under Award Numbers P30DK089503 (MNORC), P30DK020572 (MDRC), and P30DK092926 (MCDTR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Principle Investigator A. Gearhardt has received speaking honoraria from academic organizations and health-related nonprofits, consulting fees from health-related nonprofits and a law firm and receives royalties from Oxford University Press. Co-I J. Lee receives grant funding from Lilly USA, LLC, participated on the Medical Advisory Board for GoodRx, and served as a consultant to Tandem Diabetes Care. These consulting roles and companies were not involved in any step of the research process (e.g., funding, study design, collection and analysis of data, writing or approval of manuscript) and did not influence the content, analysis, or conclusions of this research project or manuscript. No other conflicts of interest are declared with respect to the authorship or the publication of this article.
Ethics approval and consent to participate
This study’s procedures and materials were approved by the Institutional Review Board at the University of Michigan. All participants provided written informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Furman, C.R., Worth, I.A., Zhang, J.D. et al. Meals that heal: a randomized controlled trial testing the feasibility of commercial meal delivery as a convenient dietary intervention for depression. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45991-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45991-3