Abstract
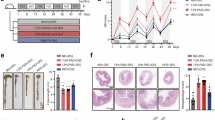
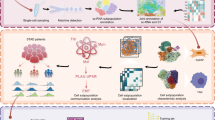
Gastric cancer remains highly lethal, yet how protein S-palmitoylation shapes tumour ecosystems and clinical outcome is unclear. We integrated single-cell RNA sequencing (119,931 cells from 25 gastric tumours) with spatial transcriptomics and bulk cohorts to delineate palmitoylation-linked states across malignant, immune, and stromal compartments. A palmitoylation-high malignant programme partitioned into three metastasis-enriched subclusters with increased fatty-acid metabolism and Ras–MAPK signalling and predicted worse survival. Spatial mapping and ligand–receptor inference revealed co-localised niches where palmitoylation-high tumour cells interacted with immunosuppressive myeloid cells and distinct CAF subsets, with strengthened pro-angiogenic and pro-fibrotic cues. We derived and validated an 87-gene multicellular palmitoylation signature for risk stratification, and higher scores were consistently associated with adverse outcomes in external cohorts. Drug-response modelling highlighted vulnerabilities involving the HSP90–PI3K/MAPK axis. Functional assays and xenografts confirmed SH3BGRL as a key driver within this poor-prognosis programme.
Similar content being viewed by others
Data Availability
The datasets generated and/or analysed during the current study are available in the corresponding repositories. RNA sequencing data and associated clinical data for GC samples were retrieved from The Cancer Genome Atlas (TCGA) and can be accessed through the TCGA portal. Single-cell RNA-seq and spatial transcriptomics data are available from the GEO database (accession numbers: GSE167297, GSE184198, OMIX001073). Further inquiries regarding the datasets should be directed to the corresponding authors.
References
Sundar, R. et al. Gastric cancer. Lancet 405, 2087–2102 (2025).
Duan, Y. et al. Helicobacter pylori and gastric cancer: mechanisms and new perspectives. J. Hematol. Oncol. 18, 10 (2025).
Chen, Y. et al. Identification of core immune-related genes CTSK, C3, and IFITM1 for diagnosing Helicobacter pylori infection-associated gastric cancer through transcriptomic analysis. Int J. Biol. Macromol. 287, 138645 (2025).
Wu, L. W. et al. Diffuse gastric cancer: a comprehensive review of molecular features and emerging therapeutics. Target Oncol. 19, 845–865 (2024).
Chen, Y. et al. The current landscape of gastric cancer and gastroesophageal junction cancer diagnosis and treatment in China: a comprehensive nationwide cohort analysis. J. Hematol. Oncol. 18, 42 (2025).
Ford, A. C. et al. Eradication therapy to prevent gastric cancer in helicobacterpylori-positive individuals: systematic review and meta-analysis of randomized controlled trials and observational studies. Gastroenterology 169, 261–276 (2025).
Liu, Y. et al. Traditional Chinese medicine in the treatment of chronic atrophic gastritis, precancerous lesions and gastric cancer. J. Ethnopharmacol. 337, 118812 (2025).
Pei, L. et al. Electroacupuncture reduces duration of postoperative ileus after laparoscopic gastrectomy for gastric cancer: a multicenter randomized trial. Gastroenterology 169, 73–84 (2025).
Shah, S. C. et al. AGA clinical practice update on screening and surveillance in individuals at increased risk for gastric cancer in the united states: expert review. Gastroenterology 168, 405–416.e1 (2025).
Ren, L. et al. Effectiveness of the CANCER-AIMS intervention on nutritional status and symptom management in patients with gastric cancer following gastrectomy: a randomized controlled trial. Int J. Nurs. Stud. 159, 104873 (2024).
Yu, H. et al. Alkannin triggered apoptosis and ferroptosis in gastric cancer by suppressing lipid metabolism mediated by the c-Fos/SREBF1 axis. Phytomedicine 140, 156604 (2025).
Shoji, H. et al. Phosphoproteomic subtyping of gastric cancer reveals dynamic transformation with chemotherapy and guides targeted cancer treatment. Cell Rep. 43, 114774 (2024).
Hwang, K. et al. Targeted degradation of METTL3 against acute myeloid leukemia and gastric cancer. Eur. J. Med Chem. 279, 116843 (2024).
Jiang, X. et al. Eleutheroside A inhibits PI3K/AKT1/mTOR-mediated glycolysis in MDSCs to alleviate their immunosuppressive function in gastric cancer. Int Immunopharmacol. 159, 114907 (2025).
Zhao, Z. et al. Helicobacter pylori activates SLFN4(+)MDSCs to accelerate gastric intestinal metaplasia. iScience 27, 111255 (2024).
Shen, Y., et al. Rg3-lipo biomimetic delivery of paclitaxel enhances targeting of tumors and myeloid-derived suppressor cells. J. Clin. Invest. 134, e178617 (2024).
Mo, Y. et al. ZDHHC20 mediated S-palmitoylation of fatty acid synthase (FASN) promotes hepatocarcinogenesis. Mol. Cancer 23, 274 (2024).
Chaturvedi, S. & Sonawane, A. Recapitulating the potential contribution of protein S-palmitoylation in cancer. Cancer Metastasis Rev. 44, 20 (2024).
Natale, F. et al. Inhibition of zDHHC7-driven protein S-palmitoylation prevents cognitive deficits in an experimental model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 121, e2402604121 (2024).
Wei, F. et al. ZDHHC7-mediated S-palmitoylation of ATG16L1 facilitates LC3 lipidation and autophagosome formation. Autophagy 20, 2719–2737 (2024).
Huang, B. et al. Palmitoylation-dependent regulation of GPX4 suppresses ferroptosis. Nat. Commun. 16, 867 (2025).
Zheng, W. et al. S-palmitoylation modulates ATG2-dependent non-vesicular lipid transport during starvation-induced autophagy. Embo J. 44, 2596–2619 (2025).
Xin, L. et al. Methionine Restriction Exerts Anti-Tumor Immunity via Joint Intervention of T-Bet palmitoylation in gastric cancer. Biotechnol. J. 20, e202400574 (2025).
Liu, S. et al. Helicobacter pylori CagA promotes gastric cancer immune escape by upregulating SQLE. Cell Death Dis. 16, 17 (2025).
Xu, M. et al. Palmitoyltransferase ZDHHC3 aggravates nonalcoholic steatohepatitis by targeting s-palmitoylated IRHOM2. Adv. Sci. 10, e2302130 (2023).
Tang, F. et al. A pan-cancer single-cell panorama of human natural killer cells. Cell 186, 4235–4251 (2023).
Mahat, D. B. et al. Single-cell nascent RNA sequencing unveils coordinated global transcription. Nature 631, 216–223 (2024).
Ma, L. et al. Single-cell RNA-seq reveals novel interaction between muscle satellite cells and fibro-adipogenic progenitors mediated with FGF7 signalling. J. Cachexia Sarcopenia Muscle 15, 1388–1403 (2024).
Jiang, H. et al. Revealing the transcriptional heterogeneity of organ-specific metastasis in human gastric cancer using single-cell RNA Sequencing. Clin. Transl. Med. 12, e730 (2022).
Zhai, Y. et al. Single-cell RNA sequencing integrated with bulk RNA sequencing analysis reveals diagnostic and prognostic signatures and immunoinfiltration in gastric cancer. Comput. Biol. Med. 163, 107239 (2023).
Wang, Z. et al. Identifying mitophagy-related genes as prognostic biomarkers and therapeutic targets of gastric carcinoma by integrated analysis of single-cell and bulk-RNA sequencing data. Comput. Biol. Med. 163, 107227 (2023).
Kuppe, C. et al. Spatial multi-omic map of human myocardial infarction. Nature 608, 766–777 (2022).
Li, J., Wu, Z., and Lin, R. Impact of Helicobacter pylori on immunotherapy in gastric cancer. J. Immunother. Cancer 12, e010354 (2024).
Kim, H. D. et al. Survival outcomes of patients with gastric cancer treated with first-line nivolumab plus chemotherapy based on claudin 18.2 expression. Gastric Cancer 28, 74–82 (2025).
Kumar, V. et al. Single-cell atlas of lineage states, tumor microenvironment, and subtype-specific expression programs in gastric cancer. Cancer Discov. 12, 670–691 (2022).
Ko, P. J. & Dixon, S. J. Protein palmitoylation and cancer. EMBO Rep. 19, e46666 (2018).
Yao, H. et al. Inhibiting PD-L1 palmitoylation enhances T-cell immune responses against tumours. Nat. Biomed. Eng. 3, 306–317 (2019).
Shen, Z. H. et al. Unveiling the role of protein palmitoylation in gastric cancer diagnosis via machine learning. Discov. Oncol. 16, 1982 (2025).
Zhang, Z. et al. Palmitoylation of TIM-3 promotes immune exhaustion and restrains antitumor immunity. Sci. Immunol. 9, eadp7302 (2024).
He, Y. et al. Palmitic acid accelerates endothelial cell injury and cardiovascular dysfunction via palmitoylation of PKM2. Adv. Sci. 12, e2412895 (2025).
Rocks, O. et al. An acylation cycle regulates localization and activity of palmitoylated Ras isoforms. Science 307, 1746–1752 (2005).
Plowman, S. J. et al. H-ras, K-ras, and inner plasma membrane raft proteins operate in nanoclusters with differential dependence on the actin cytoskeleton. Proc. Natl. Acad. Sci. USA 102, 15500–15505 (2005).
Runkle, K. B. et al. Inhibition of DHHC20-mediated EGFR palmitoylation creates a dependence on EGFR signaling. Mol. Cell 62, 385–396 (2016).
Zhang, Z. et al. Cardiolipin-mimic lipid nanoparticles without antibody modification delivered senolytic in vivo CAR-T therapy for inflamm-aging. Cell Rep. Med. 6, 102209 (2025).
Papp, T. E. et al. CD47 peptide-cloaked lipid nanoparticles promote cell-specific mRNA delivery. Mol. Ther. 33, 3195–3208 (2025).
Song, P. et al. Palmitic acid and palmitoylation in cancer: understanding, insights, and challenges. Innovation 6, 100918 (2025).
Yang, K. et al. The role of lipid metabolic reprogramming in tumor microenvironment. Theranostics 13, 1774–1808 (2023).
Acknowledgements
This study was supported by the General Programme of Health Commission of WuXi (No. M202330) and the collaborative custom development of RNA probes at Soochow University (No. P112213323). The authors would like to acknowledge the contributions of the research participants and the technical support from the institutions involved in this project.
Author information
Authors and Affiliations
Contributions
J.X., Y.H., and Q.Q. contributed equally to this work. J.X. conceived and designed the study. Y.H. and Q.Q. performed data acquisition, preprocessing, and integration of the TCGA and GEO datasets. Y.D.L. and F.C. developed and optimised the machine-learning framework and conducted model training and validation. S.S.H. and H.B.Y. performed the single-cell and spatial transcriptomic analyses. J.L. supervised the computational experiments and provided methodological guidance. Y.Q. and S.H.X. coordinated the project, provided critical revisions, and secured funding. J.X., Y.H., and Q.Q. drafted the manuscript. Y.D.L., F.C., S.S.H., and H.B.Y. prepared figures and contributed to data visualisation. J.L., Y.Q., and S.H.X. reviewed and edited the manuscript. All authors discussed the results, approved the final version, and agree to be accountable for all aspects of the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, J., Hu, Y., Qiao, Q. et al. AI-enabled single-cell dissection of the palmitoylation landscape identifies a multicellular prognostic program in gastric cancer. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01359-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01359-4