Abstract
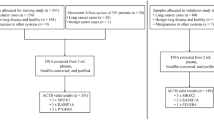
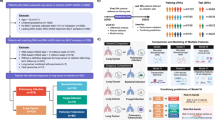
Lung cancer diagnosis poses a significant clinical challenge, with emphasis on enhancing the positivity rate and accuracy of early detection. The use of bronchoalveolar lavage fluid (BALF) for detecting the methylation of ras-association domain family member 1 A (RASSF1A) and short stature homeobox 2 (SHOX2) genes has emerged as a novel molecular diagnostic technique for lung cancer. Nonetheless, this method’s positivity rate can vary due to factors such as BALF quality, and its diagnostic consistency is uncertain. It was a prospective diagnostic study with randomized sampling. In this study, 310 patients with lung lesions detected by computed tomography (CT) imaging were enrolled, and they were randomized 1:1 into pre-biopsy BALF group and post-biopsy BALF group. RASSF1A and SHOX2 methylation in BALF were detected, and CT images and tumor markers of patients were collected to develop a multimodal model based on BALF methylation for predicting malignant lung lesions. An internal validation set was employed to gauge the model’s effectiveness. The findings revealed a statistically significant increase in gene methylation positivity rate and pathological cytology rates in the post-biopsy BALF group compared to the pre-biopsy BALF group (P < 0.05). The model demonstrated an area under the curve (AUC) of 0.985 for predicting malignant lung masses and 0.903 for lung nodules in the training set. When tested on the validation set, the AUC for predicting malignant lung masses and lung nodules was 0.930 and 0.811, respectively. The multimodal prediction model constructed based on RASSF1A and SHOX2 methylation of post-biopsy BALF demonstrates a high predictive value for identifying malignant lung lesions.
Similar content being viewed by others
Acknowledgements
This work was supported by the Technology Innovation and Application Development Special Key Project of Chongqing, China (No: CSTC2021jscx-gksb-N0029) and the Chongqing Talent Project (Innovation Leader) in China (No: CQYC20220303525).
Funding
This work was funded by the Technology Innovation and Application Development Special Key Project of Chongqing, China (No: CSTC2021jscx-gksb-N0029) and the Chongqing Talent Project (Innovation Leader) in China (No: CQYC20220303525).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Institutional review board statement
Approval of the research protocol by an Institutional Review Board: The study received approval from the Ethics Committee of the Second Affiliated Hospital of Army Medical University.Registry and the Registration No. of the study/trial: The study is registered with the Ethics Committee of the Second Affiliated Hospital of Army Medical University (No: 2022-Research 415-01). The clinical trial is registered with the Chinese Clinical Trial Registry (No: ChiCTR2300077871) on 22th November 2023.
Informed consent
Informed consent was obtained from all patients or their guardians.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, J., You, C., Bai, L. et al. Multimodal modeling based on DNA methylation analysis in bronchoalveolar lavage fluid for early lung cancer detection. Sci Rep (2026). https://doi.org/10.1038/s41598-026-50119-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-50119-8