Abstract
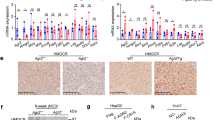
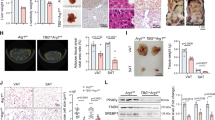
The quantity and quality of dietary protein profoundly influence satiety, body growth, and systemic metabolism. Among the 20 major proteinogenic amino acids, arginine (Arg) is considerably associated with hepatic steatosis; when animals fed an Arg-deficient diet (ΔArg), triacylglyceride (TAG) dramatically accumulates in the liver. To explore the underlying mechanism, we first investigated the role of ornithine (Orn), as Orn is the primary metabolite of Arg and it is reportedly involved in the regulation of liver metabolism. While male Wistar rats fed a ΔArg diet exhibited a significant increase in liver TAG levels due to an attenuated TAG secretion, and consistently marked reduction of TAG-rich lipoproteins in the circulation, Orn addition to the diet completely abolished all these metabolic changes. Orn was only effective when taken orally, but not through intraperitoneal administration, suggesting that the intestine plays an essential role for Orn to regulate liver metabolism. The metabolic features similar to those of our rat model was also observed in the analyses of clinical samples, implying the common mechanism in humans. Conclusively, dietary Arg deficiency lowers local Arg-to-Orn conversion in the intestine, which in turn inhibits hepatic lipid secretion remotely via gut-liver axis.
Similar content being viewed by others
Data availability
Raw 16 S ribosomal DNA amplicon sequencing data related to Supplementary Fig 5 are available in the DDBJ BioProject database with links to BioProject accession number #PRJDB40630. The human datasets generated and/or analyzed in this study are not publicly available for protecting personal privacy, but are available upon reasonable request. The other data described in this manuscript and the analytical code will be made available upon reasonable request. Further information and requests should be directed to and will be fulfilled by the lead contact, Hiroki Nishi ([shwest@mail.saitama-u.ac.jp](mailto: shwest@mail.saitama-u.ac.jp)).
References
Hyde, P. N. et al. Dietary carbohydrate restriction improves metabolic syndrome independent of weight loss. JCI Insight 4, (2019).
Mardinoglu, A. et al. An integrated understanding of the rapid metabolic benefits of a carbohydrate-restricted diet on hepatic steatosis in humans. Cell. Metab. 27, 559–571e5 (2018).
Gao, Y., Hua, R., Hu, K. & Wang, Z. Carbohydrates deteriorate fatty liver by activating the inflammatory response. Nutr. Res. Rev. 35, 252–267 (2022).
Yki-Järvinen, H., Luukkonen, P. K., Hodson, L. & Moore, J. B. Dietary carbohydrates and fats in nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 18, 770–786 (2021).
Lyu, J. et al. Protein source associated with risk of metabolic syndrome in adults with low and adequate protein intake: A prospective cohort study of middle-aged and older adults. J. Nutr. Health Aging. 28, (2024).
Nishi, H. et al. Metabolic effects of short-term high-fat intake vary depending on dietary amino acid composition. Curr. Dev. Nutr. 8, 103768 (2024).
Hu, X. & Guo, F. Amino acid sensing in metabolic homeostasis and health. Endocr. Rev. 42, 56–76 (2021).
Taya, Y. et al. Depleting dietary valine permits nonmyeloablative mouse hematopoietic stem cell transplantation. Sci. (1979). 354, 1152–1155 (2016).
He, L., Ding, Y., Zhou, X., Li, T. & Yin, Y. Serine signaling governs metabolic homeostasis and health. Trends Endocrinol. Metab. 34, 361–372 (2023).
Kosakamoto, H. et al. Sensing of the non-essential amino acid tyrosine governs the response to protein restriction in Drosophila. Nat. Metab. 4, 944–959 (2022).
Nishi, H. et al. Importance of serum amino acid profile for induction of hepatic steatosis under protein malnutrition. Sci. Rep. 8, 1–11 (2018).
Otani, L. et al. Low-arginine and low-protein diets induce hepatic lipid accumulation through different mechanisms in growing rats. Nutr. Metab. (Lond). 17, 1–13 (2020).
Jenkinson, C. P., Grody, W. W. & Cederbaum, S. D. Comparative properties of arginases. Biochem. Physiol. 114 (1996).
Canbay, A. & Sowa, J. P. l-Ornithine l-Aspartate (LOLA) as a novel approach for therapy of non-alcoholic fatty liver disease. Drugs. 79, 39–44. https://doi.org/10.1007/s40265-018-1020-5 (2019).
Vela, C. I. B. & Ramírez, J. L. P. Efficacy of oral L-ornithine L-aspartate in cirrhotic patients with hyperammonemic hepatic encephalopathy. Ann. Hepatol. 10, (2011).
Fukushima, S. et al. A novel amino acid signaling process governs glucose-6-phosphatase transcription. iScience 24, 102778 (2021).
Olofsson, S. O., Boren, J. & Apolipoprotein, B. A clinically important apolipoprotein which assembles atherogenic lipoproteins and promotes the development of atherosclerosis. J. Intern. Med. 258, 395–410 (2005).
Wang, S. et al. IRE1α-XBP1s induces PDI expression to increase MTP activity for hepatic VLDL assembly and lipid homeostasis. Cell. Metab. 16, 473–486 (2012).
Wetterau, J. R., Combs, K. A., McLean, L. R., Spinner, S. N. & Aggerbeck, L. P. Protein disulfide isomerase appears necessary to maintain the catalytically active structure of the microsomal triglyceride transfer protein. Biochemistry 30, 9728–9735 (1991).
Wang, X. et al. Receptor-mediated ER export of lipoproteins controls lipid homeostasis in mice and humans. Cell. Metab. 33, 350–366e7 (2021).
Albillos, A., De Gottardi, A. & Rescigno, M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J. Hepatol. 72, 558–577 (2020).
Pabst, O. et al. Gut–liver axis: Barriers and functional circuits. Nat. Rev. Gastroenterol. Hepatol. 20, 447–461 (2023).
Hsu, C. L. & Schnabl, B. The gut–liver axis and gut microbiota in health and liver disease. Nat. Rev. Microbiol. 21, 719–733 (2023).
Zhong, J. et al. Hyodeoxycholic acid ameliorates nonalcoholic fatty liver disease by inhibiting RAN-mediated PPARα nucleus-cytoplasm shuttling. Nat. Commun. 14, (2023).
Saxton, R. A. & Sabatini, D. M. mTOR signaling in growth, metabolism, and disease. Cell. 168, 960–976. https://doi.org/10.1016/j.cell.2017.02.004 (2017).
Goul, C., Peruzzo, R. & Zoncu, R. The molecular basis of nutrient sensing and signalling by mTORC1 in metabolism regulation and disease. Nat. Rev. Mol. Cell Biol. 24, 857–875. https://doi.org/10.1038/s41580-023-00641-8 (2023).
Rodríguez-Correa, E., González-Pérez, I., Clavel-Pérez, P. I., Contreras-Vargas, Y. & Carvajal, K. Biochemical and nutritional overview of diet-induced metabolic syndrome models in rats: What is the best choice? Nutr. Diabetes 10, (2020).
Bays, H. E. et al. Obesity, dyslipidemia, and cardiovascular disease: A joint expert review from the Obesity Medicine Association and the National Lipid Association 2024. J. Clin. Lipidol. 18, e320–e350 (2024).
Yanai, H., Adachi, H., Hakoshima, M., Iida, S. & Katsuyama, H. Metabolic-dysfunction-associated steatotic liver disease—Its pathophysiology, association with atherosclerosis and cardiovascular disease, and treatments. Int J. Mol. Sci. 24, (2023).
Deprince, A., Haas, J. T. & Staels, B. Dysregulated lipid metabolism links NAFLD to cardiovascular disease. Mol. Metab 42, (2020).
Nishi, H. et al. Alteration of serum amino acid profiles by dietary adenine supplementation inhibits fatty liver development in rats. Sci. Rep. 10, 1–10 (2020).
Yamakado, M. et al. Plasma free amino acid profiles predict four-year risk of developing diabetes, metabolic syndrome, dyslipidemia, and hypertension in Japanese population. Sci. Rep. 5, (2015).
Chantranupong, L. et al. The CASTOR proteins are arginine sensors for the mTORC1 pathway. Cell. 165, 153–164 (2016).
Igarashi, M. & Guarente, L. mTORC1 and SIRT1 cooperate to foster expansion of gut adult stem cells during calorie restriction. Cell 166, 436–450 (2016).
Folch, J., Lees, M. & Sloane Stanley, G. H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 55, 999–1033 (1957).
Athar, H., Iqbal, J., Jiang, X. C. & Hussain, M. M. A simple, rapid, and sensitive fluorescence assay for microsomal triglyceride transfer protein. J. Lipid Res. 45, 764–772 (2004).
Anaganti, N., Rajan, S. & Hussain, M. M. An improved assay to measure the phospholipid transfer activity of microsomal triglyceride transport protein. J. Lipid Res. 62, (2021).
Zhang, Y. et al. L-[5-11 C]Glutamine PET imaging noninvasively tracks dynamic responses of glutaminolysis in non-alcoholic steatohepatitis. Acta Pharm. Sin. B. https://doi.org/10.1016/j.apsb.2024.07.023 (2024).
Ding, Y. S., Antoni, G., Fowler, J. S., Wolf, A. P. & Langstrom, B. Synthesis of L‐[5‐11 C]ornithine. J. Label. Comp. Radiopharm. 27, 1079–1090 (1989).
Jarry, A. C. et al. Neuromedin U is a gut peptide that alters oral glucose tolerance by delaying gastric emptying via direct contraction of the pylorus and vagal-dependent mechanisms. FASEB J. 33, 5377–5388 (2019).
Fagarasan, S. et al. Critical roles of activation-induced cytidine deaminase in the homeostasis of gut flora. Sci. (1979). 298, 1421–1424 (2002).
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004).
Jimba, S. et al. Prevalence of non-alcoholic fatty liver disease and its association with impaired glucose metabolism in Japanese adults. Diabet. Med. 22, 1141–1145 (2005).
Zhuang, X., Yang, Z. & Cordes, D. A technical review of canonical correlation analysis for neuroscience applications. Hum. Brain Mapp. 41, 3807–3833. https://doi.org/10.1002/hbm.25090 (2020).
Acknowledgements
We thank Dr. Minoru Yoshida and Dr. Yasuhiro Ogata (Japanese Red Cross Kumamoto Health Care Center) for the human data collection. We also thank Ms. Tomomi Ueda and Ms. Noyumi Nakamura (Department of Veterinary Medical Sciences, the University of Tokyo) for their technical support. Further, we thank Dr. Shin-Ichiro Takahashi (The University of Tokyo) for his contribution in supervising our experiments. We would like to thank Editage (www.editage.com) for the English language editing.
Funding
This work was supported in part by the Japan Society for the Promotion of Science (JSPS) KAKENHI, #23K13923 granted to H.N., 24K21295 and 23K27558 granted to M.R.Z.; 15H04583, 23H00358 and 23K23793 granted to F.H.; and the Moonshot Research and Development Program (##21zf0127003h001) granted to M.R.Z.; and the Cross-ministerial Moonshot Agriculture, Forestry, and Fishers Research, from the Bio-oriented Technology Research Advancement Institution, BRAIN (#20350956) granted to N.H. and F.H.
Author information
Authors and Affiliations
Contributions
H.N. designed the study; H.N. and S.N. conducted most experiments; L.X., M.F., Y.Z., and M.R.Z. contributed to 11 C-Orn synthesis and related experiments; D.Y. contributed to LC-MS-based metabolite analyses; J.S. and K.O. collected human blood samples, performed human data curation, examined patients, and collected clinical samples; S.F. contributed to the clinical data processing and informatics analyses; F.H. supervised the study; H.N., S.F., and F.H. wrote the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nishi, H., Nakanishi, S., Xie, L. et al. Remote regulation of hepatic lipid secretion in the intestine by metabolic interaction of dietary arginine with ornithine. Sci Rep (2026). https://doi.org/10.1038/s41598-026-47841-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-47841-8