Abstract
Imatinib is the first-line treatment for advanced gastrointestinal stromal tumors (GISTs) harboring KIT or PDGFRA mutations. Unfortunately, resistance invariably develops, typically through secondary KIT/PDGFRA mutations. Here, we describe an unprecedented case of acquired imatinib resistance associated with an oncogenic driver switch, from a KIT mutation to an NTRK3 fusion. The index case was a KIT exon 11-mutated gastric GIST that progressed on imatinib. Despite retaining the original KIT mutation and DOG1 expression, the relapsed tumor lost KIT expression and exhibited a dedifferentiated phenotype. Transcriptomic profiling revealed a de novo EML4::NTRK3 gene fusion. In vitro modeling demonstrated that EML4::NTRK3 confers imatinib resistance, while sensitizing GIST cells to NTRK inhibitors. This first reported instance of an NTRK fusion as a secondary event in GIST progression underscores the importance of testing for NTRK alterations in tumors that have developed resistance to tyrosine kinase inhibitors to ensure patients are offered all available therapeutic options.
Similar content being viewed by others
Introduction
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal neoplasms of the gastrointestinal tract. The majority of GISTs are driven by activating mutations in KIT or PDGFRA, while less common drivers include mutations in BRAF, NF1, or alterations in the SDH complex. More recently, a small subset of GISTs has been found to harbour oncogenic NTRK fusions1. Like KIT and PDGFRA, NTRK is a tyrosine kinase receptor that promotes cell proliferation and survival through MAPK/ERK, PI3K/AKT, and JAK/STAT signaling pathways. Therefore, both oncogenic KIT mutations and NTRK fusions lead to ligand-independent constitutive activation of convergent downstream signaling cascades2,3,4.
Tyrosine kinase inhibitors (TKIs) are the standard of care for advanced KIT/PDGFRA mutant GISTs, with imatinib as the first-line agent1,5,6,7. However, secondary resistance to these treatments eventually develops, often due to additional on-target gene mutations that impair drug binding8,9,10,11,12,13,14,15,16,17 or, less frequently, off-target gene alterations18,19,20 (Table 1).
Here, we report a case of acquired imatinib resistance associated with an oncogenic driver switch, from KIT exon 11 mutation to EML4::NTRK3 fusion. To our knowledge, this is the first report of an NTRK3 fusion being involved as a mechanism of oncogenic driver switch and imatinib resistance in GISTs. Since NTRK fusion-positive tumors have been shown to be sensitive to NTRK inhibitors21,22, the case presented here discloses new therapeutic opportunities for the treatment of GISTs that progress under TKIs therapy.
Results
Clinical case
The index case was a 35-years old man who was admitted to the Arezzo General Hospital in November 2014 for abdominal pain. Computed tomography (CT) showed a large gastric mass and several diaphragm nodules. The patient underwent partial gastrectomy and splenectomy.
Histological analysis revealed that the tumor was composed of monomorphic spindle cells with scant eosinophilic cytoplasm and paranuclear vacuolation arranged in a fascicular pattern of growth (Fig. 1a, b). Numerous non-atypical mitotic figures were detected (approximately 50 mitoses/5 mm2) (Fig. 1c). Tumor cells showed strong and diffuse expression of CD117 and DOG1 (Fig. 1d, e). Molecular analysis identified an imatinib-sensitizing KIT exon 11 mutation (c.1670_1675del, corresponding to pW557_V559delinsF) (Fig. 1f). The final diagnosis was of metastatic GIST with conventional morphology.
a, b Primary resection specimen showing spindle cell morphology with paranuclear vacuolation admixed with areas of increased cellularity and nuclear atypia. c Numerous mitotic figures were detected. d, e Tumor cells display strong, diffuse expression of CD117 and DOG1. f DNA mutation analysis of the primary GIST (tumor cell content 90%) revealed the presence of an imatinib-sensitizing KIT exon 11 mutation consisting of a 6-nucleotide deletion (c.1670_1675del, corresponding to pW557_V559delinsF), underlined in the figure. g–i Tumor relapse exhibits atypical undifferentiated spindle and ovoid cells within a myxoid stroma, with areas of necrosis and a high mitotic index, including atypical mitoses. j, k The neoplastic cells are negative for CD117, while strong multifocal expression of DOG1 was maintained. l The same pW557_V559delinsF KIT mutation detected in the primary tumor was present in the DNA of the recurrence (tumor cell content 60%). Images were taken at 10X magnification.
Post-surgical imaging confirmed disease-free status. Considering the advanced status of the disease, adjuvant imatinib (400 mg/day) was initiated (February 2015) but discontinued after 18 months at the patient’s request. Clinical follow-up was maintained thereafter. In June 2017 (31 months post-diagnosis), the patient presented with epigastric pain and a palpable mass; imaging revealed a 26 mm hepatic lesion and multiple epigastric nodules consistent with neoplastic recurrence. Imatinib (400 mg/day) was restarted, leading to marked regression of lesions (Fig. 2a, b). In February 2018 (39 months post-diagnosis), PET/CT (Positron Emission Tomography/Computed Tomography) showed diffuse peritoneal “sarcomatosis”, including a gastric mass, and peritoneal and periesophageal nodules (Fig. 2c). Imatinib was escalated (800 mg/day) but disease progressed (Fig. 2d). Debulking surgery was performed (April 2018).
a, b Follow-up CT scan from September 2017 shows a partial response to imatinib, with reduced size of periesophageal and hepatic lesions (red arrows). c By February 2018, CT imaging reveals disease progression, with new intra-abdominal lesions, including an 8 cm perigastric mass with central necrosis (red arrow). d Following imatinib dose escalation, further progression is observed, including enlargement of the perigastric mass. e Imaging obtained at the late-stage of disease showing a large tumor mass occupying most of the abdominal cavity.
Pathological examination revealed an atypical undifferentiated spindle and ovoid cell morphology set in a myxoid stroma. Diffuse areas of necrosis were present and mitotic index was elevated, including atypical forms (Fig. 1g–i). The tumor was CD117 negative but showed strong, albeit multifocal, DOG1 positivity (Fig. 1j, k). Molecular analysis of tumor DNA revealed the KIT exon 11 mutation (pW557_V559delinsF) originally detected in the primary lesion (Fig. 1l). No canonical KIT resistance mutation was identified. The combination of clinical history, immunophenotype, and molecular data, despite the absence of classic GIST morphology, was consistent with the diagnosis of relapse of GIST that had undergone dedifferentiation. Sunitinib (37.5 mg/day) was initiated but the patient experienced rapid disease progression and died shortly after (June 2018) (Fig. 2e).
RNA-sequencing identifies a novel EML4::NTRK3 fusion transcript in the relapse
To gain insights into the mechanisms of imatinib resistance and GIST dedifferentiation, both primary tumor and relapse were subsequently transcriptionally profiled by RNA-sequencing. In line with immunohistochemistry, the relapse showed a dramatic decrease in KIT mRNA levels. Interestingly, RNA-sequencing revealed an in-frame EML4::NTRK3 fusion, which involves exons 1–2 of EML4 (chr 2p21, NM_019063) and exons 14–20 of NTRK3 (chr 15q25.3, NM_001012338). EML4 (Echinoderm Microtubule Associated Protein Like 4) is a microtubule-associated protein that has been reported to participate in oncogenic gene fusion events in various cancers23,24. NTRK3 (Neurotrophic Receptor Tyrosine Kinase 3) is a member of the tropomyosin receptor kinase family that, upon binding its ligand NTF3 (neurotrophin-3), undergoes autophosphorylation and activates pro-proliferative signaling cascades, including MAPK/ERK, PI3K/AKT, and JAK/STAT4. In the identified EML4::NTRK3 fusion, the 429-amino acid chimeric protein encompasses the EML4 coiled-coil domain (aa 1–69) and the NTRK3 kinase domain (aa 70–429) (Fig. 3a; Supplementary Table 1). No in-frame fusion was detected in the primary tumor. RT-PCR confirmed the expression of the EML4::NTRK3 chimera solely in the relapse, and Sanger sequencing validated the breakpoint identified by RNA-sequencing (Fig. 3b, c; Supplementary Fig. S1). The EML4 gene is well expressed in GISTs (data not shown), indicating that the EML4 promoter likely supports significant expression of the fusion. Accordingly, immunohistochemistry for pan-TRK showed strong and diffused staining in the relapse whilst the primary tumor was negative (Fig. 3d, e).
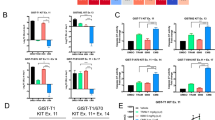
a Schematic representation of the EML4::NTRK3 fusion identified by RNA-sequencing in the relapse. b The selective expression of the EML4::NTRK3 fusion transcript in the relapse but not in the primary tumor was confirmed by RT-PCR. c Sequencing of the RT-PCR product of the relapse displaying the junction between EML4 and NTRK3. d Immunohistochemistry for pan-TRK shows strong and diffuse staining in the recurrence. e Negative pan-TRK immunostaining in the primary tumor (10X magnification). f Western blot analysis for KIT and the NTRK-fused protein product in the GIST-E4N3 cell model engineered to express the EML4::NTRK3 chimera. GIST-CTR cells served as a negative control. GAPDH was used as a loading control. A representative image of three biologically independent replicates is shown. g GIST-E4N3 cells show reduced sensitivity to imatinib relative to GIST-CTR. Relative cell count (RCC) was assessed by the SRB assay at 48 h post-treatment. *p-value ≤ 0.05, **p-value ≤ 0.01, ****p-value ≤ 0.0001. Data are presented as mean ± SEM (n = 6). h Colony growth assay demonstrating the capacity of GIST-E4N3 cells to survive a chronic, 3-week imatinib treatment (50 nM). For better visualization of individual colonies of vehicle-treated cells, images taken at 2-week DMSO treatment are shown. i Compared to GIST-CTR, imatinib-resistant derivatives of GIST-E4N3 cells (GIST-E4N3R) exhibit increased sensitivity to NTRK inhibitors (200 nM larotrectinib, dark green; 200 nM entrectinib, light green; 48 h-treatment). ****p-value ≤ 0.0001. Data are presented as mean ± SEM (n = 6).
Overall, these data indicate that after imatinib treatment the tumor acquired an EML4::NTRK3 gene fusion while losing the expression of the activated KIT allele. This suggests that an oncogenic driver switch occurred during treatment, and that the expression of the EML4::NTRK3 fusion product supports imatinib resistance.
EML4::NTRK3-expressing cells are resistant to imatinib and sensitive to NTRK inhibitors
To investigate the hypothesis that the EML4::NTRK3 fusion product supports the proliferation of GIST cells under imatinib pressure, we utilized the GIST-T1 cell line25. This cell line carries an imatinib-sensitizing, heterozygous KIT exon 11 mutation (p.Val560_Tyr578del), thereby mimicking the genetic context of the primary tumor in the index case. GIST-T1 cells were engineered to ectopically express the EML4::NTRK3 chimera (GIST-E4N3), while cells transduced with the empty vector served as a control (GIST-CTR) (Fig. 3f; Supplementary Fig. S2).
Compared to GIST-CTR cells, GIST-E4N3 cells exhibited reduced sensitivity to imatinib under both acute (Fig. 3g) and chronic (Fig. 3h) treatment conditions. Specifically, in both the SRB cell viability assay performed after a 48-hour treatment with imatinib at different dosages, and the colony growth assay evaluated after 3 weeks of treatment, GIST-E4N3 cells showed increased survival. These results suggest that the EML4::NTRK3 fusion transcript confers a growth advantage to GIST-T1 cells under imatinib pressure.
Imatinib-resistant GIST-E4N3 cells (GIST-E4N3R), obtained following prolonged imatinib challenge, were evaluated for their response to NTRK inhibitors. Compared to GIST-CTR cells, GIST-E4N3R cells exhibited increased sensitivity to both larotrectinib and entrectinib, supporting the relevance of the EML4::NTRK3 fusion in their viability (Fig. 3i).
Discussion
Although systemic treatment with imatinib or other TKIs can achieve long-term disease control in patients with KIT or PDGFRA mutations, most GISTs eventually progress due to the development of acquired resistance. Imatinib resistance is commonly associated with the acquisition of secondary mutations in the ATP-binding pocket or the activation loop of the primary driver (KIT or PDGFRA)1,8,9,10,11,12,13,14,15,16,17, although other mechanisms (e.g., KIT or PDGFRA amplification)9,10,15 or off-target gene alterations (e.g., mutations in downstream effectors or activation of alternative receptor tyrosine kinases)18,19,20 may also be involved. Recently, a FGFR2::TACC2 gene fusion was detected in a multi TKI-resistant KIT exon 11-driven GIST, prompting the authors to speculate a role for the fusion in drug resistance20.
We and others have reported the detection of NTRK activating gene fusions in GISTs devoid of canonical oncogenic drivers21,22,26. Although the existence of this group of GISTs has been long questioned27, a recent survey has provided evidence that GISTs carrying NTRK fusions do exist and may be sensitive to NTRK inhibitors22. Importantly, in these tumors the fusion represents the primary driver event.
The case reported here highlights an unprecedented scenario: NTRK fusions can represent a mechanism of oncogenic driver switch and acquired resistance to imatinib. The oncogenic driver switch, from a KIT mutation to an NTRK3 fusion, occurred after imatinib treatment and was associated with concomitant transcriptional silencing of the original KIT driver mutation. Notably, this molecular switch was accompanied by a morphological transition toward a dedifferentiated GIST phenotype. GIST dedifferentiation is characterized by atypical morphology, loss of canonical markers such as CD117, DOG1, and CD34, and it is often associated with long-term imatinib therapy and disease progression28,29,30,31. Dedifferentiation poses significant diagnostic challenges, as the dedifferentiated component may histologically mimic high-grade sarcomas or other high-grade malignancies, potentially resulting in misclassification. In our case, although the post-imatinib tumor exhibited atypical morphology, the maintenance of the original KIT exon 11 mutation at the genomic level and the expression of DOG1 supported the diagnosis of dedifferentiated GIST. This highlights the importance of integrating immunohistochemistry, molecular, and clinical data to ensure an accurate diagnosis and avoid diagnostic pitfalls.
To date, approximately 20 cases of GISTs harboring NTRK fusions have been reported in the literature, with CD117 and/or DOG1 expression documented in about half of them22. Our study adds further support to the notion that NTRK fusion-driven GISTs do exist and represent a distinct molecular entity. Importantly, to the best of our knowledge, this is the first report demonstrating an NTRK fusion as a mechanism of oncogenic driver switch and acquired resistance to imatinib.
Through in vitro modeling, we demonstrated that the EML4::NTRK3 fusion confers a growth advantage under imatinib treatment to KIT-driven GIST cells, supporting its functional role in resistance. Moreover, the expression of the fusion conveys sensitivity to NTRK inhibitors to imatinib-resistant cells. Unfortunately, the patient did not benefit from our findings since the EML4::NTRK3 fusion was identified through RNA sequencing after the patient had already passed away. Consequently, NTRK inhibitor therapy was not administered, and its therapeutic efficacy in our patient remains unassessed, representing a limitation of our study. It is also important to note that transcriptional profiling is not part of the standard diagnostic workflow for GIST. Therefore, our finding that NTRK fusions can emerge as secondary events in GIST progression underscores the clinical relevance of including NTRK fusion analysis into the molecular workup of GISTs with acquired TKI resistance to inform personalized treatment approaches.
Methods
Histopathological diagnosis
The case was sent for consultation to APDT and the revised diagnosis was made by APDT and MS based on morphology, immunophenotype, and molecular data.
Immunohistochemistry was performed on 3 μm formalin-fixed, paraffin-embedded tissue sections using a panel of antibodies to evaluate tumor phenotype. The antibodies included CD117 (polyclonal serum, dilution 1:400; Dako; Carpentaria, CA), DOG1 (clone SP31, prediluted; Cell Marque; Rocklin, CA), S100 (clone EP32, prediluted; Leica Biosystem; Nussloch, DE), SMA (clone 1A4, dilution 1:100; Cell Marque; Rocklin, CA), Desmin (clone D33, dilution 1:400; Dako; Carpentaria, CA), and pan-TRK (clone EPR17341, prediluted; Ventana; Cupertino, CA). Detection was carried out using a standard immunoperoxidase method, diaminobenzidine chromogen and hematoxylin counterstaining. The study was approved by the local ethics committee (Marca 456/CE). Informed consent was provided by the study participants. All procedures were carried out in accordance with the principles of the Declaration of Helsinki.
DNA mutation analysis and RNA-sequencing
DNA and RNA extraction were essentially as described in ref. 32. Briefly, DNA and RNA were extracted from FFPE tissue sections mounted on glass slides. Areas with high tumor cell content (90% in the primary tumor and 60% in the relapse) were marked by the pathologists on a reference hematoxylin-eosin-stained slide. Corresponding regions were scraped from matched unstained slides and dewaxed with the Qiagen deparaffinization solution (Qiagen, Germantown, MD, USA). The Ambion RecoverAll™ Total Nucleic Acid Isolation Kit for FFPE (Thermo Fisher Scientific, Carlsbad, CA, USA) was used for DNA and RNA purification. KIT, PDGFRA, SDHA-D, BRAF, H/K/NRAS and NF1 gene mutation analysis was performed using a targeted NGS sequencing panel32. For whole RNA-sequencing analysis, libraries were prepared using the Illumina TruSeq Stranded Total RNA Kit with Ribo-Zero (Illumina, San Diego, CA, USA) and processed as described33. Libraries were sequenced on an Illumina NextSeq550 apparatus to an average of 60 million paired-end reads per sample. The quality of the row sequencing data was evaluated with the FastQC software. Reads were aligned to the hg38 reference genome (GRCh38.p13) with STAR v2.7.3.a, and RSEM v1.3.1 was utilized for transcript quantification34,35. Arriba and Dragen tools were used for fusion transcript identification36,37.
PCR/Sanger sequencing validation of KIT exon 11 mutation and EML4::NTRK3 fusion
KIT exon 11 was PCR-amplified from genomic DNA of the primary tumor and relapse samples using the GoTaq® Master Mix (Promega, Madison, WI, USA) and the following primers: forward 5’-TCCAGAGTGCTCTAATGACTGAG-3’ and reverse 5’-GCCTGTTTCTGGGAAACTCC-3’. PCR-Sanger sequencing was then performed using the same primers to confirm the presence of the c.1670_1675del mutation.
Total RNA (500 ng), isolated from the primary tumor and relapse samples, was reverse-transcribed using SuperScript III (Thermo Fisher Scientific, Carlsbad, CA, USA). To confirm the expression of the EML4::NTRK3 fusion transcript, RT-PCR was performed using the following primers: EML4 forward 5’-TTGAGGCGTCTTGCAATCT-3’ and NTRK3 reverse 5’-GCTGAGTCCTCCTCACCACTGATG-3’. The resulting PCR product was subsequently sequenced using the same primers.
Cell model generation
LinxA were obtained from Greg Hannon (Cancer Research UK Cambridge Institute, Cambridge, UK). GIST-T1 were obtained from Cosmo Bio (Carlsbad, CA, USA). Cells were regularly authenticated by STR profiling (Supplementary Table 2). Both cell lines were maintained in DMEM (Thermo Fisher Scientific, Carlsbad, CA, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS, Thermo Fisher Scientific) and gentamicin (8 μg/mL), at 37 °C and 5% CO2.
To generate the GIST-E4N3 cell model, the EML4::NTRK3 cDNA was PCR-amplified and cloned into the pLPC-puro retroviral vector as previously described26. LinxA cells were transfected with either the pLPC-E4N3 or the empty pLPC vector. Forty-eight hours post-transfection, the supernatants were collected, filtered through a 0.22 µm filter, and used to infect GIST-T1 cells. Infected cells were enriched by puromycin selection.
Imatinib-resistant GIST-E4N3R cells were derived by chronic imatinib treatment (20 nM for >30 days) of GIST-E4N3 cells.
Cell viability and colony growth assays
For drug sensitivity assays, cells were seeded in sextuplicate in 96-well plates and treated the following day with imatinib, larotrectinib, or entrectinib, as indicated (Selleckchem, Munich, DE). Stock solutions of each drug were prepared at a 1000× concentration in DMSO (vehicle).
The sulforhodamine B (SRB) cell viability assay was used to evaluate cell viability after a short-term drug treatment (48 h). The relative cell count (RCC) was calculated as the percentage of cells surviving the 48h-treatment, normalized to DMSO-treated cells.
To evaluate cell survival under chronic treatment conditions, a colony growth assay was performed. GIST-E4N3 and GIST-CTR cells were seeded onto 100 mm dishes at a density of 1 × 104 cells per dish. Cells were cultured for 10 days before treatment with 50 nM imatinib (Selleckchem) or vehicle (DMSO) for three weeks. Media supplemented with imatinib or DMSO was refreshed every 3–4 days. Colony growth was assessed by crystal violet staining according to a standard protocol38. Experiments were conducted at least in triplicate and results were confirmed on two independent cell infections. p-values were calculated with 2-tailed unpaired t test. Data were analyzed using GraphPad Prism Software.
Western blotting
Protein expression was determined by Western blot. Briefly, cells were lysed in RIPA buffer (Santa Cruz Biotechnology, Dallas, TX, USA). Whole cell lysates were quantified using the Bradford protein assay (Bio-Rad Laboratories, Hercules, CA, USA). Proteins were separated by SDS-PAGE on 4–15% Criterion TGX precast gels (Bio-Rad Laboratories) and transferred onto a nitrocellulose membrane (Protran Whatman, Merck, Darmstadt, DE). Proteins were detected by chemiluminescence (LiteUp kit, Euroclone, Pero, Milan, Italy). Immunoblots were scanned using a ChemiDoc MP apparatus and analyzed with the ImageLab software (Bio-Rad Laboratories). The following antibodies were used: anti-KIT (clone E-1; Santa Cruz Biotechnology) and anti-TRK (polyclonal serum; Santa Cruz Biotechnology). Anti-GAPDH (clone 6C5; Santa Cruz Biotechnology) was used for loading control.
Data availability
Data is provided within the manuscript or in the supplementary information file. Raw data (FASTQ files) are available on NCBI's SRA database (PRJNA1373022).
References
Cicala, C. M., Bauer, S., Heinrich, M. C. & Serrano, C. Gastrointestinal Stromal Tumor: Current Approaches and Future Directions in the Treatment of Advanced Disease. Hematol. Oncol. Clin. North Am. 39, 773–784 (2025).
Szucs, Z. et al. Molecular subtypes of gastrointestinal stromal tumors and their prognostic and therapeutic implications. Future Oncol. 13, 93–107 (2017).
Bauer, S., George, S., von Mehren, M. & Heinrich, M. C. Early and Next-Generation KIT/PDGFRA Kinase Inhibitors and the Future of Treatment for Advanced Gastrointestinal Stromal Tumor. Front. Oncol. 11, 672500 (2021).
Cocco, E., Scaltriti, M. & Drilon, A. NTRK fusion-positive cancers and TRK inhibitor therapy. Nat. Rev. Clin. Oncol. 15, 731–747 (2018).
Joensuu, H. et al. Effect of the Tyrosine Kinase Inhibitor STI571 in a Patient with a Metastatic Gastrointestinal Stromal Tumor. N. Engl. J. Med. 344, 1052–1056 (2001).
Demetri, G. D. et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet Lond. Engl. 381, 295–302 (2013).
Blay, J.-Y. et al. Ripretinib in patients with advanced gastrointestinal stromal tumours (INVICTUS): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 21, 923–934 (2020).
Antonescu, C. R. et al. Acquired Resistance to Imatinib in Gastrointestinal Stromal Tumor Occurs Through Secondary Gene Mutation. Clin. Cancer Res. 11, 4182–4190 (2005).
Debiec-Rychter, M. et al. Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the PKC412 inhibitor against imatinib-resistant mutants. Gastroenterology 128, 270–279 (2005).
Heinrich, M. C. et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J. Clin. Oncol. J. Am. Soc. Clin. Oncol. 24, 4764–4774 (2006).
Wardelmann, E. et al. Polyclonal Evolution of Multiple Secondary KIT Mutations in Gastrointestinal Stromal Tumors under Treatment with Imatinib Mesylate. Clin. Cancer Res. 12, 1743–1749 (2006).
Desai, J. et al. Clonal Evolution of Resistance to Imatinib in Patients with Metastatic Gastrointestinal Stromal Tumors. Clin. Cancer Res. 13, 5398–5405 (2007).
Miselli, F. C. et al. c-Kit/PDGFRA gene status alterations possibly related to primary imatinib resistance in gastrointestinal stromal tumors. Clin. Cancer Res. J. Am. Assoc. Cancer Res. 13, 2369–2377 (2007).
Heinrich, M. C. et al. Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumor. J. Clin. Oncol. J. Am. Soc. Clin. Oncol. 26, 5352–5359 (2008).
Liegl, B. et al. Heterogeneity of kinase inhibitor resistance mechanisms in GIST. J. Pathol. 216, 64–74 (2008).
Du, J. et al. Identifying Secondary Mutations in Chinese Patients with Imatinib-Resistant Gastrointestinal Stromal Tumors (GISTs) by Next Generation Sequencing (NGS). Pathol. Oncol. Res. POR 26, 91–100 (2020).
Li, J. et al. Clinicopathological characteristics of progressive gastrointestinal stromal tumors and heterogeneity analyses of secondary mutations. Oncologist 30, oyaf110 (2025).
Mahadevan, D. et al. A novel tyrosine kinase switch is a mechanism of imatinib resistance in gastrointestinal stromal tumors. Oncogene 26, 3909–3919 (2007).
Agaram, N. P. et al. Novel V600E BRAF mutations in imatinib-naive and imatinib-resistant gastrointestinal stromal tumors. Genes. Chromosomes Cancer 47, 853–859 (2008).
Dermawan, J. K. et al. FGFR2::TACC2 fusion as a novel KIT-independent mechanism of targeted therapy failure in a multidrug-resistant gastrointestinal stromal tumor. Genes. Chromosomes Cancer 61, 412–419 (2022).
Machado, I. et al. ETV6::NTRK3 Fusion-Positive Wild-Type Gastrointestinal Stromal Tumor (GIST) with Abundant Lymphoid Infiltration (TILs and Tertiary Lymphoid Structures): A Report on a New Case with Therapeutic Implications and a Literature Review. Int. J. Mol. Sci. 25, 3707 (2024).
Ranjbarian, T. et al. A Systematic Review with a Demonstrative Case of KIT and DOG-1 Expressing Gastrointestinal Stromal Tumors Harboring ETV6–NTRK3 Fusions. Clin. Cancer Res. 31, 2056–2061 (2025).
Soda, M. et al. Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007).
Tang, H. et al. Fusion genes in cancers: Biogenesis, functions, and therapeutic implications. Genes Dis. 12, 101536 (2025).
Taguchi, T. et al. Conventional and molecular cytogenetic characterization of a new human cell line, GIST-T1, established from gastrointestinal stromal tumor. Lab. Investig. J. Tech. Methods Pathol. 82, 663–665 (2002).
Brenca, M. et al. Transcriptome sequencing identifies ETV6–NTRK3 as a gene fusion involved in GIST. J. Pathol. 238, 543–549 (2016).
Atiq, M. A. et al. Mesenchymal tumors of the gastrointestinal tract with NTRK rearrangements: a clinicopathological, immunophenotypic, and molecular study of eight cases, emphasizing their distinction from gastrointestinal stromal tumor (GIST). Mod. Pathol. 34, 95–103 (2021).
Antonescu, C. R. et al. Dedifferentiation in gastrointestinal stromal tumor to an anaplastic KIT-negative phenotype: a diagnostic pitfall: morphologic and molecular characterization of 8 cases occurring either de novo or after imatinib therapy. Am. J. Surg. Pathol. 37, 385–392 (2013).
Choi, J. J., Sinada-Bottros, L., Maker, A. V. & Weisenberg, E. Dedifferentiated gastrointestinal stromal tumor arising de novo from the small intestine. Pathol. Res. Pract. 210, 264–266 (2014).
Karakas, C., Christensen, P., Baek, D., Jung, M. & Ro, J. Y. Dedifferentiated gastrointestinal stromal tumor: Recent advances. Ann. Diagn. Pathol. 39, 118–124 (2019).
Malik, F. et al. Dedifferentiation in SDH-Deficient Gastrointestinal Stromal Tumor: A Report With Histologic, Immunophenotypic, and Molecular Characterization. Pediatr. Dev. Pathol. J. Soc. Pediatr. Pathol. Paediatr. Pathol. Soc. 22, 492–498 (2019).
Gasparotto, D. et al. Quadruple-Negative GIST Is a Sentinel for Unrecognized Neurofibromatosis Type 1 Syndrome. Clin. Cancer Res. J. Am. Assoc. Cancer Res. 23, 273–282 (2017).
Sigalotti, L. et al. Proximal and Classic Epithelioid Sarcomas are Distinct Molecular Entities Defined by MYC/GATA3 and SOX17/Endothelial Markers, Respectively. Mod. Pathol. J. U. S. Can. Acad. Pathol. Inc. 38, 100647 (2025).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinforma. Oxf. Engl. 29, 15–21 (2013).
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinforma. 12, 323 (2011).
Uhrig, S. et al. Accurate and efficient detection of gene fusions from RNA sequencing data. Genome Res 31, 448–460 (2021).
Behera, S. et al. Comprehensive genome analysis and variant detection at scale using DRAGEN. Nat. Biotechnol. 43, 1177–1191 (2025).
Franken, N. A. P., Rodermond, H. M., Stap, J., Haveman, J. & van Bree, C. Clonogenic assay of cells in vitro. Nat. Protoc. 1, 2315–2319 (2006).
Acknowledgements
The authors are grateful to the members of the Laboratory of Oncogenetics and Functional Oncogenomics for their assistance and suggestions, and to AIRC, the Italian Ministries of Health and of University and Research, and CRO for their support. This work was funded by Fondazione AIRC per la ricerca sul cancro ETS (project nr. 19975), Ministero della Salute (Ricerca Corrente), 5x1000 CRO intramural grant, and Ministero dell'Università e della Ricerca (PRIN 2022ZY8XVR).
Author information
Authors and Affiliations
Contributions
R.M., A.P.D.T.: conceptualization and supervision; S.G., M.S., E.Bn, D.G., E.Bi, S.P.: pathobiological investigations and formal analysis; D.B.: bioinformatics analysis; G.R., E.M., M.P.R., A.C.: clinical investigations and sample collection; S.G., M.S., A.P.D.T., R.M.: manuscript drafting. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gloazzo, S., Sbaraglia, M., Bellan, E. et al. KIT Mutation-NTRK fusion oncogenic driver switch: a novel mechanism of acquired imatinib resistance in GIST. npj Precis. Onc. 10, 75 (2026). https://doi.org/10.1038/s41698-026-01289-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41698-026-01289-1