Abstract
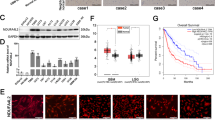
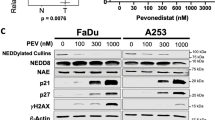
Head and neck squamous cell carcinoma (HNSCC) represents a leading global malignancy among head and neck cancers. While chemotherapy serves as a standard adjuvant treatment, cisplatin resistance frequently compromises therapeutic outcomes. PANoptosis is an integrated inflammatory cell death pathway governed by PANoptosome complexes. It critically influences chemotherapy response, though its regulatory mechanisms remain incompletely characterized. NADH dehydrogenase (ubiquinone) 1 alpha subcomplex subunit 4-like 2 (NDUFA4L2), a subunit of respiratory chain complex I, has been identified as a critical regulator of cell survival. Our multi-platform investigation employed HNSCC cell lines, patient-derived organoids, tongue orthotopic xenograft models in C57BL/6 mice and Tgfbr1/Pten 2cKO mice to elucidate the role of NDUFA4L2 in cisplatin resistance. Bioinformatic analysis and clinical samples indicate that elevated NDUFA4L2 is associated with poor survival rates and low sensitivity to chemotherapy in HNSCC patients. Through in vitro and in vivo studies, we found that NDUFA4L2-KO in combination with cisplatin suppresses glycolysis levels, thereby inhibiting AIM2 inflammasome activation. Consequently, it triggers tumor cell PANoptosis, remodels the immunosuppressive tumor microenvironment, and enhances antitumor efficacy. These findings establish NDUFA4L2 as both a prognostic biomarker and therapeutic target for overcoming cisplatin resistance in HNSCC through PANoptosis modulation.
Similar content being viewed by others
Data availability
All data relevant to the study are included in the article or uploaded as supplementary information. Additional supporting data are available from the corresponding authors upon reasonable request, and the raw sequence data reported in this paper have been deposited in the Genome Sequence Archive (Genomics, Proteomics & Bioinformatics 2025) in National Genomics Data Center (Nucleic Acids Res 2025), China National Center for Bioinformation/Beijing Institute of Genomics, Chinese Academy of Sciences53,54 (GSA-Human: HRA013828) that are publicly accessible at https://ngdc.cncb.ac.cn/gsa-human.
Code availability
All analyses were performed using conventional analysis code, which is available at https://github.com/carmonalab/GeneNMF55, and no new codes were generated.
References
Johnson, D. E. et al. Head and neck squamous cell carcinoma. Nat. Rev. Dis. Prim. 6, 92 (2020).
Barsouk, A., Aluru, J. S., Rawla, P., Saginala, K. & Barsouk, A. Epidemiology, risk factors, and prevention of head and neck squamous cell carcinoma. Med. Sci. 11, https://doi.org/10.3390/medsci11020042 (2023).
Galluzzi, L. et al. Molecular mechanisms of cisplatin resistance. Oncogene 31, 1869–1883 (2012).
Peng, F. et al. Regulated cell death (RCD) in cancer: key pathways and targeted therapies. Signal Transduct. Target. Ther. 7, 286 (2022).
Wu, D., Wang, S., Yu, G. & Chen, X. Cell death mediated by the pyroptosis pathway with the aid of nanotechnology: prospects for cancer therapy. Angew. Chem. Int Ed. Engl. 60, 8018–8034 (2021).
Bedoui, S., Herold, M. J. & Strasser, A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat. Rev. Mol. Cell Biol. 21, 678–695 (2020).
Malireddi, R. K. S., Kesavardhana, S. & Kanneganti, T. ZBP1 and TAK1: master regulators of NLRP3 inflammasome/pyroptosis, apoptosis, and necroptosis (PAN-optosis). Front. Cell. Infect. Microbiol. 9, 406 (2019).
Lee, S. et al. AIM2 forms a complex with pyrin and ZBP1 to drive PANoptosis and host defence. Nature 597, 415–419 (2021).
Cridland, J. A. et al. The mammalian PYHIN gene family: phylogeny, evolution and expression. BMC Evol. Biol. 12, 140 (2012).
Zhang, M., Zhang, L., Miao, T. & Hu, X. Characterization of PANoptosis-related expression pattern, prognosis and tumor microenvironment in head and neck squamous cell carcinoma. Discov. Oncol. https://doi.org/10.1007/s12672-025-04328-8 (2025).
Yang, P. et al. Identification of PANoptosis-related biomarkers and analysis of prognostic values in head and neck squamous cell carcinoma. Sci. Rep. 14, 9824 (2024).
Tello, D. et al. Induction of the mitochondrial NDUFA4L2 protein by HIF-1alpha decreases oxygen consumption by inhibiting Complex I activity. Cell Metab. 14, 768–779 (2011).
Lai, R. K. et al. NDUFA4L2 fine-tunes oxidative stress in hepatocellular carcinoma. Clin. Cancer Res. 22, 3105–3117 (2016).
Ye, N. et al. Hypoxia-induced the upregulation of NDUFA4L2 promoted colon adenocarcinoma progression through ROS-mediated PI3K/AKT pathway. Cytotechnology 75, 461–472 (2023).
Meng, L., Yang, X., Xie, X. & Wang, M. Mitochondrial NDUFA4L2 protein promotes the vitality of lung cancer cells by repressing oxidative stress. Thorac. Cancer 10, 676–685 (2019).
Yuan, Y. et al. NDUFA4L2 promotes trastuzumab resistance in HER2-positive breast cancer. Ther. Adv. Med. Oncol. 13, 17527428 (2021).
Wang, D., Zhang, P., Liu, Z., Xing’, Y. & Xiao, Y. NXPH4 promotes gemcitabine resistance in bladder cancer by enhancing reactive oxygen species and glycolysis activation through modulating NDUFA4L2. Cancers. 14, https://doi.org/10.3390/cancers14153782 (2022).
Zhou, L. et al. Transcriptional regulation of NDUFA4L2 by NFIB induces sorafenib resistance by decreasing reactive oxygen species in hepatocellular carcinoma. Cancer Sci. 114, 793–805 (2023).
Chamoli, A. et al. Overview of oral cavity squamous cell carcinoma: Risk factors, mechanisms, and diagnostics. Oral. Oncol. 121, 105451 (2021).
Warburg, O., Wind, F. & Negelein, E. The metabolism of tumors in the body. J. Gen. Physiol. 8, 519–530 (1927).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Li, F. et al. Acetylation accumulates PFKFB3 in cytoplasm to promote glycolysis and protects cells from cisplatin-induced apoptosis. Nat. Commun. 9, 508 (2018).
Faes, S. et al. Acidic tumor microenvironment abrogates the efficacy of mTORC1 inhibitors. Mol. Cancer 15, 78 (2016).
Lin, Y. et al. Knockdown of PKM2 enhances radiosensitivity of cervical cancer cells. Cancer Cell Int. 19, 129 (2019).
Zong, S., Dai, W., Fang, W., Guo, X. & Wang, K. SIK2 promotes cisplatin resistance induced by aerobic glycolysis in breast cancer cells through PI3K/AKT/mTOR signaling pathway. Biosci. Rep. https://doi.org/10.1042/BSR20201302 (2020).
Tan, Y. et al. Pyroptosis: a new paradigm of cell death for fighting against cancer. J. Exp. Clin. Cancer Res. 40, 153 (2021).
Slee, E. A. et al. Benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone (Z-VAD.FMK) inhibits apoptosis by blocking the processing of CPP32. Biochem. J. 315, 21–24 (1996).
Xu, W. et al. Gasdermin E-derived caspase-3 inhibitors effectively protect mice from acute hepatic failure. Acta Pharmacol. Sin. 42, 68–76 (2021).
Cao, L. & Mu, W. Necrostatin-1 and necroptosis inhibition: pathophysiology and therapeutic implications. Pharmacol. Res. 163, 105297 (2021).
Wang, Y. et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature 547, 99–103 (2017).
Yu, X. & He, S. GSDME as an executioner of chemotherapy-induced cell death. Sci. China Life Sci. 60, 1291–1294 (2017).
Zong, Z., Ren, J., Yang, B., Zhang, L. & Zhou, F. Emerging roles of lysine lactyltransferases and lactylation. Nat. Cell Biol. 27, 563–574 (2025).
Hu, H. et al. New advances into cisplatin resistance in head and neck squamous carcinoma: mechanisms and therapeutic aspects. Biomed. Pharmacother. 163, 114778 (2023).
Mei, Q. et al. Elevated of NDUFA4L2 expression in colon adenocarcinoma is correlated with an unfavorable prognosis and increased immune cell infiltration. Heliyon 10, e25462 (2024).
Lv, Y. et al. Overexpression of NDUFA4L2 is associated with poor prognosis in patients with colorectal cancer. ANZ J. Surg. 87, E251–E255 (2017).
He, H. et al. Nucleotide metabolism-associated drug resistance gene NDUFA4L2 promotes colon cancer progression and 5-FU resistance. Sci. Rep. 15, 570 (2025).
Li, L. et al. The identification of induction chemo-sensitivity genes of laryngeal squamous cell carcinoma and their clinical utilization. Eur. Arch. Otorhinolaryngol. 275, 2773–2781 (2018).
Mohammad, R. M. et al. Broad targeting of resistance to apoptosis in cancer. Semin. Cancer Biol. 35, S78–S103 (2015).
Zhu, P. et al. Advances in mechanism and regulation of PANoptosis: prospects in disease treatment. Front. Immunol. 14, 1120034 (2023).
Malireddi, R. K. S., Tweedell, R. E. & Kanneganti, T. PANoptosis components, regulation, and implications. Aging 12, 11163–11164 (2020).
Sagulenko, V. et al. AIM2 and NLRP3 inflammasomes activate both apoptotic and pyroptotic death pathways via ASC. Cell Death Differ. 20, 1149–1160 (2013).
Wang, J. et al. DNASE1L3-mediated PANoptosis enhances the efficacy of combination therapy for advanced hepatocellular carcinoma. Theranostics 14, 6798–6817 (2024).
Gao, W. et al. Targeting SKA3 suppresses the proliferation and chemoresistance of laryngeal squamous cell carcinoma via impairing PLK1-AKT axis-mediated glycolysis. Cell Death Dis. 11, 919 (2020).
Wang, L. et al. Wnt1-inducible signaling protein 1 regulates laryngeal squamous cell carcinoma glycolysis and chemoresistance via the YAP1/TEAD1/GLUT1 pathway. J. Cell. Physiol. 234, 15941–15950 (2019).
Rastogi, S., Banerjee, S., Chellappan, S. & Simon, G. R. Glut-1 antibodies induce growth arrest and apoptosis in human cancer cell lines. Cancer Lett. 257, 244–251 (2007).
Munoz-Pinedo, C., Ruiz-Ruiz, C., Ruiz De Almodovar, C., Palacios, C. & Lopez-Rivas, A. Inhibition of glucose metabolism sensitizes tumor cells to death receptor-triggered apoptosis through enhancement of death-inducing signaling complex formation and apical procaspase-8 processing. J. Biol. Chem. 278, 12759–12768 (2003).
Sun, F. et al. Impact of glycolysis enzymes and metabolites in regulating DNA damage repair in tumorigenesis and therapy. Cell Commun. Signal. 23, 44 (2025).
Chen, H. et al. NBS1 lactylation is required for efficient DNA repair and chemotherapy resistance. Nature 631, 663–669 (2024).
Cong, J. et al. Dysfunction of natural killer cells by FBP1-induced inhibition of glycolysis during lung cancer progression. Cell Metab. 28, 243–255 (2018).
Noe, J. T. et al. Lactate supports a metabolic-epigenetic link in macrophage polarization. Sci. Adv. 7, i8602 (2021).
Zhang, D. et al. Metabolic regulation of gene expression by histone lactylation. Nature 574, 575–580 (2019).
Liu, S. et al. Crosstalk between lactylation and RNA modifications in tumorigenesis: mechanisms and therapeutic implications. Biomark. Res. 13, 110 (2025).
Zhang, S. et al. The GSA family in 2025: a broadened sharing platform for multi-omics and multimodal data. Genom. Proteom. Bioinforma. 23, qzaf072 (2025).
Database Resources of the National Genomics Data Center, China National Center for Bioinformation in 2025. Nucleic. Acids. Res. 53, D30-D44 (2025).
Barkley, D. et al. Cancer cell states recur across tumor types and form specific interactions with the tumor microenvironment. Nat. Genet. 54, 1192–1201 (2022).
Acknowledgements
We acknowledge Yongchu Pan, Mifang Yang, Dongling Yan, and Linlin Jiang for the use of experimental instruments (Jiangsu Key Laboratory of Oral Diseases, Nanjing Medical University). The authors thank Prof. Zhi-Jun Sun for the kind gift of the time-inducible tissue-specific Tgfbr1/Pten 2cKO HNSCC mice. This research was funded by the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (Grant No. 23KJA320003).
Author information
Authors and Affiliations
Contributions
J.J.C.: Data curation, conceptualization, methodology, formal analysis, investigation, writing-original draft. Y.Y.: Data curation, methodology, formal analysis, investigation, writing-original draft. J.H.Z.: Data curation, formal analysis, methodology, investigation. Y.J.G.: Data curation, formal analysis, methodology. M.R.Z.: Data curation, formal analysis, investigation. R.Z.: Data curation, methodology. Y.H.L.: Investigation. J.Y.W.: Investigation. X.S.: Conceptualization, supervision, funding acquisition, project administration, resources, writing-review, and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cui, JJ., Yang, Y., Zhao, JH. et al. NDUFA4L2 regulates the progression and chemotherapy sensitivity of HNSCC by inhibiting PANoptosis. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01358-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01358-5