Abstract
Macrophages, the predominant immune-related stromal cells within tumor microenvironment, could be polarized into M1 and alternative M2 macrophages. M1 macrophages are pro-inflammatory and eliminate cancer cells, whereas M2 macrophages are immunosuppressive and facilitate tumor growth. Mounting evidence has shown that M2 macrophages are responsible for a poor prognosis in triple negative breast cancer (TNBC) patients. Since neoadjuvant chemotherapy is becoming the foundation treatment for TNBC, the relationship between M2 macrophages and response to neoadjuvant chemotherapy remains to be elucidated. Infiltration of M2 macrophages were checked by analyzing the CD163 levels on surgical resection specimens from 42 Chinese TNBC patients treated with neoadjuvant chemotherapy. Evaluation of M2 macrophages together with other immune markers including CD8-positive cytotoxic T cells and PD-L1 expression in TNBC and its association with pCR after neoadjuvant chemotherapy were performed. Pathological complete response (pCR) was significantly associated with younger age, higher tumor or stromal PD-L1 expression, higher levels of tumor or stromal CD8-positive cytotoxic T cells, but lower infiltration of CD163-positive M2 macrophages, with the level of CD163-positive M2 macrophages in stromal area as the strongest factor. Our study showed that high infiltration of CD163-positive M2 macrophages was negatively associated with the chemotherapeutic response to neoadjuvant chemotherapy in Chinese TNBC patients and could be used as a promising prognostic candidate for Chinese TNBC patients treated with neoadjuvant chemotherapy.
Similar content being viewed by others
Introduction
Breast cancer is the most common malignancy among women worldwide, and the subtype of particular note is triple negative breast cancer (TNBC) which accounts for 10–20% of all breast cancers and is characterized by the lack of estrogen receptors, progesterone receptors, and human epidermal growth factor receptor-2 (HER-2) expression1. Neoadjuvant chemotherapy is becoming the foundation treatment for TNBC patients2,3. Tumor response to neoadjuvant chemotherapy is commonly evaluated by microscopically checking the surgical resection specimens, and pathological complete response (pCR) is defined as no residual invasive carcinoma. Standard neoadjuvant strategy for TNBC includes regimens with cycles of anthracycline and cyclophosphamide followed by cycles of taxanes (EC-T regimen)2,3. Accumulating studies have shown that the utility of neoadjuvant chemotherapy results in a significantly higher pCR rate in TNBC patients than in hormone receptor-positive breast cancer patients4.
Neoadjuvant chemotherapy could modify the tumor immune microenvironment in several ways5. Moreover, changes in tumor immunity after neoadjuvant chemotherapy were more common in TNBC with respect to other subtypes6. Macrophages, the most abundant immune-related stromal cells within tumor microenvironment, could be polarized into M1 and alternative M2 macrophages. M1 macrophages are pro-inflammatory and eliminate cancer cells, whereas M2 macrophages are immunosuppressive and facilitate tumor growth7. Mounting evidence has shown that M2 macrophages could promote chemoresistance, invasion and metastasis of tumor cells, and inhibit anti-tumor immune response8. M2 macrophages have also been proved to be responsible for a poor prognosis in breast cancer9. Tumor-infiltrating lymphocytes, which are indicators of immune response monitoring, were associated with better response to neoadjuvant chemotherapy and could predict recurrence of breast cancer after neoadjuvant chemotherapy including TNBC patients10,11. Several studies have analyzed programmed cell death 1 ligand (PD-L1) alterations after neoadjuvant chemotherapy in breast cancer and confirmed that PD-L1 was related to response to chemotherapy and overall survival, especially in TNBC patients12.
In order to evaluate the relationship between M2 macrophages and response to neoadjuvant chemotherapy, we evaluated the levels of CD163-positive M2 macrophages together with PD-L1 and CD8-positive cytotoxic T cells in TNBC and their associations with pCR after neoadjuvant chemotherapy.
Methods
Patients and samples
This study was approved by the ethics committee of Changzhou No.2 People’s Hospital, the Third Affiliated Hospital of Nanjing Medical University. Collection, preservation, and analyzation of clinical samples were conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of Changzhou No.2 People’s Hospital, the Third Affiliated Hospital of Nanjing Medical University. Informed written consent was obtained from all individual patients.
From February 2022 to May 2024, a total of 42 TNBC patients confirmed histologically by core needle biopsy with immunohistochemistry (IHC) were enrolled in the present study. Inclusion criteria were being female and having primary TNBC that was stages II or stage III. Patients with carcinoma in situ, stage IV breast cancer, inflammatory breast cancer and those who received any endocrine agents or radiotherapy before neoadjuvant chemotherapy were excluded. Detailed clinical data were collected by retrospective evaluation of patients’ medical records. All these patients received 4 cycles of anthracycline and cyclophosphamide followed by weekly paclitaxel or dose-dense paclitaxel (EC-T regimen). After neoadjuvant chemotherapy, either mastectomy or breast-conserving surgery were performed to remove the primary tumor, and sentinel lymph node biopsy or axillary lymph node dissection were conducted to excise the lymph nodes. Surgical resection specimens were microscopically checked to identify any residual tumor. When residual invasive tumor was not found in breast tissues and metastasis was not detected in lymph node, the diagnosis of pCR was rendered and confirmed by two different pathologists who were unaware of the original and each other’s decision. Then, the collected samples were submitted for IHC examination.
Pathology and immunohistochemistry
Nottingham grading system is the most commonly used method for histological grading. Such technique involves evaluation of three morphological features: percentage of tubule formation, degree of nuclear pleomorphism, and an accurate mitotic count13. In the present study, specimens were histologically graded by the Nottingham grading system using the pre-neoadjuvant chemotherapy core needle biopsy tissues. Two different pathologists who were blinded to the clinical and pathological data checked the IHC results on the surgical resection samples. M2 macrophages, cytotoxic T cells, and PD-L1 expressions were evaluated by IHC with three antibodies including CD163, CD8, and PD-L1 (Abcam, Cambridge, MA, USA) under the manufacturer’s instructions. In particular, M2 macrophages and cytotoxic T cells were respectively evaluated by analyzing the percentage of tumoral and stromal areas infiltrated by CD163-positive and CD8-positive cells. PD-L1 expression was checked by calculating the percentage of tumor cells and stromal cells with membranous PD-L1 staining. When inconsistencies were more than 5%, reevaluation was conducted for consensus.
Statistical analysis
Data were analyzed using the SPSS 20.0 package. Associations between clinical variables and response to neoadjuvant chemotherapy were analyzed by Pearson’s chi-square test or Fisher’s exact test. All the continuous variables have been tested for normality using Kolmogorov-Smirnov test. A multivariate logistic regression analysis was performed when a variable was an independent predictor of pCR in the univariate analysis. Variables with p > 0.05 were sequentially removed from the multivariate model using forward selection method. All data were expressed as mean or mean (min, max), and p < 0.05 was considered statistically significant.
Results
Clinical and pathological characteristics of TNBC patients
The specific clinical and pathological features of 42 TNBC patients with neoadjuvant chemotherapy and subsequent surgical resection were shown in Table 1. The mean age was 51.8 years old (26–71 years). Forty cases (95.2%) were invasive ductal carcinoma, and the rest 2 cases (4.8%) were metaplastic carcinoma. All cases were Nottingham grade 3 or grade 2 with an average of 2.75. About 34 cases (81.0%) were HER-2 negative, and the other 8 cases (19.0%) showed equivocal HER-2, but negative in fluorescence in-situ hybridization. A total of 23 cases (54.8%) showed pCR. Among 19 non-pCR cases (45.2%) with residual tumor, 10 cases were positive for lymph nodes.
Univariate analysis of factors related to response to neoadjuvant chemotherapy in TNBC patients
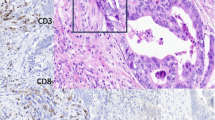
Twenty-three cases (54.8%) had pCR and 19 non-pCR cases (45.2%) had residual tumor. Among 42 cases, tumoral PD-L1 expression was identified in 38 (90.5%) cases with at least 1%, and PD-L1 expression in stromal cells was also identified in 38 (90.5%) cases. Both tumoral and stromal CD8-positive cytotoxic T cell infiltration were found in 39 (92.9%) cases. CD163-positive M2 macrophages were found in tumoral area for all cases and in stromal area for all cases. Univariate analysis was used to check the associations between the response to neoadjuvant chemotherapy and clinical variables, including age, histology type, grade, HER-2 expression, PD-L1, CD8, and CD163 levels. As shown in Table 2, patients in the pCR group were younger than patients in the non-pCR group (48.8 vs. 55.4, p = 0.039). Moreover, increased tumoral and stromal PD-L1 expression were found in the pCR group, with respect to the non-pCR group (tumoral: 5.4% vs. 2.3%, p = 0.002; stromal: 5.6% vs. 1.9%, p = 0.001). Higher levels of tumoral and stromal CD8-positive cytotoxic T cells were observed in the pCR group than in the non-pCR group (tumoral: 9.9% vs. 6.9%, p = 0.020; stromal: 12.1% vs. 7.9%, p = 0.009). When compared to patients in the non-pCR group, patients in the pCR group showed reduced CD163-positive M2 macrophages in both tumoral and stromal areas (tumoral: 26.4% vs. 35.3%, p = 0.015; stromal: 28.1% vs. 37.0%, p = 0.008) (Fig. 1; Table 2).
Comparison of CD163, CD8, and PD-L1 expressions between pCR group and non-pCR group. Higher PD-L1 expression, higher level of CD8-positive cytotoxic T cells, but lower infiltration of CD163-positive M2 macrophages were identified in the pCR group with respect to the non-pCR group in TNBC patients treated with neoadjuvant chemotherapy (x 200).
Multivariable analysis of factors related to response to neoadjuvant chemotherapy in TNBC patients
Variables with p < 0.10 in the univariate analysis, namely, age, PD-L1 tumoral, PD-L1 stromal, CD8 tumoral, CD8 stromal, CD163 tumoral, and CD163 stromal, were then selected for multivariable logistic regression analysis. Besides, variables with p > 0.05 were sequentially removed from the multivariate model using forward selection method. It was found that increased stromal PD-L1 expression (p = 0.001) and decreased CD163-positive M2 macrophages in stromal areas (p = 0.007) were significantly associated with pCR to neoadjuvant chemotherapy in TNBC patients (Table 3).
Discussion
Application of neoadjuvant chemotherapy brings the reduction of primary breast tumor and aims to eliminate lymph nodes. Although novel agents are being developed for breast cancer treatment, the most commonly used chemotherapy regimen in a neoadjuvant setting for TNBC is anthracycline and cyclophosphamide given in dose-dense schedule followed by weekly paclitaxel or dose-dense paclitaxel (EC-T regimen)2,3. Data from our present study are consistent with the results of previous work showing that the pCR rate ranges from 49% to 71% in early-stage TNBC patients12,14. According to CTNeoBC pooled analysis data, achieving pCR is associated with better long-term survival15. While pCR is a critical endpoint, residual cancer burden has also been shown to better predict long-term outcomes. Indeed, TNBC patients in the I-SPY2 clinical trial had worse event-free survival as residual cancer burden increased16. However, the long-term resuls are not the study purpose of the present work, residual cancer burden classification, progression-free survival, and overall survival are therefore not included in the present work.
Accumulating studies demonstrated that M2 macrophages, the predominant immune-related stromal cells within tumor microenvironment, play an essential role in oncogenesis, progression, invasion, and metastasis8. However, research of M2 macrophages’ effect on response to neoadjuvant chemotherapy in breast cancer patients is still lacking. CD163 is a well-known specific marker for M2 macrophages. Although several previous publications showed that high infiltration of CD163-positive M2 macrophages was related to a poor response to neoadjuvant chemotherapy in TNBC patients17,18, there are still some limitations. For example, Ye et al. showed that CD163-positive M2 macrophages may predict chemotherapeutic response in Chinese TNBC patients treated with three different neoadjuvant chemotherapy regimens, but only 1/5 patients received the classic EC-T regimen17. Arole et al. revealed that the level of CD163-positive M2 macrophages was significantly higher in TNBC patients with incomplete response than patients with complete response, but all research data were collected from Caucasian and no Asian data were available18. In the present work, we have also demonstrated that the level of CD163-positive M2 macrophages, especially in stromal area, were negatively associated with pCR to neoadjuvant chemotherapy in Chinese TNBC patients receiving EC-T regimen. Our findings could be very helpful with respect to Ye et al.‘s and Arole’s cohort studies, as it is important to recognize the race and regimen factors that may influence response to neoadjuvant chemotherapy. Besides, we know that chemotherapy itself can modify the immune microenvironment; however, tumors can also orchestrate their immune microenvironment via secreted factors, organizing a complex and dynamic network of macrophage polarization. Therefore, there is no need to clarify whether M2 macrophages were elevated before therapy initiation, as reported by prior studies on M2 macrophages in TNBC17,18.
Remarkably, the detailed mechanism about M2 macrophages and chemoresistance of neoadjuvant chemotherapy in TNBC has not been fully clarified. Xu et al. reported that neoadjuvant chemotherapy could increase M2 macrophages infiltration and M2 macrophages-derived IL-6 would mediate resistance of breast cancer cells to hedgehog inhibition19. Our group has found that chemoresistant breast cancer cells could remodel macrophages within tumor microenvironment by secreting exosomal miR-222, which directly targeted PTEN and caused Akt cascade activation and macrophages M2 polarization20. Although this is the first study to investigate the relationship between M2 macrophages and the response to neoadjuvant chemotherapy using EC-T regimen in Chinese TNBC patients, the detailed mechanisms and crosstalk about how M2 macrophages modify the drug resistance during neoadjuvant chemotherapy warrant an in-depth research.
In the present work, several other clinical variables including age, PD-L1 expression, and CD8-positive cytotoxic T cells were also associated with response to neoadjuvant chemotherapy. By using univariate analysis, it was found that younger age, higher level of PD-L1 expression, and increased CD8-positive cytotoxic T cells were positive predictors for pCR in TNBC patients of our cohort study. Moreover, multivariate analysis confirmed that increased stromal PD-L1 expression was positively related to pCR in TNBC patients receiving neoadjuvant chemotherapy. Previous studies have demonstrated the value of tumor-infiltrating lymphocytes for predicting the response to neoadjuvant chemotherapy in breast cancer10,11. Moreover, Zhu et al. found that the combination of tumor mutational burden and the ratio of CD8-positive cytotoxic T cells to M2 macrophages may be a potential biomarker for prediction of neoadjuvant chemotherapy21. Another research showed that high adipogenesis TNBC was not sensitive to neoadjuvant chemotherapy and was infiltrated with low CD8-positive T cells and high M2 macrophages22. Failure of CD8-positive cytotoxic T cells as a significant predictor during multivariable analysis is probably caused by the small sample size of our cohort study, although the mean percentage of CD8-positive cells in pCR group is greater with respect to non-pCR group without statistically significant difference. The relationship between PD-L1 expression and cancer prognosis is still controversial. We have observed the positive association between PD-L1 expression and pCR in the present work. Ahmed also found that PD-L1 expression on both tumor cells and macrophages were positively associated with response to neoadjuvant chemotherapy in TNBC23. However, our cellular research has confirmed that exosomal HMGB1 from TNBC could target Tim-3 ligand on macrophages and accelerate glycolysis and lactate production to induce PD-L1+-macrophages, thereby suppressing T lymphocytes immunity and generating a pre-metastasis immune-suppressive niche24. This findings might generate much enthusiasm in exploring the multiple roles of PD-L1.
Our study has several limitations. First, the sample size is relatively small. We could not establish the positive association between CD8-positive T cells and pCR in.
multivariable analysis with statistical significance, although a significant correlation was found in univariate analysis. Second, residual cancer burden was not analyzed in the present work since previous TNBC cohort study showed that long-term prognostic risk after neoadjuvant chemotherapy was associated with residual cancer burden25. Finally, genomic and transcriptomic landscape of TNBC were quite different26, and taking the aforementioned limitations together with a small sample size suggest that our findings need to be validated with prospective large cohort studies, and more cellular research should be performed in this field.
In conclusion, high infiltration of CD163-positive M2 macrophages was negatively associated with the chemotherapeutic response to neoadjuvant chemotherapy in Chinese TNBC patients receiving EC-T regimen and could be used as a promising prognostic candidate for Chinese TNBC patients treated with neoadjuvant chemotherapy.
Data availability
The data sets analyzed during the current study are available from the corresponding author on reasonable request.
References
Angela, N. et al. Breast Cancer Stat. 2022 CA Cancer J. Clin. ;72(6):524–541. (2022).
Larissa, A. et al. Neoadjuvant Chemotherapy, Endocrine Therapy, and Targeted Therapy for Breast Cancer: ASCO Guideline. J. Clin. Oncol. 39 (13), 1485–1505 (2021).
Jieun Lee. Current Treatment Landscape for Early Triple-Negative Breast Cancer (TNBC). J. Clin. Med. 12 (4), 1524 (2023).
Milos Holanek, I. et al. Neoadjuvant Chemotherapy of Triple-Negative Breast Cancer: Evaluation of Early Clinical Response, Pathological Complete Response Rates, and Addition of Platinum Salts Benefit Based on Real-World Evidence. Cancers (Basel). 13 (7), 1586 (2021).
Galizia, D. et al. How Risk Factors Affect Head and Neck Squamous Cell Carcinoma (HNSCC) Tumor Immune Microenvironment (TIME): Their Influence on Immune Escape Mechanisms and Immunotherapy Strategy. Biomedicines 10 (10), 2498 (2022).
Margaret, L. et al. Changes in Peripheral and Local Tumor Immunity after Neoadjuvant Chemotherapy Reshape Clinical Outcomes in Patients with Breast Cancer. Clin. Cancer Res. 26 (21), 5668–5681 (2020).
Siamon Gordon, Fernando, O. & Martinez Alternative activation of macrophages: mechanism and functions. Immunity 32 (5), 593–604 (2010).
Zhang, W., Wang, M., Ji, C., Liu, X. & Gu, B. Ting Dong. Macrophage polarization in the tumor microenvironment: Emerging roles and therapeutic potentials. Biomed. Pharmacother. 177, 116930 (2024).
Eleanor Allison, S., Edirimanne, J., Matthews, Stephen, J. & Fuller Breast Cancer Survival Outcomes and Tumor-Associated Macrophage Markers: A Systematic Review and Meta-Analysis. Oncol. Ther. 11 (1), 27–48 (2023).
Faur, I. F. et al. The Value of Tumor Infiltrating Lymphocytes (TIL) for Predicting the Response to Neoadjuvant Chemotherapy (NAC) in Breast Cancer according to the Molecular Subtypes. Biomedicines 11 (11), 3037 (2023).
Shinichiro Kashiwagi, Y. et al. Outcome Prediction After Neoadjuvant Chemotherapy (NAC) for Breast Cancer, Using Tumor-infiltrating Lymphocytes Within Fibrotic Foci of Tumor Stroma (FF-TILs). Anticancer Res. 42 (3), 1421–1431 (2022).
Javier Cortes, D. W. et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): a randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet 396 (10265), 1817–1828 (2020).
Elston, C. W. & Ellis, I. O. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19 (5), 403–410 (1991).
Schmid, P. et al. Pembrolizumab plus chemotherapy as neoadjuvant treatment of high-risk, early-stage triple-negative breast cancer: results from the phase 1b open-label, multicohort KEYNOTE-173 study. Ann. Oncol. 31 (5), 569–581 (2020).
Cortazar, P. et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet 384 (9938), 164–172 (2014).
Roberto, A. et al. Combined prognostic impact of initial clinical stage and residual cancer burden after neoadjuvant systemic therapy in triple-negative and HER2-positive breast cancer: an analysis of the I-SPY2 randomized clinical trial. Breast Cancer Res. 27 (1), 115 (2025).
Ye, J. H. et al. Tumor-associated macrophages are associated with response to neoadjuvant chemotherapy and poor outcomes in patients with triple-negative breast cancer. J. Cancer. 12 (10), 2886–2892 (2021).
Vidya Arole, H. et al. M2 tumor-associated macrophages play important role in predicting response to neoadjuvant chemotherapy in triple-negative breast carcinoma. Breast Cancer Res. Treat. 188 (1), 37–42 (2021).
Xu, X., Ye, J., Huang, C., Yan, Y. & Li, J. M2 macrophage-derived IL6 mediates resistance of breast cancer cells to hedgehog inhibition. Toxicol. Appl. Pharmacol. 364, 77–82 (2019).
Chen, W. X. et al. Exosomal miR-222 from adriamycin-resistant MCF-7 breast cancer cells promote macrophages M2 polarization via PTEN/Akt to induce tumor progression. Aging (Albany NY). 13 (7), 10415–10430 (2021).
Zhu, Y., Zhang, H., Pan, C., He, G. & Cui, X. Integrated tumor genomic and immune microenvironment analysis identifies predictive biomarkers associated with the efficacy of neoadjuvant therapy for triple-negative breast cancer. Cancer Med. 12 (5), 5846–5858 (2023).
Masanori Oshi, Y. et al. Adipogenesis in triple-negative breast cancer is associated with unfavorable tumor immune microenvironment and with worse survival. Sci. Rep. 11 (1), 12541 (2021).
Ahmed, F. S. et al. PD-L1 Protein Expression on Both Tumor Cells and Macrophages are Associated with Response to Neoadjuvant Durvalumab with Chemotherapy in Triple-negative Breast Cancer. Clin. Cancer Res. 26 (20), 5456–5461 (2020).
Chen, W. X., Wu, J. H. & Cheng, T. C. Sujin Yang. Exosomal HMGB1 induce PD-L1+-tumor associated macrophages through glycometabolic reprogramming to promote lung-tropic metastasis of triple negative breast cancer [abstract]. In: Proceedings of the 2023 San Antonio Breast Cancer Symposium; Dec 5–9; San Antonio, TX. Philadelphia (PA): AACR; Cancer Res 2024;84(9 Suppl):Abstract nr PO1-06-11. (2023).
Symmans, W. F. et al. Long-Term Prognostic Risk After Neoadjuvant Chemotherapy Associated With Residual Cancer Burden and Breast Cancer Subtype. J. Clin. Oncol. 35 (10), 1049–1060 (2017).
Jiang, Y. Z., Ma, D., Suo, C., Shi, J. & Xin, M. X. Genomic and Transcriptomic Landscape of Triple-Negative Breast Cancers: Subtypes and Treatment Strategies. Cancer Cell. 35 (3), 428–440e5 (2019).
Acknowledgements
The authors thank Dr Wei Liang for providing technical assistance.
Funding
This present work was kindly supported by grants from the Changzhou Medical Center (CMCB202401), the Jiangsu Provincial Health Commission General Project (MQ2024036), the Excellent Post-doctoral Program of Jiangsu Province (2022ZB820), the Top Talent of Changzhou ‘’The 14th Five-Year Plan’’ High-Level Health Talents Training Project (2022CZBJ065), the Post-doctoral Foundation of China (2022M720543).
Author information
Authors and Affiliations
Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Wx., Yang, M., Shao, Qf. et al. M2 macrophages predict response to neoadjuvant chemotherapy in triple negative breast cancer patients. Sci Rep 16, 10638 (2026). https://doi.org/10.1038/s41598-026-46026-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-46026-7