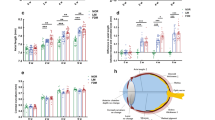
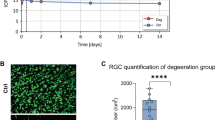
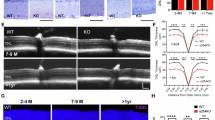
Abstract
The pathological mechanism underlying retinal apoptosis in X-linked retinoschisis (XLRS), a disease caused by retinoschisin 1 (RS1) deficiency, remains incompletely understood. This study aimed to investigate the role of transient receptor potential melastatin 1 (TRPM1) in retinal tissues and cells. Retinal function and structure were assessed by electroretinography (ERG) and optical coherence tomography (OCT). Protein expression was evaluated by immunofluorescence staining and western blotting (WB). Retinal morphology was examined by hematoxylin and eosin (H&E) staining. Apoptotic retinal cells were detected by TUNEL staining. Key proteins were screened using proteomics data obtained by mass spectrometry. Intracellular calcium levels were measured using Rhod-2 AM. TRPM1 expression in Rs1-KO mice was 1.3-fold higher than that in wild-type mice (p < 0.05), whereas TRPM1-overexpressing ARPE19 cells exhibited approximately twofold higher expression than the empty vector control group. Mechanistically, TRPM1-mediated calcium influx promoted calcium/calmodulin-dependent protein kinase II (CAMKII) phosphorylation. Concomitantly, the accumulation of the autophagy-related proteins P62 and LC3B, increased BAX expression, and decreased BCL2 expression were observed in both Rs1-KO retinal tissues and TRPM1-overexpressing ARPE19 cells. These findings collectively suggest that TRPM1 may contribute to cell’s apoptosis. Our study provides new insight into the mechanism of retinal apoptosis in XLRS.
Similar content being viewed by others
Data availability
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) via the iProX partner repository with the dataset identifier PXD031901.
References
Wu, W. W., Wong, J. P., Kast, J. & Molday, R. S. RS1, a discoidin domain-containing retinal cell adhesion protein associated with X-linked retinoschisis, exists as a novel disulfide-linked octamer. J. Biol. Chem. 280, 10721–10730. https://doi.org/10.1074/jbc.M413117200 (2005).
Sergeev, Y. V. et al. Molecular modeling of retinoschisin with functional analysis of pathogenic mutations from human X-linked retinoschisis. Hum. Mol. Genet. 19, 1302–1313. https://doi.org/10.1093/hmg/ddq006 (2010).
Reid, S. N. et al. The mouse X-linked juvenile retinoschisis cDNA: expression in photoreceptors. Gene 227, 257–266. https://doi.org/10.1016/s0378-1119(98)00578-2 (1999).
Reid, S. N., Yamashita, C. & Farber, D. B. Retinoschisin, a photoreceptor-secreted protein, and its interaction with bipolar and muller cells. J. neuroscience: official J. Soc. Neurosci. 23, 6030–6040. https://doi.org/10.1523/JNEUROSCI.23-14-06030.2003 (2003).
Reid, S. N. & Farber, D. B. Glial transcytosis of a photoreceptor-secreted signaling protein, retinoschisin. Glia 49, 397–406. https://doi.org/10.1002/glia.20131 (2005).
Vijayasarathy, C., Zeng, Y., Brooks, M. J., Fariss, R. N. & Sieving, P. A. Genetic Rescue of X-Linked Retinoschisis Mouse (Rs1(-/y)) Retina Induces Quiescence of the Retinal Microglial Inflammatory State Following AAV8-RS1 Gene Transfer and Identifies Gene Networks Underlying Retinal Recovery. Hum. Gene Ther. 32, 667–681. https://doi.org/10.1089/hum.2020.213 (2021).
Guo, Q. et al. Phenotype Heterogeneity and the Association Between Visual Acuity and Outer Retinal Structure in a Cohort of Chinese X-Linked Juvenile Retinoschisis Patients. Front. Genet. 13, 832814. https://doi.org/10.3389/fgene.2022.832814 (2022).
Jin, X. et al. Retinal Proteomic Alterations and Combined Transcriptomic-Proteomic Analysis in the Early Stages of Progression of a Mouse Model of X-Linked Retinoschisis. Cells 11, (2022). https://doi.org/10.3390/cells11142150
Liu, M. et al. Longitudinal Photoreceptor Phenotype Observation and Therapeutic Evaluation of a Carbonic Anhydrase Inhibitor in a X-Linked Retinoschisis Mouse Model. Front. Med. (Lausanne). 9, 886947. https://doi.org/10.3389/fmed.2022.886947 (2022).
Takada, Y. et al. Synaptic pathology in retinoschisis knockout (Rs1-/y) mouse retina and modification by rAAV-Rs1 gene delivery. Investig. Ophthalmol. Vis. Sci. 49, 3677–3686. https://doi.org/10.1167/iovs.07-1071 (2008).
Ziccardi, L., Vijayasarathy, C., Bush, R. A. & Sieving, P. A. Loss of retinoschisin (RS1) cell surface protein in maturing mouse rod photoreceptors elevates the luminance threshold for light-driven translocation of transducin but not arrestin. J. neuroscience: official J. Soc. Neurosci. 32, 13010–13021. https://doi.org/10.1523/JNEUROSCI.1913-12.2012 (2012).
Ou, J. et al. Synaptic pathology and therapeutic repair in adult retinoschisis mouse by AAV-RS1 transfer. J. Clin. Invest. 125, 2891–2903. https://doi.org/10.1172/JCI81380 (2015).
Bush, R. A. et al. Preclinical Dose-Escalation Study of Intravitreal AAV-RS1 Gene Therapy in a Mouse Model of X-linked Retinoschisis: Dose-Dependent Expression and Improved Retinal Structure and Function. Hum. Gene Ther. 27, 376–389. https://doi.org/10.1089/hum.2015.142 (2016).
Vijayasarathy, C., Pasha, S., Sieving, P. A. & S. P. B. & Of men and mice: Human X-linked retinoschisis and fidelity in mouse modeling. Prog Retin Eye Res. 87, 100999. https://doi.org/10.1016/j.preteyeres.2021.100999 (2022).
Ye, E. A. et al. XLRS Rat with Rs1-/Y Exon-1-Del Shows Failure of Early Postnatal Outer Retina Development. Genes 13 https://doi.org/10.3390/genes13111995 (2022).
Cukras, C. et al. Retinal AAV8-RS1 Gene Therapy for X-Linked Retinoschisis: Initial Findings from a Phase I/IIa Trial by Intravitreal Delivery. Mol. Ther. 26, 2282–2294. https://doi.org/10.1016/j.ymthe.2018.05.025 (2018).
Wang, W. et al. Intravitreal Injection of an Exosome-Associated Adeno-Associated Viral Vector Enhances Retinoschisin 1 Gene Transduction in the Mouse Retina. Hum. Gene Ther. 32, 707–716. https://doi.org/10.1089/hum.2020.328 (2021).
van der Veen, I. et al. The Road towards Gene Therapy for X-Linked Juvenile Retinoschisis: A Systematic Review of Preclinical Gene Therapy in Cell-Based and Rodent Models of XLRS. Int. J. Mol. Sci. 25 https://doi.org/10.3390/ijms25021267 (2024).
Molday, R. S., Kellner, U. & Weber, B. H. X-linked juvenile retinoschisis: clinical diagnosis, genetic analysis, and molecular mechanisms. Prog Retin Eye Res. 31, 195–212. https://doi.org/10.1016/j.preteyeres.2011.12.002 (2012).
Klooster, J. et al. Ultrastructural localization and expression of TRPM1 in the human retina. Investig. Ophthalmol. Vis. Sci. 52, 8356–8362. https://doi.org/10.1167/iovs.11-7575 (2011).
Xu, Y. et al. mGluR6 deletion renders the TRPM1 channel in retina inactive. J. Neurophysiol. 107, 948–957. https://doi.org/10.1152/jn.00933.2011 (2012).
Agosto, M. A., Anastassov, I. A. & Wensel, T. G. Differential epitope masking reveals synapse-specific complexes of TRPM1. Vis. Neurosci. 35, E001. https://doi.org/10.1017/S0952523817000360 (2018).
Devi, S. et al. Calcium homeostasis in human melanocytes: role of transient receptor potential melastatin 1 (TRPM1) and its regulation by ultraviolet light. Am. J. Physiol. Cell. Physiol. 297, C679–C687. https://doi.org/10.1152/ajpcell.00092.2009 (2009).
Devi, S. et al. Metabotropic glutamate receptor 6 signaling enhances TRPM1 calcium channel function and increases melanin content in human melanocytes. Pigment Cell. Melanoma Res. 26, 348–356. https://doi.org/10.1111/pcmr.12083 (2013).
Peachey, N. S. et al. Depolarizing bipolar cell dysfunction due to a Trpm1 point mutation. J. Neurophysiol. 108, 2442–2451. https://doi.org/10.1152/jn.00137.2012 (2012).
Takeuchi, H. et al. Different Activity Patterns in Retinal Ganglion Cells of TRPM1 and mGluR6 Knockout Mice. Biomed. Res. Int. 2018 (2963232). https://doi.org/10.1155/2018/2963232 (2018).
Xu, X. Z., Moebius, F., Gill, D. L. & Montell, C. Regulation of melastatin, a TRP-related protein, through interaction with a cytoplasmic isoform. Proc. Natl. Acad. Sci. U S A. 98, 10692–10697. https://doi.org/10.1073/pnas.191360198 (2001).
Liu, F. S. et al. Ca(2+) Regulates Autophagy Through CaMKKbeta/AMPK/mTOR Signaling Pathway in Mechanical Spinal cord Injury: An in vitro Study. Neurochem Res. 48, 447–457. https://doi.org/10.1007/s11064-022-03768-w (2023).
Hsieh, C. C. et al. TRPM1 promotes tumor progression in acral melanoma by activating the Ca(2+)/CaMKIIδ/AKT pathway. J. Adv. Res. 43, 45–57. https://doi.org/10.1016/j.jare.2022.03.005 (2023).
Zhang, Y. B., Gong, J. L., Xing, T. Y., Zheng, S. P. & Ding, W. Autophagy protein p62/SQSTM1 is involved in HAMLET-induced cell death by modulating apotosis in U87MG cells. Cell. Death Dis. 4, e550. https://doi.org/10.1038/cddis.2013.77 (2013).
Wang, Q. et al. Mechanistic study of TRPM2-Ca(2+)-CAMK2-BECN1 signaling in oxidative stress-induced autophagy inhibition. Autophagy 12, 1340–1354. https://doi.org/10.1080/15548627.2016.1187365 (2016).
Plössl, K. et al. Retinoschisin is linked to retinal Na/K-ATPase signaling and localization. Mol. Biol. Cell. 28, 2178–2189. https://doi.org/10.1091/mbc.E17-01-0064 (2017).
Plössl, K., Weber, B. H. & Friedrich, U. The X-linked juvenile retinoschisis protein retinoschisin is a novel regulator of mitogen-activated protein kinase signalling and apoptosis in the retina. J. Cell. Mol. Med. 21, 768–780. https://doi.org/10.1111/jcmm.13019 (2017).
Gayet-Primo, J. & Puthussery, T. Alterations in Kainate Receptor and TRPM1 Localization in Bipolar Cells after Retinal Photoreceptor Degeneration. Front. Cell. Neurosci. 9, 486. https://doi.org/10.3389/fncel.2015.00486 (2015).
Lambert, S. et al. Transient receptor potential melastatin 1 (TRPM1) is an ion-conducting plasma membrane channel inhibited by zinc ions. J. Biol. Chem. 286, 12221–12233. https://doi.org/10.1074/jbc.M110.202945 (2011).
Bond, K. et al. Double-Stranded RNA Induces Retinal Pigment Epithelium Cell Degeneration and Inflammation. FASEB J. 39, e70786. https://doi.org/10.1096/fj.202402181R (2025).
Agosto, M. A., Adeosun, A. A. R., Kumar, N. & Wensel, T. G. The mGluR6 ligand-binding domain, but not the C-terminal domain, is required for synaptic localization in retinal ON-bipolar cells. J. Biol. Chem. 297, 101418. https://doi.org/10.1016/j.jbc.2021.101418 (2021).
Koike, C. et al. TRPM1 is a component of the retinal ON bipolar cell transduction channel in the mGluR6 cascade. Proc. Natl. Acad. Sci. U S A. 107, 332–337. https://doi.org/10.1073/pnas.0912730107 (2010).
Zhang, X. et al. Mechanisms underlying morphological and functional changes of cilia in fibroblasts derived from patients bearing ARL3(T31A) and ARL3(T31A/C118F) mutations. FASEB J. 38, e23519. https://doi.org/10.1096/fj.202301906R (2024).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 82402170, funder: W.P.W.; No. 82271084, funder: B.L.). The Youth Basic Research Project of Henan Eye Hospital (No. 23JCQN001, funder: W.P.W.). The Henan Provincial Medical Science and Technology Tackling Program (No. LHGJ20220087, funder: W.P.W.). Henan Academy of Innovations in Medical Science Eye Institute Applied Research Special Project (No. 20250301, funder: W.P.W.; No. 20250203, funder: X.X.J.). Henan Academy of Innovations in Medical Science Basic Research Operations Program (No.JBKY250311, funder: X.X.J.).
Author information
Authors and Affiliations
Contributions
W.P.W. and B.L. conceived and supervised the study. W.P.W. and J.Y.L. designed the methodology and performed validation experiments. W.P.W. and X.X.J. conducted the formal analysis. W.P.W., M.Z.Y., and R.Q.Q. performed the investigation. S.Y. provided resources. G.M.L. and M.Y.Q. curated the data. W.P.W. wrote the main manuscript text and prepared the figures. B.L. reviewed and edited the manuscript. W.P.W., X.X.J., and B.L. acquired funding. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, W., Liu, J., Jin, X. et al. Upregulated TRPM1 is associated with apoptosis in Rs1 knockout mice and in ARPE19 cells through increased intracellular calcium. Sci Rep (2026). https://doi.org/10.1038/s41598-026-47523-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-47523-5