Abstract
Background
The estimated plasma volume status (ePVS) is an independent risk factor for poor prognosis in certain cardiovascular disease populations. However, the relationship between ePVS and prognosis in critically ill patients with non-traumatic subarachnoid hemorrhage (NSAH) remains unclear. This study aims to explore the association between ePVS and all-cause mortality (ACM) in NSAH patients.
Methods
A total of 750 NSAH patients were included in this retrospective study. The primary outcome was 1-month ACM. A Cox proportional hazards model was constructed to examine the relationship between ePVS and ACM in critically ill NSAH patients. A restricted cubic spline model was used to assess the relationship between ePVS and the outcome.
Results
In this patient cohort, 1-month, 3-month, and 1-year ACM rates were 21%, 25%, and 29%, respectively. After adjusting for potential confounders, we found a U-shaped relationship between ePVS and 1-month ACM, with an inflection point at 3.94. Specifically, when ePVS is below the inflection point, each unit increase in ePVS reduces the mortality risk by 56% [HR, 0.44 (95% CI 0.25, 0.76)]. However, once the inflection point is surpassed, each unit increase in ePVS increases the mortality risk by 1.8 times [HR, 2.80 (95% CI 1.58, 4.97)].
Conclusion
We identified a U-shaped relationship between ePVS and ACM in critically ill patients with NSAH, suggesting both low and high ePVS levels may portend adverse outcomes. Prospective multicenter studies are warranted to validate these findings and elucidate underlying mechanisms.
Similar content being viewed by others
Introduction
Subarachnoid hemorrhage (SAH), primarily caused by ruptured aneurysms, is a severe cerebrovascular disorder accounting for approximately 5% of all strokes and is associated with high rates of disability and mortality1. In addition to advanced age, the severity of the initial hemorrhage—measured by tools such as the Hunt-Hess grade and Fisher score—and aneurysm characteristics such as location and size2, previous studies have identified secondary brain injuries as the primary contributors to poor clinical outcomes. These complications include rebleeding, delayed cerebral ischemia (DCI), and cerebral vasospasm3,4,5.
Optimal fluid management following SAH aims to enhance cerebral blood flow and oxygenation, playing a crucial role in preventing secondary brain injury, including delayed cerebral ischemia (DCI)6. Previous studies have indicated that both hypervolemia and hypovolemia are associated with poorer outcomes and higher complication rates. However, definitions of normal blood volume in the literature vary, and no clear standard exists. While the underlying fluid status is critical for managing critically ill patients, accurately estimating it remains a daily challenge7. The use of central venous pressure alone to evaluate volume status is unreliable8. Invasive measurements of cardiac output, including advanced hemodynamic monitoring via transurethral thermodilution, have been employed9–10. The additional significance of these monitoring techniques is still uncertain. Estimated plasma volume status (ePVS), a potential indicator of fluid balance, can be derived from routine blood counts using the Duarte formula, which is based on the Strauss formula11. Unlike invasive methods, this approach may offer a practical alternative for clinical use. As an alternative marker of blood volume, recent studies have reported an association between elevated ePVS levels and an increased risk of poor prognosis in patients with cardiac disease and intracerebral hemorrhage12,13,14. However, its value in patients with non-traumatic subarachnoid hemorrhage (NSAH) has yet to be explored.
Therefore, this study aimed to assess the potential relationship between ePVS and all-cause mortality (ACM) in critically ill patients with NSAH using the Medical Information Mart for Intensive Care IV (MIMIC-IV) database, providing insights for prognostic risk identification and fluid management in this population.
Materials and methods
Data source
This study employed a retrospective cohort design and utilized the MIMIC-IV database (version 2.2), which encompasses extensive clinical information on patients admitted to intensive care units (ICUs) at the Beth Israel Deaconess Medical Center (BIDMC) from 2008 to 2019. Data extraction was systematically performed using Navicat Premium (version 17). The principal investigator, Hui Shen, completed the “Protecting Human Research Participants” online course (ID: 1741944), thereby gaining authorization to access the MIMIC-IV database. The Institutional Review Board (IRB) at BIDMC granted a waiver for informed consent and approved the use of this resource for research purposes. As the study utilized publicly available and anonymized data, individual patient consent was not required. The manuscript was prepared in strict accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Criteria for population selection
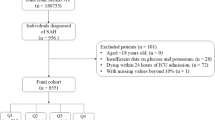
The retrospective cohort comprised all adult ICU patients diagnosed with NSAH, identified using ICD-9 code 430 and ICD-10 codes I60 from the MIMIC-IV database (version 2.2). The exclusion criteria were as follows: (1) ICU stays shorter than 24 h; (2) younger than 18 years; (3) non-first hospital and ICU admissions; (3) incomplete data, such as missing hematocrit or hemoglobin levels. A total of 750 patients were included in the study cohort. (Fig. 1).
Flow chart for patient selection in this study.
Data collection
Data on baseline characteristics within first 24 h of ICU admission were extracted from the MIMIC-IV 2.2 database. The extraction process covered six main components: (1)demographic variables, gender, age, race; (2)clinical severity, simplified acute physiology score (SAPS III), Glasgow Coma Scale (GCS), World Federation of Neurosurgical Society (WFNS), systemic inflammatory response syndrome (SIRS), sequential organ failure assessment (SOFA); (3)the mean values of vital signs, heart rate(HR), systolic blood pressure(SBP), diastolic blood pressure (DBP), mean blood pressure (MBP), respiratory rate (RR), temperature (temp), percutaneous oxygen saturation (SpO2);(4)the average laboratory results, hematocrit, hemoglobin(Hb), white blood cell(WBC), glucose, sodium, potassium; (5)comorbidities, Charlson comorbidity index(CCI), myocardial infarction(MI), chronic heart failure(CHF), chronic pulmonary disease(CPD), peripheral vascular disease(PVD), diabetes mellitus (DM), renal disease(RD), sepsis; and (6)treatments during ICU stay, the use of Statin, Nimodipine, or Nicardipine and mechanical ventilation (MV).
Estimated Plasma Volume Status
The Duarte formula incorporating hematocrit and Hb was utilized as follows:
ePVS = 100 × (1-hematocrit)/hemoglobin(g/dL). Hematocrit, and Hb were obtained directly within 24-hour on admission to ICU from MIMIC IV 2.2 database.
Clinical outcomes
The primary outcome was all-cause mortality (ACM) at 30 days. Secondary outcomes included ACM at 90 days and 1 year. Survival time was calculated from ICU admission to death from any cause, with censoring at the respective time points for survivors.
Statistical analysis
Continuous variables were analyzed using the Kruskal-Wallis test and are presented as medians with interquartile ranges (IQR). Categorical variables were assessed with Pearson’s Chi-squared test or Fisher’s exact test and are reported as absolute numbers with percentages (%). The distribution of ePVS was illustrated using a density plot. Restricted cubic splines (RCS) were applied to elucidate the U-shaped dose-response relationship with 1-month ACM. The optimal cutoff points for ePVS were determined by maximizing discriminative ability in a univariable Cox regression model using the R package ‘CatPredi’.
Kaplan-Meier (KM) curves were used to evaluate the incidence of primary and secondary outcomes, stratified by ePVS. The proportional-hazards assumption was preliminarily validated using Schoenfeld residuals; covariates with residual P-values ≥ 0.05 were deemed compliant and subsequently analyzed via Cox proportional hazards regression. To determine the independent predictive value of ePVS, two multivariate Cox models were constructed to adjust for potential confounders. Model 1 adjusted for age, race, and gender, while Model 2 adjusted for age, gender, race, SIRS, WFNS, MI, CHF, CPD, DM, sepsis, and MV. The trend P-value was calculated using Cox regression by entering the median value of each ePVS subgroup as a continuous variable in the model. A two-piece Cox proportional hazards model (“segmented” package) was used to further explore the relationship between ePVS and 1-month all-cause mortality, with separate analyses on either side of the inflection point. Additionally, stratified analysis was conducted based on gender (female, male), age (< 60 years or ≥ 60 years), race, WFNS, sepsis, and MV.
All statistical analyses were performed using R software (version 4.4.1), and P-values < 0.05 were considered statistically significant for two-sided tests.
Results
Baseline characteristics of participants
Initially, 1,176 patients were screened based on predefined diagnostic criteria for critical NSAH. After excluding those who were not initially admitted to the hospital or intensive care unit (ICU), those with an ICU stay of less than 24 h, and those with missing data, the final cohort consisted of 750 patients (Fig. 1).
The median ePVS across the cohort was 4.6 (IQR: 4.0–5.3), with the distribution shown in Fig. 2. RCS analysis revealed a U-shaped association between ePVS and 1-month all-cause mortality (Fig. 3). To further explore this relationship, the R package “CatPredi” was used to stratify the cohort into three groups: T1 (< 3.58), T2 (3.59–5.10), and T3 (≥ 5.11). Group T2 served as the reference category in subsequent analyses. The median ePVS values for each group were as follows: T1, 3.4 mL/g (IQR: 3.2–3.5); T2, 4.4 mL/g (IQR: 4.0–4.7); and T3, 5.9 mL/g (IQR: 5.4–6.8).
A density plot displaying the distribution of ePVS in the study.
Restricted cubic spline curve of the ePVS and 1-month all-cause mortality. ePVS, estimated plasma volume status.
Baseline demographic and clinical characteristics were stratified by ePVS groups (Table 1). Compared to patients in the other groups, those in the T2 group were generally younger, with lower APSIII and SOFA scores, as well as reduced heart rate (HR) and respiratory rate (RR). They also had a lower incidence of congestive heart failure (CHF), renal dysfunction (RD), and sepsis, along with a reduced all-cause mortality rate at 1 month, 3 months, and 1 year. Significant differences were found in gender, diastolic blood pressure (DBP), mean blood pressure (MBP), SpO₂, hematocrit, hemoglobin (Hb), white blood cell (WBC) count, glucose, potassium, Charlson comorbidity index (CCI), and treatment with nimodipine or nicardipine across the three groups (all P < 0.05).
Clinical outcomes
Kaplan-Meier (K-M) survival analysis was conducted to assess all-cause mortality (ACM) rates across different ePVS groups (Fig. 4). Individuals in the ePVS T2 group exhibited a significantly lower risk of mortality at 1 month, 3 months, and 1 year (all P < 0.001). Subsequently, Cox proportional hazards regression models were used to evaluate the relationship between ePVS and clinical outcomes (Table 2). The results indicated that ePVS was a significant predictor of 3-month mortality in both Model 1 (HR: 1.18; 95% CI: 1.06–1.31; P = 0.002) and Model 2 (HR: 1.16; 95% CI: 1.04–1.30; P = 0.01) when analyzed as a continuous variable. Similar results were observed for 1-year mortality (all P < 0.001), but no significant association was found for 1-month mortality (all P > 0.05).
Kaplan-Meier survival analysis curves for (A) 1-month, and (B) 3-month and (C)1-year all-cause mortality. ePVS, estimated plasma volume status. HR: hazard ratio; CI: confidence interval.
When ePVS was treated as an ordinal variable, patients in the T1 and T3 groups had a significantly higher risk of 1-month ACM compared to those in the T2 group. This increased risk was observed in Cox regression models for both Model 1 (T1 vs. T2: HR 2.38, 95% CI 1.51–3.75, P < 0.001; T3 vs. T2: HR 1.89, 95% CI 1.35–2.66, P < 0.001) and Model 2 (T1 vs. T2: HR 2.27, 95% CI 1.45–3.58, P < 0.001; T3 vs. T2: HR 1.79, 95% CI 1.27–2.53, P < 0.001). These findings suggest a U-shaped relationship between ePVS levels and mortality risk, which was also observed in multivariate Cox regression analyses for 3-month and 1-year ACM.
The two-piecewise Cox proportional hazards regression model identified an ePVS inflection point at 3.94 (P for log-likelihood ratio test = 0.004) (Table 3). When ePVS was below 3.94, there was a significant association with a decreased risk of 1-month mortality (HR: 0.44, 95% CI: 0.25–0.76, P = 0.004). Conversely, when ePVS exceeded 3.94, the association reversed, indicating an increased risk of mortality (HR: 2.80, 95% CI: 1.58–4.97, P < 0.001).
Subgroup analysis
To further explore the relationship between ePVS and 1-month ACM, stratified analyses were conducted based on gender, age (< 60 years or ≥ 60 years), race, WFNS score, presence of sepsis, and use of MV (Fig. 5). Compared to patients in the T2 group, those in the T1 and T3 groups who were male, aged ≥ 60 years, of any race, had a WFNS score of Ⅰ–Ⅲ, experienced sepsis, or required MV exhibited a significantly higher risk of 1-month ACM (all P < 0.05). However, no significant interactions were observed between these stratification variables and ePVS (all P for interaction > 0.05).
Subgroup analysis of the relationship between ePVS and 1-month all-cause mortality. ePVS. estimated plasma volume status.
Discussion
To our knowledge, this study is the first comprehensive investigation of the association between ePVS and mortality in critically ill patients with NSAH. The primary finding of this study is a unique U-shaped correlation between ePVS and ACM in this patient cohort. Specifically, both elevated and reduced ePVS values were significantly associated with increased ACM, as demonstrated in most of the selected subgroups.
To elucidate the dose-response relationship, we fitted a two-piecewise Cox regression model. Multivariable adjustment revealed an inflection point at ePVS = 3.94. In the left segment (ePVS < 3.94), each unit decrease was associated with reduced mortality, whereas in the right segment (ePVS > 3.94), each unit increase was associated with elevated mortality. This U-shaped association implies an optimal ePVS range in NSAH, with divergence from this range—whether toward lower or higher values—portending worse prognosis. These thresholds were derived empirically from the current dataset and require prospective validation before clinical implementation.
Initially, ePVS was used in patients with heart failure. Duarte et al. found that, in patients with heart failure and myocardial infarction, transient measurements of congestion and plasma volume derived from the Strauss formula predicted early cardiovascular events beyond conventional clinical evaluation11. Follow-up studies further demonstrated that ePVS is associated with early clinical outcomes in decompensated heart failure and may enhance risk stratification in heart failure patients13. In a retrospective cohort of 195 patients with advanced heart failure, Wu et al. reported that ePVS improved the predictive performance of hemodynamic parameters for clinical outcomes, although no direct association between ePVS and these parameters was observed15. The prognostic relevance of ePVS extends beyond cardiology. Recent investigations have demonstrated that ePVS is independently associated with heightened thrombosis risk and inferior survival outcomes in patients with primary myelofibrosis16. Surveys conducted in U.S. adults have found that elevated ePVS is strongly linked to increased all-cause, cardiovascular, and cancer-related mortality17. A retrospective study from Japan showed that ePVS was closely related to clinical outcomes in patients with lower limb arterial disease, with the prognostic ability for mortality and major adverse cardiovascular events (MALE) significantly improved by adding ePVS to basic predictors18. Additionally, a multicenter retrospective study by Shen et al. demonstrated a linear correlation between ePVS and 30-day or 1-year mortality in patients with intracerebral hemorrhage14. This study extends the above findings by examining the role of ePVS in patients with NSAH.
Fluid therapy constitutes a cornerstone of neurocritical care (NIC), encompassing both standard indications—volume resuscitation and maintenance of euvolemia—and neurospecificity applications, including intracranial pressure (ICP) management and prevention of delayed cerebral ischemia (DCI)6. Notably, the present study revealed a distinct U-shaped association between ePVS burden and mortality in patients with NSAH, which contrasts with the J-shaped relationships reported in prior investigations. This finding is consistent with established principles of fluid management in SAH. Specifically, diminished ePVS may reflect reduced effective circulating blood volume, thereby compromise cerebral perfusion pressure and precipitate hemodynamic instability secondary to cerebral vasospasm or elevated ICP19. Reduced blood volume may exacerbate DCI and secondary brain injury7–20. Furthermore, insufficient blood volume may impair renal perfusion, activate the renin-angiotensin-aldosterone system (RAAS) and worsen cerebral hypoxia21. On the other hand, high ePVS may lead to fluid overload, increasing the risk of heart failure or pulmonary edema. In many previous studies exploring the benefits of liberal fluid administration in aSAH patients—regarding DCI, cerebral infarction, mortality, or functional outcomes—pulmonary edema was more commonly observed in patients receiving liberal or high-volume fluid strategies, although none of these studies reported statistically significant findings22,23,24. In the volume management of NSAH patients, accurate assessment of circulatory blood volume is always a clinical challenge, whether the target is high or normal blood volume. Traditional methods, such as fluid balance calculations and CVP monitoring, have limited reliability and are associated with an increased risk of hypovolemia7. ePVS offers a rapid, inexpensive alternative derived from routine laboratory tests, with particular applicability in acute care. However, because ePVS is calculated from hemoglobin and hematocrit—parameters acutely modified by hemorrhage, transfusion, and fluid therapy—it may reflect acute disease severity or early interventions rather than authentic baseline volume status. This potential for reverse causality warrants careful interpretation of findings.
To elucidate the dose-response relationship, we fitted a two-piecewise Cox regression model. Multivariable adjustment revealed an inflection point at ePVS = 3.94. In the left segment (ePVS < 3.94), each unit decrease was associated with reduced mortality, whereas in the right segment (ePVS > 3.94), each unit increase was associated with elevated mortality. This U-shaped association implies an optimal ePVS range in NSAH, with divergence from this range—whether toward lower or higher values—portending worse prognosis. These thresholds were derived empirically from the current dataset and require prospective validation before clinical implementation.
Limitation
This study has several limitations. First, the absence of imaging validation for diagnostic codes may have resulted in misclassification of non-aneurysmal cases, introducing disease heterogeneity. Second, as a retrospective single-center observational study, our findings are susceptible to residual confounding and do not establish causality. Third, data derived from third-party public databases lacked critical clinical variables—including hematoma burden, hydrocephalus, intraventricular extension, DCI, and treatment modalities—potentially resulting in unmeasured confounding. Fourth, the severely ill nature of the cohort limits external validity to less acute NSAH populations. Finally, single-timepoint ePVS measurement precludes assessment of longitudinal changes; future studies should evaluate whether ePVS trajectories predict mortality independent of baseline values.
Conclusion
In conclusion, our study reveals a U-shaped association between ePVS and mortality risk in critically ill NSAH patients. Notably, both low and high ePVS are significantly associated with increased mortality in this specific patient population. This suggests that timely monitoring of ePVS levels and adjusting therapeutic strategies may help mitigate the potential risk of death in this group, emphasizing the critical importance of maintaining optimal blood volume control in this clinical context. Future large-scale, multicenter prospective studies are warranted to validate these observations and refine clinical strategies for improving outcomes in this vulnerable patient group.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Claassen, J. & Park, S. Spontaneous subarachnoid haemorrhage. Lancet 400 (10355), 846–862. https://doi.org/10.1016/S0140-6736(22)00938-2 (2022).
Yang, Y. et al. Association between Braden Scale and all-cause mortality in critically ill patients with non-traumatic subarachnoid hemorrhage: analysis of the MIMIC-IV database. Neurosurg. Rev. 48 (1), 345. https://doi.org/10.1007/s10143-025-03508-y (2025). Published 2025 Apr 2.
Brilstra, E. H., Rinkel, G. J., Algra, A. & van Gijn, J. Rebleeding, secondary ischemia, and timing of operation in patients with subarachnoid hemorrhage. Neurology 55 (11), 1656–1660. https://doi.org/10.1212/wnl.55.11.1656 (2000).
Macdonald, R. L. et al. Clazosentan, an endothelin receptor antagonist, in patients with aneurysmal subarachnoid haemorrhage undergoing surgical clipping: a randomised, double-blind, placebo-controlled phase 3 trial (CONSCIOUS-2). Lancet Neurol. 10 (7), 618–625. https://doi.org/10.1016/S1474-4422(11)70108-9 (2011).
Macdonald, R. L. Origins of the Concept of Vasospasm. Stroke 47 (1), e11–e15. https://doi.org/10.1161/STROKEAHA.114.006498 (2016).
Oddo, M. et al. Fluid therapy in neurointensive care patients: ESICM consensus and clinical practice recommendations. Intensive Care Med. 44 (4), 449–463. https://doi.org/10.1007/s00134-018-5086-z (2018).
Hoh, B. L. et al. 2023 Guideline for the Management of Patients With Aneurysmal Subarachnoid Hemorrhage: A Guideline From the American Heart Association/American Stroke Association [published correction appears in Stroke. ;54(12):e516. (2023). https://doi.org/10.1161/STR.0000000000000449.]. Stroke. 2023;54(7):e314-e370. doi:10.1161/STR.0000000000000436.
Gress, DR & Participants in the International Multi-Disciplinary Consensus Conference on the Critical Care Management of Subarachnoid Hemorrhage. Monitoring of volume status after subarachnoid hemorrhage. Neurocrit Care. 15 (2), 270–274. https://doi.org/10.1007/s12028-011-9604-x (2011).
Mutoh, T. et al. Early intensive versus minimally invasive approach to postoperative hemodynamic management after subarachnoid hemorrhage. Stroke 45 (5), 1280–1284. https://doi.org/10.1161/STROKEAHA.114.004739 (2014).
Anetsberger, A. et al. Impact of Goal-Directed Therapy on Delayed Ischemia After Aneurysmal Subarachnoid Hemorrhage: Randomized Controlled Trial [published correction appears in Stroke. ;51(9):e272. doi: 10.1161/STR.0000000000000348.]. Stroke. 2020;51(8):2287–2296. (2020). https://doi.org/10.1161/STROKEAHA.120.029279
Duarte, K. et al. Prognostic Value of Estimated Plasma Volume in Heart Failure. JACC Heart Fail. 3 (11), 886–893. https://doi.org/10.1016/j.jchf.2015.06.014 (2015).
Kobayashi, M. et al. Estimated plasma volume status in heart failure: clinical implications and future directions. Clin. Res. Cardiol. 110 (8), 1159–1172. https://doi.org/10.1007/s00392-020-01794-8 (2021).
Fudim, M. et al. Plasma Volume Status and Its Association With In-Hospital and Postdischarge Outcomes in Decompensated Heart Failure. J. Card Fail. 27 (3), 297–308. https://doi.org/10.1016/j.cardfail.2020.09.478 (2021).
Shen, Y. et al. Associations of estimated plasma volume status with 30-day mortality and 1-year mortality in patients with intracerebral hemorrhage: a study of the MIMIC-IV database and the hospital information system. Front. Neurol. 16, 1548064. https://doi.org/10.3389/fneur.2025.1548064 (2025). Published 2025 Apr 1.
Wu, Y. et al. Estimated plasma volume status adds prognostic value to hemodynamic parameters in advanced heart failure. Intern. Emerg. Med. 18 (8), 2281–2291. https://doi.org/10.1007/s11739-023-03422-5 (2023).
Lucijanic, M. et al. Higher estimated plasma volume status is associated with increased thrombotic risk and impaired survival in patients with primary myelofibrosis. Biochem. Med. (Zagreb). 33 (2), 020901. https://doi.org/10.11613/BM.2023.020901 (2023).
Marawan, A. & Qayyum, R. Estimated plasma volume and mortality: analysis from NHANES 1999–2014. Clin. Res. Cardiol. 109 (9), 1148–1154. https://doi.org/10.1007/s00392-020-01606-z (2020).
Kurokawa, T. et al. Impact of estimated plasma volume status on clinical outcomes in patients with lower extremity artery disease who underwent endovascular therapy. Hypertens. Res. 46 (8), 2005–2015. https://doi.org/10.1038/s41440-023-01315-w (2023).
Dhar, R. et al. Relationship between angiographic vasospasm and regional hypoperfusion in aneurysmal subarachnoid hemorrhage. Stroke 43 (7), 1788–1794. https://doi.org/10.1161/STROKEAHA.111.646836 (2012).
Labib, H. et al. Sodium and Its Impact on Outcome After Aneurysmal Subarachnoid Hemorrhage in Patients With and Without Delayed Cerebral Ischemia. Crit. Care Med. 52 (5), 752–763. https://doi.org/10.1097/CCM.0000000000006182 (2024).
Yang, X. et al. Association between estimated plasma volume status and acute kidney injury in patients who underwent coronary revascularization: A retrospective cohort study from the MIMIC-IV database. PLoS One. 19 (6), e0300656. https://doi.org/10.1371/journal.pone.0300656 (2024). Published 2024 Jun 12.
Mayer, S. A. et al. Effect of 5% albumin solution on sodium balance and blood volume after subarachnoid hemorrhage. Neurosurgery 42 (4), 759–768. https://doi.org/10.1097/00006123-199804000-00048 (1998).
Lennihan, L. et al. Effect of hypervolemic therapy on cerebral blood flow after subarachnoid hemorrhage: a randomized controlled trial. Stroke 31 (2), 383–391. https://doi.org/10.1161/01.str.31.2.383 (2000).
Egge, A. et al. Prophylactic hyperdynamic postoperative fluid therapy after aneurysmal subarachnoid hemorrhage: a clinical, prospective, randomized, controlled study. Neurosurgery 49 (3), 593–606. https://doi.org/10.1097/00006123-200109000-00012 (2001).
Acknowledgements
None.
Funding
This work was supported by Beijing Natural Science Foundation (7222053), China National Natural Science Foundation of (82003134) and Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-009 A).
Author information
Authors and Affiliations
Contributions
HS conceived the study and participated in the design of the study. QM and HS collected the data, JZ and HS analyzed and interpreted the data. QM and HS drafted and edited the manuscript. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript *.*.
Corresponding author
Ethics declarations
Consent for publication
All authors of the manuscript agreed to its publication.
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
Not applicable.
Clinical trial number
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mei, Q., Zhang, J. & Shen, H. Association between estimated plasma volume status and all-cause mortality in critically ill patients with non-traumatic subarachnoid hemorrhage: analysis of the MIMIC-IV database. Sci Rep 16, 11725 (2026). https://doi.org/10.1038/s41598-026-47116-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-47116-2