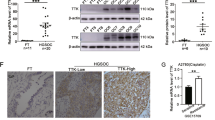
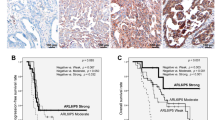
Abstract
The identification of novel molecular drivers and the development of new state-of-the-art therapies are critical challenges in ovarian cancer (OC) treatment. Cyclin-dependent kinase 12 (CDK12) is a promising target, as it’s functional activity promotes genomic stability. Here, we examined the anticancer efficacy of the dual CDK12/13-inhibitor SR-4835 in platinum-sensitive and -resistant OC cell lines, as well as its potential as a drug partner for platinum or olaparib combination therapy. SR-4835 exhibited potent anti-proliferative effects on most OC cell lines with IC50 values within the nanomolar range. A tendency for increased sensitivity of the cisplatin-resistant compared to their sensitive, parental cell lines was observed. Transcriptome analyses indicated gross changes in gene expression in numerous signaling pathways by SR-4835. Gene downregulation was in part due to alternative exon usage, which correlated with the number of intronic polyadenylation sites per gene and gene length. Furthermore, SR-4835 lead to the downregulation of key homologous recombination pathway genes rendering a BRCAness phenotype. However, the combination of SR-4835 with cisplatin or olaparib primarily exhibited an additive, not synergistic, effect. In summary, the present findings indicate that CDK12/13 inhibitor SR-4835 has potent anti-cancer effects accompanied by a BRCAness induction, but fails to achieve synergistic effects with cisplatin or olaparib in OC cells.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are provided in the Supplementary Information of this article or are available from the corresponding author upon request. The datasets generated and/or analysed during the current study are available in the ArrayExpress repository under accession number E-MTAB-15987 (https://www.ebi.ac.uk/biostudies/ArrayExpress/studies/E-MTAB-15987). Raw sequencing reads (FASTQ) can be retrieved from the European Nucleotide Archive under accession number PRJEB93935 (https://www.ebi.ac.uk/ena/browser/view/PRJEB93935).
Abbreviations
- CDK:
-
Cyclin-dependent kinase
- DDR:
-
DNA damage response
- HRD:
-
Homologous recombination repair deficiency
- MTT:
-
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide
- OC:
-
Ovarian cancer
- PARP:
-
Poly (ADP-ribose) polymerase
- PARPi:
-
PARP inhibitors
- poly(A):
-
Polyadenylation
- qPCR:
-
QuantitaTive real-time PCR
References
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 71, 209–249 (2021).
Karam, A. et al. Fifth Ovarian Cancer Consensus Conference of the Gynecologic Cancer InterGroup: First-line interventions. Ann. Oncol. 28, 711–717 (2017).
Vergote, I. et al. Treatment algorithm in patients with ovarian cancer. Facts Views Vis. Obgyn. 12, 227–239 (2020).
Moore, K. et al. Maintenance Olaparib in patients with newly diagnosed advanced ovarian cancer. N. Engl. J. Med. 379, 2495–2505 (2018).
González-Martín, A. et al. Niraparib in patients with newly diagnosed advanced ovarian cancer. N. Engl. J. Med. 381, 2391–2402 (2019).
Friedlander, M. et al. Clinical trials in recurrent ovarian cancer. Int. J. Gynecol. Cancer 21, 771–775 (2011).
Miller, R. E., El-Shakankery, K. H. & Lee, J.-Y. PARP inhibitors in ovarian cancer: Overcoming resistance with combination strategies. J. Gynecol. Oncol. 33, e44 (2022).
Greenleaf, A. L. Human CDK12 and CDK13, multi-tasking CTD kinases for the new millennium. Transcription 10, 91–110 (2019).
Sokol, E. S. et al. Pan-cancer analysis of CDK12 loss-of-function alterations and their association with the focal tandem-duplicator phenotype. Oncologist 24, 1526–1533 (2019).
Popova, T. et al. Ovarian cancers harboring inactivating mutations in CDK12 display a distinct genomic instability pattern characterized by large tandem duplications. Cancer Res. 76, 1882–1891 (2016).
Bajrami, I. et al. Genome-wide profiling of genetic synthetic lethality identifies CDK12 as a novel determinant of PARP1/2 inhibitor sensitivity. Cancer Res. 74, 287–297 (2014).
Blazek, D. The cyclin K/Cdk12 complex: An emerging new player in the maintenance of genome stability. Cell Cycle 11, 1049–1050 (2012).
Blazek, D. et al. The cyclin K/Cdk12 complex maintains genomic stability via regulation of expression of DNA damage response genes. Genes Dev. 25, 2158–2172 (2011).
Joshi, P. M., Sutor, S. L., Huntoon, C. J. & Karnitz, L. M. Ovarian cancer-associated mutations disable catalytic activity of CDK12, a kinase that promotes homologous recombination repair and resistance to cisplatin and poly(ADP-ribose) polymerase inhibitors. J. Biol. Chem. 289, 9247–9253 (2014).
Ekumi, K. M. et al. Ovarian carcinoma CDK12 mutations misregulate expression of DNA repair genes via deficient formation and function of the Cdk12/CycK complex. Nucleic Acids Res. 43, 2575–2589 (2015).
Krajewska, M. et al. CDK12 loss in cancer cells affects DNA damage response genes through premature cleavage and polyadenylation. Nat. Commun. 10, 1757 (2019).
Dubbury, S. J., Boutz, P. L. & Sharp, P. A. CDK12 regulates DNA repair genes by suppressing intronic polyadenylation. Nature 564, 141–145 (2018).
Quereda, V. et al. Therapeutic targeting of CDK12/CDK13 in triple-negative breast cancer. Cancer Cell 36, 545-558.e7 (2019).
Johnson, S. F. et al. CDK12 inhibition reverses de novo and acquired PARP inhibitor resistance in BRCA wild-type and mutated models of triple-negative breast cancer. Cell Rep. 17, 2367–2381 (2016).
Wang, C. et al. CDK12 inhibition mediates DNA damage and is synergistic with sorafenib treatment in hepatocellular carcinoma. Gut 69, 727–736 (2020).
Iniguez, A. B. et al. EWS/FLI confers tumor cell synthetic lethality to CDK12 inhibition in Ewing sarcoma. Cancer Cell 33, 202-216.e6 (2018).
Cheng, L. et al. Dual inhibition of CDK12/CDK13 targets both tumor and immune cells in ovarian cancer. Cancer Res. 82, 3588–3602 (2022).
Houles, T. et al. The CDK12 inhibitor SR-4835 functions as a molecular glue that promotes cyclin K degradation in melanoma. Cell Death Discov. 9, 459 (2023).
Rajan, N. et al. Differential activity of specific inhibitors of transcription regulating cyclin-dependent kinases in thyroid cancer cells. Endocr Relat Cancer https://doi.org/10.1530/ERC-24-0124 (2025).
CDK12 DepMap Gene Summary. https://depmap.org/portal/gene/CDK12?tab=characterization&characterization=expression.
Arafeh, R., Shibue, T., Dempster, J. M., Hahn, W. C. & Vazquez, F. The present and future of the Cancer Dependency Map. Nat Rev Cancer 25, 59–73 (2025).
Bliss, C. I. The toxicity of poisons applied jointly. Ann. Appl. Biol. 26, 585–615 (1939).
Davies, M., Boyce, M. & Conway, E. Short circuit: Transcription factor addiction as a growing vulnerability in cancer. Curr. Opin. Struct. Biol. 89, 102948 (2024).
Santos-Pereira, J. M., Carnero, A. & Muñoz-Galván, S. Integrative multi-omic analysis reveals a PAX8-driven gene network linking tumor stemness to therapy response in ovarian cancer. NAR Genom. Bioinform. 7, lqaf113 (2025).
Cesari, E. et al. Dual inhibition of CDK12 and CDK13 uncovers actionable vulnerabilities in patient-derived ovarian cancer organoids. J. Exp. Clin. Cancer Res. 42, 126 (2023).
Kaufman, B. et al. Olaparib monotherapy in patients with advanced cancer and a germline BRCA1/2 mutation. J. Clin. Oncol. 33, 244–250 (2015).
Dieter, S. M. et al. Degradation of CCNK/CDK12 is a druggable vulnerability of colorectal cancer. Cell Rep. 36, 109394 (2021).
Parson, W. et al. Cancer cell line identification by short tandem repeat profiling: Power and limitations. FASEB J. 19, 434–436 (2005).
Fiegl, H. et al. Dubious effects of methadone as an ‘anticancer’ drug on ovarian cancer cell-lines and patient-derived tumor-spheroids. Gynecol. Oncol. 165, 129–136 (2022).
Di Veroli, G. Y. et al. Combenefit: An interactive platform for the analysis and visualization of drug combinations. Bioinformatics 32, 2866–2868 (2016).
Malyutina, A. et al. Drug combination sensitivity scoring facilitates the discovery of synergistic and efficacious drug combinations in cancer. PLoS Comput. Biol. 15, e1006752 (2019).
Nicoletti, I., Migliorati, G., Pagliacci, M. C., Grignani, F. & Riccardi, C. A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J. Immunol. Methods 139, 271–279 (1991).
Acknowledgements
We thank Kathrin Ausserlechner, Brigitte Greiderer-Kleinlercher, Petra Hechenberger, Stefanie Rainer, Sarah Ritscher, Martin Fleisch and the team of the High Throughput DNA Database Laboratory, Institute of Legal Medicine, Medical University of Innsbruck for their excellent technical assistance and Kaisa Huhtinen, Olli Carpén and Robert Zeillinger for providing cell lines.
Funding
This work was supported by the Verein zur Krebsforschung in der Frauenheilkunde.
Author information
Authors and Affiliations
Contributions
H.F. and C.M. developed the concept and designed the study. L.H., F.H., F.S., I.T., V.W., M.J.A., W.P, S.S., A.G.Z., C.M., and H.F. were involved in the data acquisition and quality control of the data and algorithms. L.H., F.H., F.S., I.T., V.W., M.J.A., W.P, A.G.Z., C.M., and H.F. analyzed and interpreted the data. L.H., F.H., F.S and H.F. performed statistical analyses and prepared the manuscript. L.H., F.H., F.S., I.T., V.W., M.J.A., W.P, S.S., A.G.Z., C.M., and H.F. edited the manuscript and reviewed the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Santer, F.R., Hovdar, L., Handle, F. et al. Absence of synergistic effects by CDK12/13 inhibition in combination with cisplatin or olaparib in ovarian cancer cells. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46634-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46634-3