Abstract
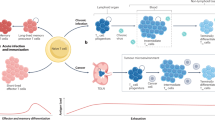
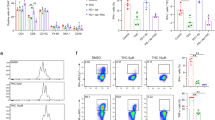
The prolonged interaction between the immune system and tumor antigens can result in T cell exhaustion. Extensive research has been conducted on strategies to reactivate exhausted T cells within the tumor microenvironment. However the exact contribution of the endocannabinoid system (ECS) and nociceptors in regulating CD8+ T cells within the framework of cancer-related inflammation has not been thoroughly studied. This study investigated the use of a TRPM8 antagonist (RQ-00203078), a selective cannabinoid receptor 1 (CB1) antagonist (AM251), and alpelisib (BYL-719) to control CD8+ T cell exhaustion. Our findings showed that administration of the CB1 antagonist AM251, either alone or in combination with alpelisib, significantly reduced the expression of PD-1 and Lag-3 on CD8+ T cells. Interestingly, treatment with the TRPM8 antagonist led to a notable increase in PD-1 expression on CD8+ T cells. These findings suggest that the decreased expression of inhibitory receptors on CD8+ T cells after treatment with the CB1 antagonist whether alone or with alpelisib and TRPM8 highlights the potential of ECS as a promising therapeutic target in cancer treatment.
Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article or its supplementary materials, and can also be obtained from the corresponding author upon request.
Abbreviations
- CNS:
-
Central nervous system
- ECS:
-
Endocannabinoid system
- CB1:
-
Cannabinoid receptor 1
- CB1 ant:
-
Cannabinoid receptor 1 antagonist
- TRPM8:
-
Transient receptor potential cation channel subfamily M member 8
- TRPM8 ant:
-
Transient receptor potential cation channel subfamily M member 8 antagonist
- PI3K:
-
Phosphoinositide 3-kinase
- PD-1:
-
Programmed cell death protein 1
- CTLA-4:
-
Cytotoxic T-lymphocyte-associated antigen 4
- Lag-3:
-
Lymphocyte activation gene 3
- TIM-3:
-
T cell immunoglobulin domain and mucin domain
- BTLA:
-
B- and T-lymphocyte attenuator
- TIGIT:
-
T cell immunoreceptor with immunoglobulin and ITIM domain
- AEA:
-
Anandamide
- 2-AG:
-
2-Arachidonoylglycerol
- ICBs:
-
Immune checkpoint inhibitors
- IHC:
-
Immunohistochemistry
References
Giles, J. R., Globig, A. M., Kaech, S. M. & Wherry, E. J. CD8(+) T cells in the cancer-immunity cycle. Immunity 56, 2231–2253. https://doi.org/10.1016/j.immuni.2023.09.005 (2023).
Napolitano, F. et al. CD8+ T cells in the tumor microenvironment modulate response to endocrine therapy in breast cancer. J. Clin. Invest. https://doi.org/10.1172/jci188458 (2025).
Han, K.-C. et al. Streamlined selection of cancer antigens for vaccine development through integrative multi-omics and high-content cell imaging. Sci. Rep. 10, 5885. https://doi.org/10.1038/s41598-020-62244-z (2020).
Hosonuma, M. & Yoshimura, K. Association between pH regulation of the tumor microenvironment and immunological state. Front. Oncol. 13, 1175563. https://doi.org/10.3389/fonc.2023.1175563 (2023).
Yang, W. et al. T-cell infiltration and its regulatory mechanisms in cancers: Insights at single-cell resolution. J. Exp. Clin. Cancer Res. 43, 38. https://doi.org/10.1186/s13046-024-02960-w (2024).
Li, X. et al. Targeting tumor innervation: Premises, promises, and challenges. Cell Death Discov. 8, 131. https://doi.org/10.1038/s41420-022-00930-9 (2022).
Mardelle, U., Bretaud, N., Daher, C. & Feuillet, V. From pain to tumor immunity: Influence of peripheral sensory neurons in cancer. Front. Immunol. 15, 1335387. https://doi.org/10.3389/fimmu.2024.1335387 (2024).
Matsueda, S., Chen, L., Li, H., Yao, H. & Yu, F. Recent clinical researches and technological development in TIL therapy. Cancer Immunol. Immunother. 73, 232. https://doi.org/10.1007/s00262-024-03793-4 (2024).
Koh, C.-H., Lee, S., Kwak, M., Kim, B.-S. & Chung, Y. CD8 T-cell subsets: Heterogeneity, functions, and therapeutic potential. Exp. Mol. Med. 55, 2287–2299. https://doi.org/10.1038/s12276-023-01105-x (2023).
Huang, Y., Jia, A., Wang, Y. & Liu, G. CD8+ T cell exhaustion in anti-tumour immunity: The new insights for cancer immunotherapy. Immunology 168, 30–48. https://doi.org/10.1111/imm.13588 (2023).
Turnis, M. E., Andrews, L. P. & Vignali, D. A. A. Inhibitory receptors as targets for cancer immunotherapy. Eur. J. Immunol. 45, 1892–1905. https://doi.org/10.1002/eji.201344413 (2015).
Xiang, S., Li, S. & Xu, J. Unravelling T cell exhaustion through co-inhibitory receptors and its transformative role in cancer immunotherapy. Clin. Transl. Med. 15, e70345. https://doi.org/10.1002/ctm2.70345 (2025).
Cheng, L. et al. Efficacy and safety of bispecific antibodies vs. immune checkpoint blockade combination therapy in cancer: A real-world comparison. Mol. Cancer 23, 77. https://doi.org/10.1186/s12943-024-01956-6 (2024).
Park, Y.-J., Kuen, D.-S. & Chung, Y. Future prospects of immune checkpoint blockade in cancer: From response prediction to overcoming resistance. Exp. Mol. Med. 50, 1–13. https://doi.org/10.1038/s12276-018-0130-1 (2018).
Yin, X., He, L. & Guo, Z. T-cell exhaustion in CAR-T-cell therapy and strategies to overcome it. Immunology 169, 400–411. https://doi.org/10.1111/imm.13642 (2023).
Kouro, T., Himuro, H. & Sasada, T. Exhaustion of CAR T cells: Potential causes and solutions. J. Transl. Med. 20, 239. https://doi.org/10.1186/s12967-022-03442-3 (2022).
Sousa-Valente, J., Andreou, A. P., Urban, L. & Nagy, I. Transient receptor potential ion channels in primary sensory neurons as targets for novel analgesics. Br. J. Pharmacol. 171, 2508–2527. https://doi.org/10.1111/bph.12532 (2014).
Koivisto, A.-P., Belvisi, M. G., Gaudet, R. & Szallasi, A. Advances in TRP channel drug discovery: From target validation to clinical studies. Nat. Rev. Drug Discov. 21, 41–59. https://doi.org/10.1038/s41573-021-00268-4 (2022).
Zhang, M. et al. TRP (transient receptor potential) ion channel family: Structures, biological functions and therapeutic interventions for diseases. Signal Transduct. Target. Ther. 8, 261. https://doi.org/10.1038/s41392-023-01464-x (2023).
Pertusa, M., Solorza, J. & Madrid, R. Molecular determinants of TRPM8 function: Key clues for a cool modulation. Front. Pharmacol. 14, 1213337. https://doi.org/10.3389/fphar.2023.1213337 (2023).
Turcotte, C., Chouinard, F., Lefebvre, J. S. & Flamand, N. Regulation of inflammation by cannabinoids, the endocannabinoids 2-arachidonoyl-glycerol and arachidonoyl-ethanolamide, and their metabolites. J. Leukoc. Biol. 97, 1049–1070. https://doi.org/10.1189/jlb.3RU0115-021R (2015).
Lutz, B. Neurobiology of cannabinoid receptor signaling. Dialogues Clin. Neurosci. 22, 207–222. https://doi.org/10.31887/DCNS.2020.22.3/blutz (2020).
Hinz, B. & Ramer, R. Cannabinoids as anticancer drugs: Current status of preclinical research. Br. J. Cancer. 127, 1–13. https://doi.org/10.1038/s41416-022-01727-4 (2022).
Wang, D. et al. Loss of cannabinoid receptor 1 accelerates intestinal tumor growth. Cancer Res. 68, 6468–6476. https://doi.org/10.1158/0008-5472.Can-08-0896 (2008).
Rahaman, O. & Ganguly, D. Endocannabinoids in immune regulation and immunopathologies. Immunology 164, 242–252. https://doi.org/10.1111/imm.13378 (2021).
Tanaka, K. et al. Gene expression of the endocannabinoid system in endometrium through menstrual cycle. Sci. Rep. 12, 9400. https://doi.org/10.1038/s41598-022-13488-4 (2022).
Wong, T.-S. et al. G protein-coupled receptors in neurodegenerative diseases and psychiatric disorders. Signal. Transduct. Target. Ther. 8, 177. https://doi.org/10.1038/s41392-023-01427-2 (2023).
Khunluck, T. et al. Activation of cannabinoid receptors in breast cancer cells improves osteoblast viability in cancer-bone interaction model while reducing breast cancer cell survival and migration. Sci. Rep. 12, 7398. https://doi.org/10.1038/s41598-022-11116-9 (2022).
Di Donato, M. et al. Therapeutic potential of TRPM8 antagonists in prostate cancer. Sci. Rep. 11, 23232. https://doi.org/10.1038/s41598-021-02675-4 (2021).
Chodon, D. et al. Estrogen regulation of TRPM8 expression in breast cancer cells. BMC Cancer 10, 212. https://doi.org/10.1186/1471-2407-10-212 (2010).
Chakravarti, B., Ravi, J. & Ganju, R. K. Cannabinoids as therapeutic agents in cancer: current status and future implications. Oncotarget 5, 5852–5872 (2014).
Mattioli, R. et al. Doxorubicin and other anthracyclines in cancers: Activity, chemoresistance and its overcoming. Mol. Aspects Med. 93, 101205. https://doi.org/10.1016/j.mam.2023.101205 (2023).
Khan, S. U., Fatima, K., Aisha, S. & Malik, F. Unveiling the mechanisms and challenges of cancer drug resistance. Cell. Commun. Signal. 22, 109. https://doi.org/10.1186/s12964-023-01302-1 (2024).
Emerling, B. M. & Akcakanat, A. Targeting PI3K/mTOR signaling in cancer. Cancer Res. 71, 7351–7359. https://doi.org/10.1158/0008-5472.Can-11-1699 (2011).
Zhang, H. P. et al. PI3K/AKT/mTOR signaling pathway: An important driver and therapeutic target in triple-negative breast cancer. Breast Cancer 31, 539–551. https://doi.org/10.1007/s12282-024-01567-5 (2024).
Takata, H. & Trautmann, L. Transforming dysfunctional CD8+ T cells into natural controller-like CD8+ T cells: Can TCF-1 be the magic wand?. J. Clin. Invest. https://doi.org/10.1172/jci160474 (2022).
Zhang, J., Lyu, T., Cao, Y. & Feng, H. Role of TCF-1 in differentiation, exhaustion, and memory of CD8+ T cells: A review. FASEB J. 35, e21549. https://doi.org/10.1096/fj.202002566R (2021).
Zuniga, E. I. & Harker, J. A. T-cell exhaustion due to persistent antigen: Quantity not quality?. Eur. J. Immunol. 42, 2285–2289. https://doi.org/10.1002/eji.201242852 (2012).
Șerban, M., Toader, C. & Covache-Busuioc, R.-A. The endocannabinoid system in human disease: Molecular signaling, receptor pharmacology, and therapeutic innovation. Int. J. Mol. Sci. 26, 11132 (2025).
Chow, A., Perica, K., Klebanoff, C. A. & Wolchok, J. D. Clinical implications of T cell exhaustion for cancer immunotherapy. Nat. Rev. Clin. Oncol. 19, 775–790. https://doi.org/10.1038/s41571-022-00689-z (2022).
Schillebeeckx, I. et al. T cell subtype profiling measures exhaustion and predicts anti-PD-1 response. Sci. Rep. 12, 1342. https://doi.org/10.1038/s41598-022-05474-7 (2022).
Woroniecka, K. et al. T-cell exhaustion signatures vary with tumor type and are severe in Glioblastoma. Clin. Cancer Res. 24, 4175–4186. https://doi.org/10.1158/1078-0432.Ccr-17-1846 (2018).
Poorebrahim, M. et al. Counteracting CAR T cell dysfunction. Oncogene 40, 421–435. https://doi.org/10.1038/s41388-020-01501-x (2021).
Catakovic, K., Klieser, E., Neureiter, D. & Geisberger, R. T cell exhaustion: From pathophysiological basics to tumor immunotherapy. Cell Commun. Signal. 15, 1. https://doi.org/10.1186/s12964-016-0160-z (2017).
Huang, A. C. & Zappasodi, R. A decade of checkpoint blockade immunotherapy in melanoma: Understanding the molecular basis for immune sensitivity and resistance. Nat. Immunol. 23, 660–670. https://doi.org/10.1038/s41590-022-01141-1 (2022).
Sun, Q. et al. Immune checkpoint therapy for solid tumours: Clinical dilemmas and future trends. Signal Transduct. Target. Ther. 8, 320. https://doi.org/10.1038/s41392-023-01522-4 (2023).
Catozzi, S. et al. Early morning immune checkpoint blockade and overall survival of patients with metastatic cancer: An in-depth chronotherapeutic study. Eur. J. Cancer https://doi.org/10.1016/j.ejca.2024.113571 (2024).
Farina, A. et al. Neurological adverse events of immune checkpoint inhibitors and the development of paraneoplastic neurological syndromes. Lancet Neurol. 23, 81–94. https://doi.org/10.1016/S1474-4422(23)00369-1 (2024).
Schnell, A., Bod, L., Madi, A. & Kuchroo, V. K. The yin and yang of co-inhibitory receptors: Toward anti-tumor immunity without autoimmunity. Cell Res. 30, 285–299. https://doi.org/10.1038/s41422-020-0277-x (2020).
Pinho-Ribeiro, F. A., Verri, W. A. Jr. & Chiu, I. M. Nociceptor sensory neuron-immune interactions in pain and inflammation. Trends Immunol. 38, 5–19. https://doi.org/10.1016/j.it.2016.10.001 (2017).
Pan, T. et al. Pan-cancer analyses reveal the genetic and pharmacogenomic landscape of transient receptor potential channels. Npj. Genom. Med. 7, 32. https://doi.org/10.1038/s41525-022-00304-1 (2022).
Wong, C. et al. 4E-BP1–dependent translation in nociceptors controls mechanical hypersensitivity via TRIM32/type I interferon signaling. Sci. Adv. 9, eadh9603. https://doi.org/10.1126/sciadv.adh9603 (2023).
Géranton, S. M. et al. A Rapamycin-sensitive signaling pathway is essential for the full expression of persistent pain states. J. Neurosci. 29, 15017–15027. https://doi.org/10.1523/jneurosci.3451-09.2009 (2009).
Wei, F. et al. Strength of PD-1 signaling differentially affects T-cell effector functions. Proc. Natl. Acad. Sci. USA 110, E2480-2489. https://doi.org/10.1073/pnas.1305394110 (2013).
Huang, H. et al. The immunomodulatory effects of endocrine therapy in breast cancer. J. Exp. Clin. Cancer Res. 40, 19. https://doi.org/10.1186/s13046-020-01788-4 (2021).
Siddiqui, I. et al. Intratumoral Tcf1+PD-1+CD8+ T cells with stem-like properties promote tumor control in response to vaccination and checkpoint blockade immunotherapy. Immunity 50, 195-211.e110. https://doi.org/10.1016/j.immuni.2018.12.021 (2019).
Ochoa, S. V., Casas, Z., Albarracín, S. L., Sutachan, J. J. & Torres, Y. P. Therapeutic potential of TRPM8 channels in cancer treatment. Front. Pharmacol. 14, 1098448. https://doi.org/10.3389/fphar.2023.1098448 (2023).
Pagano, E. et al. TRPM8 indicates poor prognosis in colorectal cancer patients and its pharmacological targeting reduces tumour growth in mice by inhibiting Wnt/β-catenin signalling. Br. J. Pharmacol. 180, 235–251. https://doi.org/10.1111/bph.15960 (2023).
Grolez, G. P. et al. Encapsulation of a TRPM8 agonist, WS12, in lipid nanocapsules potentiates PC3 prostate cancer cell migration inhibition through channel activation. Sci. Rep. 9, 7926. https://doi.org/10.1038/s41598-019-44452-4 (2019).
Le Fèvre, C. et al. Ellipsoid calculations versus manual tumor delineations for glioblastoma tumor volume evaluation. Sci. Rep. 12, 10502. https://doi.org/10.1038/s41598-022-13739-4 (2022).
Percie du Sert, N. et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS. Biol. 18, e3000410. https://doi.org/10.1371/journal.pbio.3000410 (2020).
Djoufack-Momo, S. M., Amparan, A. A., Grunden, B. & Boivin, G. P. Evaluation of carbon dioxide dissipation within a euthanasia chamber. J. Am. Assoc. Lab. Anim. Sci. 53, 404–407 (2014).
Funding
The authors did not receive any financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
A.M. conceptualized, designed, and conducted the majority of the experiments, performed statistical analyses, and authored the manuscript. S.D. made significant contributions to cell culture, cell processing, and animal handling tasks. A.T.M. assisted with software applications, analysis, visualization of data, and manuscript writing. A.A. provided consultation on drug preparations and partially drafted the manuscript related to the drug discussions and writing. R.M., a breast cancer surgeon, and A.A.P. offered valuable insights and critical revision feedback that enhanced the results and discussion sections. All authors participated in the editing process and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
To establish the 4T1-induced breast cancer model, it was confirmed that all animal care and experimental procedures conducted in this study adhered to the ethical guidelines approved by Urmia University of Medical Sciences, in alignment with ARRIVE guidelines (ARRIVE 2.0)61. All authors acknowledge the ethical considerations relevant to this manuscript. Ethical approval for this research was obtained from the Ethics Committee of Urmia University of Medical Sciences under approval code [IR.UMSU.AEC.1401.004]. Mice were maintained under standard housing conditions with a controlled temperature of 22°C, following a 12-h light/dark cycle. Food and water were available at all times, and appropriate air conditioning was implemented to minimize potential harm or distress. All necessary measures and precautions were employed to ensure the well-being of the animals. At the conclusion of the experiment, in accordance with IACUC guidelines, all euthanasia procedures for the mice were conducted using carbon dioxide (CO2) gas at a flow rate of 3 L/min until respiration ceased62. Prior to this procedure, the animal received anesthesia to minimize distress, which involved administering a mixture of ketamine (80 mg/kg) and xylazine (5 mg/kg) via intraperitoneal injection. Euthanasia was subsequently performed using CO₂ asphyxiation, followed by the cervical dislocation method, which involved placing a pen directly behind the ears at the base of the skull and, while lifting the base of the tail, dislocating the cervical vertebrae. In this study, female BALB/c mice, weighing between 21 and 23 g and aged 6–8 weeks, were obtained from the Pasteur Institute in Tehran, Iran. Upon reaching the logarithmic growth phase, 4T1 cell lines were trypsinized (using Gibco), resuspended, and a suspension containing 1 × 105 4T1 cells in 100 μL of sterile phosphate-buffered saline (PBS) was injected into the mammary fat pad of the mice. Eight days post-implantation, the mice were evaluated for tumor growth and were randomly assigned to treatment and control groups. The experimental groups consisted of eight categories: Control or Untreated, Alp (Alpelisib treated), TRPM8 ant (TRPM8 antagonist treated), CB1 ant (CB1 antagonist treated), Alp/CB1 ant (Alpelisib & CB1 antagonist treated), TRPM8/CB1 ant (TRPM8 & CB1 antagonist treated), Alp/TRPM8 ant (Alpelisib & TRPM8 antagonist treated), and Alp/CB1/TRPM8 ant (Alpelisib & CB1 & TRPM8 antagonist treated), with n = 5 mice in each group. In total, n = 50 mice were utilized for tumor induction, resulting in n = 4–5 mice in each experimental group. Mice that did not develop a tumor mass or exhibited a tumor mass significantly larger than the others by day 8 of induction were excluded from the study. Additionally, 4–5 mice were randomly allocated to each cage (Large cage Size (W × L × H), 7.5″ × 11.75″ × 5″) for each experimental group.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mohammadzadeh, A., Moazzendizzaji, S., Mohammadi, A.T. et al. Targeting CB1 and TRPM8 receptors to counteract CD8+ T cell exhaustion. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46794-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46794-2