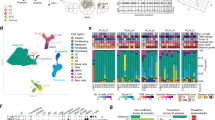
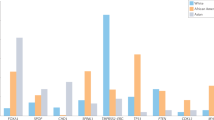
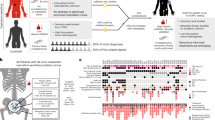
Abstract
Prostate cancer continues to impose a significant health burden worldwide and in the United States, with rising incidence, persistent mortality rates, and disparities in both incidence and patient outcomes. Advances in genomic profiling have highlighted the remarkable heterogeneity of prostate cancer, revealing frequent alterations in key oncogenes, tumor suppressors, and pathways such as androgen receptor signaling and DNA damage repair that drive disease progression and therapy resistance. These molecular discoveries and their mechanistic validation in laboratory and preclinical models are transforming prostate cancer management, enabling more precise, biomarker-driven treatments and clinical trials. However, challenges, including intra- and inter-tumor genomic diversity, remain formidable barriers to progress. This review provides an overview of the current genomic landscape of prostate cancer and discusses recent advances in precision therapy. We also discuss emerging directions in integrating molecular profiling with personalized patient care, while addressing the roadblocks hampering progress. The discussion highlights the critical need for continued innovation in the development of personalized therapies to improve the clinical outcomes for patients with prostate cancer.
Similar content being viewed by others
Data availability
All publicly available information was used and analyzed to prepare this review.
References
Kratzer, T. B. et al. Prostate cancer statistics, 2025. CA Cancer J Clin, 2025.
Siegel, R. L. et al. Cancer statistics, 2025. CA Cancer J. Clin. 75, 10–45 (2025).
Wu, F., Zhang, H. & Hao, M. Interactions between key genes and pathways in prostate cancer progression and therapy resistance. Front. Oncol. 15, 1467540 (2025).
Poon, D. M. C. et al. Treatment of metastatic castration-resistant prostate cancer: review of current evidence and synthesis of expert opinions on radioligand therapy. Front. Oncol. 15, 1530580 (2025).
Kron, K. J. et al. TMPRSS2-ERG fusion co-opts master transcription factors and activates NOTCH signaling in primary prostate cancer. Nat. Genet. 49, 1336–1345 (2017).
Hagglof, C. et al. TMPRSS2-ERG expression predicts prostate cancer survival and associates with stromal biomarkers. PLoS One 9, e86824 (2014).
Xu, Z. et al. Nuclear receptor ERRalpha and transcription factor ERG form a reciprocal loop in the regulation of TMPRSS2:ERG fusion gene in prostate cancer. Oncogene 37, 6259–6274 (2018).
Tian, T. V. et al. Identification of novel TMPRSS2:ERG mechanisms in prostate cancer metastasis: involvement of MMP9 and PLXNA2. Oncogene 33, 2204–2214 (2014).
Shah, N. et al. ERG-mediated coregulator complex formation maintains androgen receptor signaling in prostate cancer. Cancer Res. 80, 4612–4619 (2020).
Mehra, R. et al. Comprehensive assessment of TMPRSS2 and ETS family gene aberrations in clinically localized prostate cancer. Mod. Pathol. 20, 538–544 (2007).
Kumar-Sinha, C., Tomlins, S. A. & Chinnaiyan, A. M. Recurrent gene fusions in prostate cancer. Nat. Rev. Cancer 8, 497–511 (2008).
Tomlins, S. A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005).
Barbieri, C. E. et al. Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat. Genet. 44, 685–689 (2012).
Hernandez-Llodra, S. et al. SPOP and CHD1 alterations in prostate cancer: Relationship with PTEN loss, tumor grade, perineural infiltration, and PSA recurrence. Prostate 81, 1267–1277 (2021).
Zhang, H., Jin, X. & Huang, H. Deregulation of SPOP in Cancer. Cancer Res. 83, 489–499 (2023).
Blattner, M. et al. SPOP Mutation Drives Prostate Tumorigenesis In Vivo through Coordinate Regulation of PI3K/mTOR and AR Signaling. Cancer Cell 31, 436–451 (2017).
Jin, H. J. et al. Cooperativity and equilibrium with FOXA1 define the androgen receptor transcriptional program. Nat. Commun. 5, 3972 (2014).
Tam, K. J. et al. Clinically-observed FOXA1 mutations upregulate SEMA3C through transcriptional derepression in prostate cancer. Sci. Rep. 14, 7082 (2024).
Peacock, J. W. et al. SEMA3C drives cancer growth by transactivating multiple receptor tyrosine kinases via Plexin B1. EMBO Mol. Med. 10, 219–238 (2018).
Hernandez-Llodra, S. et al. SPOP and FOXA1 mutations are associated with PSA recurrence in ERG wt tumors, and SPOP downregulation with ERG-rearranged prostate cancer. Prostate 79, 1156–1165 (2019).
Parolia, A. et al. Distinct structural classes of activating FOXA1 alterations in advanced prostate cancer. Nature 571, 413–418 (2019).
Maddah, M. M. et al. Evaluation of the Prognostic Role of TP53 Gene Mutations in Prostate Cancer Outcome: A Systematic Review and Meta-Analysis. Clin. Genitourin. Cancer 22, 102226 (2024).
Tan, H. L. et al. Rb loss is characteristic of prostatic small cell neuroendocrine carcinoma. Clin. Cancer Res. 20, 890–903 (2014).
Ku, S. Y. et al. Rb1 and Trp53 cooperate to suppress prostate cancer lineage plasticity, metastasis, and antiandrogen resistance. Science 355, 78–83 (2017).
Whang, Y. E. et al. Inactivation of the tumor suppressor PTEN/MMAC1 in advanced human prostate cancer through loss of expression. Proc. Natl. Acad. Sci. USA 95, 5246–5250 (1998).
Pungsrinont, T., Kallenbach, J. & Baniahmad, A. Role of PI3K-AKT-mTOR pathway as a pro-survival signaling and resistance-mediating mechanism to therapy of prostate cancer. Int. J. Mol. Sci. 22 (2021).
Latini, J. M. et al. Loss of heterozygosity and microsatellite instability at chromosomal sites 1Q and 10Q in morphologically distinct regions of late stage prostate lesions. J. Urol. 166, 1931–1936 (2001).
Suzuki, H. et al. Interfocal heterogeneity of PTEN/MMAC1 gene alterations in multiple metastatic prostate cancer tissues. Cancer Res. 58, 204–209 (1998).
Antao, A. M., Ramakrishna, S. & Kim, K. S. The Role of Nkx3.1 in Cancers and Stemness. Int. J. Stem Cells 14, 168–179 (2021).
Pengju, Z. et al. NKX3.1 potentiates TNF-alpha/CHX-induced apoptosis of prostate cancer cells through increasing caspase-3 expression and its activity. Biochem. Biophys. Res. Commun. 398, 457–461 (2010).
Bowen, C., Zheng, T. & Gelmann, E. P. NKX3.1 Suppresses TMPRSS2-ERG Gene Rearrangement and Mediates Repair of Androgen Receptor-Induced DNA Damage. Cancer Res. 75, 2686–2698 (2015).
Sooreshjani, M. A. et al. LIMK2-NKX3.1 engagement promotes castration-resistant prostate cancer. Cancers 13, 2324 (2021).
Huang, S. et al. Recurrent deletion of CHD1 in prostate cancer with relevance to cell invasiveness. Oncogene 31, 4164–4170 (2012).
Augello, M. A. et al. CHD1 Loss Alters AR binding at lineage-specific enhancers and modulates distinct transcriptional programs to drive prostate Tumorigenesis. Cancer Cell 35, 603–617 e8 (2019).
Kari, V. et al. Loss of CHD1 causes DNA repair defects and enhances prostate cancer therapeutic responsiveness. EMBO Rep. 17, 1609–1623 (2016).
Zhou, J. et al. Human CHD1 is required for early DNA-damage signaling and is uniquely regulated by its N terminus. Nucleic Acids Res. 46, 3891–3905 (2018).
Shenoy, T. R. et al. CHD1 loss sensitizes prostate cancer to DNA damaging therapy by promoting error-prone double-strand break repair. Ann. Oncol. 28, 1495–1507 (2017).
Blattner, M. et al. SPOP mutations in prostate cancer across demographically diverse patient cohorts. Neoplasia 16, 14–20 (2014).
Kaffenberger, S. D. & Barbieri, C. E. Molecular subtyping of prostate cancer. Curr. Opin. Urol. 26, 213–218 (2016).
Jenkins, R. B. et al. Detection of c-myc oncogene amplification and chromosomal anomalies in metastatic prostatic carcinoma by fluorescence in situ hybridization. Cancer Res. 57, 524–531 (1997).
Qiu, X. et al. MYC drives aggressive prostate cancer by disrupting transcriptional pause release at androgen receptor targets. Nat. Commun. 13, 2559 (2022).
Fromont, G. et al. 8q24 amplification is associated with Myc expression and prostate cancer progression and is an independent predictor of recurrence after radical prostatectomy. Hum. Pathol. 44, 1617–1623 (2013).
Bastacky, S. et al. Use of interphase fluorescence in situ hybridization in prostate needle biopsy specimens with isolated high-grade prostatic intraepithelial neoplasia as a predictor of prostate adenocarcinoma on follow-up biopsy. Hum. Pathol. 35, 281–289 (2004).
Jin, W. H. et al. The Molecular, Immunologic, and Clinicodemographic Landscape of MYC-Amplified Advanced Prostate Cancer. Clin. Genitourin. Cancer 22, e163–e169.e1 (2024).
Fujita, K. & Nonomura, N. Role of Androgen Receptor in Prostate Cancer: A Review. World J. Mens. Health 37, 288–295 (2019).
Quistini, A. et al. Androgen Receptor Signalling in Prostate Cancer: Mechanisms of Resistance to Endocrine Therapies. Res. Rep. Urol. 17, 211–223 (2025).
Azad, A. A. et al. Androgen Receptor Gene Aberrations in Circulating Cell-Free DNA: Biomarkers of Therapeutic Resistance in Castration-Resistant Prostate Cancer. Clin. Cancer Res. 21, 2315–2324 (2015).
Anand, S. et al. MYB and HIF1alpha crosstalk drives hypoxia-induced transcriptional reprogramming and adaptive signaling alterations in pancreatic cancer. Cancer Lett. 631, 217916 (2025).
Anand, S. et al. From modulation of cellular plasticity to potentiation of therapeutic resistance: new and emerging roles of MYB transcription factors in human malignancies. Cancer Metastas. Rev. 43, 409–421 (2024).
Edwards, J. et al. Gene amplifications associated with the development of hormone-resistant prostate cancer. Clin. Cancer Res. 9, 5271–5281 (2003).
Srivastava, S. K. et al. Myb overexpression overrides androgen depletion-induced cell cycle arrest and apoptosis in prostate cancer cells, and confers aggressive malignant traits: potential role in castration resistance. Carcinogenesis 33, 1149–1157 (2012).
Srivastava, S. K. et al. MYB interacts with androgen receptor, sustains its ligand-independent activation and promotes castration resistance in prostate cancer. Br. J. Cancer 126, 1205–1214 (2022).
Khan, M. A. et al. MYB exhibits racially disparate expression, clinicopathologic association, and predictive potential for biochemical recurrence in prostate cancer. iScience 26, 108487 (2023).
Lu, X. et al. An In Vivo Screen Identifies PYGO2 as a Driver for Metastatic Prostate Cancer. Cancer Res. 78, 3823–3833 (2018).
Andrews, P. G. & Kao, K. R. Wnt/beta-catenin-dependent acetylation of Pygo2 by CBP/p300 histone acetyltransferase family members. Biochem. J. 473, 4193–4203 (2016).
Ling, J. et al. Pygo2 activates BRPF1 via Pygo2-H3K4me2/3 interaction to maintain malignant progression in colon cancer. Exp. Cell Res. 431, 113696 (2023).
Andrews, P. G. P. et al. Augmentation of Myc-Dependent Mitotic Gene Expression by the Pygopus2 Chromatin Effector. Cell Rep. 23, 1516–1529 (2018).
Kao, K. R. et al. PYGOPUS2 expression in prostatic adenocarcinoma is a potential risk stratification marker for PSA progression following radical prostatectomy. J. Clin. Pathol. 71, 402–411 (2018).
Zhu, Y. et al. Targeting the chromatin effector Pygo2 promotes cytotoxic T cell responses and overcomes immunotherapy resistance in prostate cancer. Sci. Immunol. 8, eade4656 (2023).
Melling, N. et al. Overexpression of enhancer of zeste homolog 2 (EZH2) characterizes an aggressive subset of prostate cancers and predicts patient prognosis independently from pre- and postoperatively assessed clinicopathological parameters. Carcinogenesis 36, 1333–1340 (2015).
Henrique, R. & Jeronimo, C. Molecular detection of prostate cancer: a role for GSTP1 hypermethylation. Eur. Urol. 46, 660–669 (2004). discussion 669.
Mian, O. Y. et al. GSTP1 Loss results in accumulation of oxidative DNA base damage and promotes prostate cancer cell survival following exposure to protracted oxidative stress. Prostate 76, 199–206 (2016).
Kanwal, R. et al. Protection against oxidative DNA damage and stress in human prostate by glutathione S-transferase P1. Mol. Carcinog. 53, 8–18 (2014).
Kumar, B. & Lupold, S. E. MicroRNA expression and function in prostate cancer: a review of current knowledge and opportunities for discovery. Asian J. Androl. 18, 559–567 (2016).
Acharya, S. et al. Biphasic transcriptional and posttranscriptional regulation of MYB by androgen signaling mediates its growth control in prostate cancer. J. Biol. Chem. 299, 102725 (2023).
Holtrop, P., Swails, T. & Riggs, T. Hypertriglyceridemia in extremely low birth weight infants receiving lipid emulsions. J. Neonatal Perinat. Med. 8, 133–136 (2015).
Kumar, B. et al. Cell-type specific expression of oncogenic and tumor suppressive microRNAs in the human prostate and prostate cancer. Sci. Rep. 8, 7189 (2018).
Smith, B., Agarwal, P. & Bhowmick, N. A. MicroRNA applications for prostate, ovarian and breast cancer in the era of precision medicine. Endocr. Relat. Cancer 24, R157–R172 (2017).
Doldi, V. et al. Dissecting the role of microRNAs in prostate cancer metastasis: implications for the design of novel therapeutic approaches. Cell Mol. Life Sci. 73, 2531–2542 (2016).
Zenner, M. L. et al. Prostate-derived circulating microRNAs add prognostic value to prostate cancer risk calculators. J. Extracell. Biol. 2, e122 (2023).
Rodarte, K. E. et al. Neuroendocrine Differentiation in Prostate Cancer Requires ASCL1. Cancer Res. 84, 3522–3537 (2024).
Russo, M. V. et al. SOX2 boosts major tumor progression genes in prostate cancer and is a functional biomarker of lymph node metastasis. Oncotarget 7, 12372–12385 (2016).
Wang, Z. et al. FOXA2 rewires AP-1 for transcriptional reprogramming and lineage plasticity in prostate cancer. Nat. Commun. 15, 4914 (2024).
Rotinen, M. et al. ONECUT2 is a targetable master regulator of lethal prostate cancer that suppresses the androgen axis. Nat. Med. 24, 1887–1898 (2018).
Bishop, J. L. et al. The Master Neural Transcription Factor BRN2 is an androgen receptor-suppressed driver of neuroendocrine differentiation in prostate cancer. Cancer Discov. 7, 54–71 (2017).
Imamura, J. et al. Lineage plasticity and treatment resistance in prostate cancer: the intersection of genetics, epigenetics, and evolution. Front. Endocrinol. 14, 1191311 (2023).
Dardenne, E. et al. N-Myc Induces an EZH2-Mediated Transcriptional Program Driving Neuroendocrine Prostate Cancer. Cancer Cell 30, 563–577 (2016).
Liu, B. et al. PARP Inhibition Suppresses GR-MYCN-CDK5-RB1-E2F1 Signaling and Neuroendocrine Differentiation in Castration-Resistant Prostate Cancer. Clin. Cancer Res. 25, 6839–6851 (2019).
Maylin, Z. R. et al. Therapeutic exploitation of neuroendocrine transdifferentiation drivers in prostate cancer. Cells 13, 1999 (2024).
Abida, W. et al. Analysis of the prevalence of microsatellite instability in prostate cancer and response to immune checkpoint blockade. JAMA Oncol. 5, 471–478 (2019).
Klumper, N. et al. The Role of Microsatellite Instability/DNA mismatch repair deficiency and tumor mutational burden as biomarkers in predicting response to immunotherapy in castration-resistant prostate cancer. Eur. Urol. 86, 388–390 (2024).
Lenis, A. T. et al. Microsatellite instability, tumor mutational burden, and response to immune checkpoint blockade in patients with prostate cancer. Clin. Cancer Res. 30, 3894–3903 (2024).
Mateo, J. et al. DNA-Repair Defects and Olaparib in Metastatic Prostate Cancer. N. Engl. J. Med. 373, 1697–1708 (2015).
Karlsson, Q. et al. Rare Germline Variants in ATM Predispose to Prostate Cancer: A PRACTICAL Consortium Study. Eur. Urol. Oncol. 4, 570–579 (2021).
Stolarova, L. et al. CHEK2 germline variants in cancer predisposition: stalemate rather than checkmate. Cells 9, 2675 (2020).
Nyberg, T., Tischkowitz, M. & Antoniou, A. C. BRCA1 and BRCA2 pathogenic variants and prostate cancer risk: systematic review and meta-analysis. Br. J. Cancer 126, 1067–1081 (2022).
Ledet, E. M. et al. Comparison of germline mutations in African American and Caucasian men with metastatic prostate cancer. Prostate 81, 433–439 (2021).
Nyberg, T. et al. Prostate Cancer Risks for Male BRCA1 and BRCA2 Mutation Carriers: A Prospective Cohort Study. Eur. Urol. 77, 24–35 (2020).
Agalliu, I. et al. Associations of high-grade prostate cancer with BRCA1 and BRCA2 founder mutations. Clin. Cancer Res. 15, 1112–1120 (2009).
Oh, M. et al. The association of BRCA1 and BRCA2 mutations with prostate cancer risk, frequency, and mortality: A meta-analysis. Prostate 79, 880–895 (2019).
Nyberg, T. et al. Homeobox B13 G84E Mutation and Prostate Cancer Risk. Eur. Urol. 75, 834–845 (2019).
Ewing, C. M. et al. Germline mutations in HOXB13 and prostate-cancer risk. N. Engl. J. Med. 366, 141–149 (2012).
Karlsson, R. et al. A population-based assessment of germline HOXB13 G84E mutation and prostate cancer risk. Eur. Urol. 65, 169–176 (2014).
Darst, B. F. et al. Germline Sequencing DNA Repair Genes in 5545 Men With Aggressive and Nonaggressive Prostate Cancer. J. Natl. Cancer Inst. 113, 616–625 (2021).
Zhang, X. et al. ATM-AMPKalpha mediated LAG-3 expression suppresses T cell function in prostate cancer. Cell Immunol. 393-394, 104773 (2023).
Wu, Y. et al. A comprehensive evaluation of CHEK2 germline mutations in men with prostate cancer. Prostate 78, 607–615 (2018).
Cybulski, C. et al. A novel founder CHEK2 mutation is associated with increased prostate cancer risk. Cancer Res. 64, 2677–2679 (2004).
Pritchard, C. C. et al. Inherited DNA-Repair Gene Mutations in Men with Metastatic Prostate Cancer. N. Engl. J. Med. 375, 443–453 (2016).
Heijink, A. M. et al. BRCA2 deficiency instigates cGAS-mediated inflammatory signaling and confers sensitivity to tumor necrosis factor-alpha-mediated cytotoxicity. Nat. Commun. 10, 100 (2019).
Hu, M. et al. ATM inhibition enhances cancer immunotherapy by promoting mtDNA leakage and cGAS/STING activation. J. Clin. Invest. 131, e139333 (2021).
Zhao, D. et al. Chromatin Regulator CHD1 Remodels the Immunosuppressive Tumor Microenvironment in PTEN-Deficient Prostate Cancer. Cancer Discov. 10, 1374–1387 (2020).
Vidotto, T. et al. PTEN-deficient prostate cancer is associated with an immunosuppressive tumor microenvironment mediated by increased expression of IDO1 and infiltrating FoxP3+ T regulatory cells. Prostate 79, 969–979 (2019).
Graham, M. K. et al. Convergent alterations in the tumor microenvironment of MYC-driven human and murine prostate cancer. Nat. Commun. 15, 7414 (2024).
Casey, S. C. et al. MYC regulates the antitumor immune response through CD47 and PD-L1. Science 352, 227–231 (2016).
Chesner, L. N. et al. Androgen Receptor Inhibition Increases MHC Class I Expression and Improves Immune Response in Prostate Cancer. Cancer Discov. 15, 481–494 (2025).
Wang, D. et al. IL-1beta Is an Androgen-Responsive Target in Macrophages for Immunotherapy of Prostate Cancer. Adv. Sci. 10, e2206889 (2023).
Hu, Y. M. et al. Elevated Tumor-Associated Androgen Receptor Activity Correlates with Poor Immune Infiltration and Immunotherapy Response across Cancer Types. Cancer Res Commun. 6, 17–35 (2026).
Bhinder, B. et al. Immunogenomic Landscape of Neuroendocrine Prostate Cancer. Clin. Cancer Res 29, 2933–2943 (2023).
Li, S. et al. Defined cellular reprogramming of androgen receptor-active prostate cancer to neuroendocrine prostate cancer. bioRxivp, (2025).
Zennami, K. et al. PDCD4 Is an androgen-repressed tumor suppressor that regulates prostate cancer growth and castration resistance. Mol. Cancer Res. 17, 618–627 (2019).
Xi, J. et al. miR-21 depletion in macrophages promotes tumoricidal polarization and enhances PD-1 immunotherapy. Oncogene 37, 3151–3165 (2018).
Sun, T. et al. MiR-221 promotes the development of androgen independence in prostate cancer cells via downregulation of HECTD2 and RAB1A. Oncogene 33, 2790–2800 (2014).
Tan, S. et al. Exosomal miRNAs in tumor microenvironment. J. Exp. Clin. Cancer Res. 39, 67 (2020).
Yu, E. Y. et al. Germline and Somatic Genomic Testing for Metastatic Prostate Cancer: ASCO Guideline. J. Clin. Oncol. 43, 748–758 (2025).
Antonarakis, E. S. et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N. Engl. J. Med. 371, 1028–1038 (2014).
Scher, H. I. et al. Association of AR-V7 on circulating tumor cells as a treatment-specific biomarker with outcomes and survival in castration-resistant prostate cancer. JAMA Oncol. 2, 1441–1449 (2016).
Zhang, T. et al. Androgen Receptor Splice Variant, AR-V7, as a Biomarker of Resistance to Androgen Axis-Targeted Therapies in Advanced Prostate Cancer. Clin. Genitourin. Cancer 18, 1–10 (2020).
Hille, C. et al. Detection of Androgen Receptor Variant 7 (ARV7) mRNA Levels in EpCAM-Enriched CTC Fractions for Monitoring Response to Androgen Targeting Therapies in Prostate Cancer. Cells. 8, 1067 (2019).
Nyquist, M. D. et al. Combined TP53 and RB1 loss promotes prostate cancer resistance to a spectrum of therapeutics and confers vulnerability to replication stress. Cell Rep. 31, 107669 (2020).
Jamaspishvili, T. et al. Clinical implications of PTEN loss in prostate cancer. Nat. Rev. Urol. 15, 222–234 (2018).
Yoshida, S. et al. TAS3681, an androgen receptor antagonist, prevents drug resistance driven by aberrant androgen receptor signaling in prostate cancer. Mol. Oncol. 18, 1980–2000 (2024).
Bourlon, M. T., Valdez, P. & Castro, E. Development of PARP inhibitors in advanced prostate cancer. Ther. Adv. Med. Oncol. 16, 17588359231221337 (2024).
de Bono, J. et al. Olaparib for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 382, 2091–2102 (2020).
Abida, W. et al. Rucaparib for the Treatment of Metastatic Castration-resistant Prostate Cancer Associated with a DNA Damage Repair Gene Alteration: Final Results from the Phase 2 TRITON2 Study. Eur. Urol. 84, 321–330 (2023).
Clarke, N. W. et al. Abiraterone and Olaparib for Metastatic Castration-Resistant Prostate Cancer. NEJM Evid. 1, EVIDoa2200043 (2022).
Agarwal, N. et al. Talazoparib plus enzalutamide in men with metastatic castration-resistant prostate cancer: final overall survival results from the randomised, placebo-controlled, phase 3 TALAPRO-2 trial. Lancet 406, 447–460 (2025).
Hussain, M. et al. Abiraterone, Olaparib, or Abiraterone + Olaparib in First-Line Metastatic Castration-Resistant Prostate Cancer with DNA Repair Defects (BRCAAway). Clin. Cancer Res. 30, 4318–4328 (2024).
Attard, G. et al. Niraparib and abiraterone acetate plus prednisone for HRR-deficient metastatic castration-sensitive prostate cancer: a randomized phase 3 trial. Nat. Med. 31, 4109–4118 (2025).
Fizazi, K. et al. Capivasertib plus abiraterone in PTEN-deficient metastatic hormone-sensitive prostate cancer: CAPItello-281 Phase III study. Ann. Oncol. 37, 53–68 (2026).
Rescigno, P. et al. Capivasertib in combination with enzalutamide for metastatic castration resistant prostate cancer after docetaxel and abiraterone: Results from the randomized phase II RE-AKT trial. Eur. J. Cancer 205, 114103 (2024).
Crabb, S. J. et al. Pan-AKT Inhibitor Capivasertib With Docetaxel and Prednisolone in Metastatic Castration-Resistant Prostate Cancer: A Randomized, Placebo-Controlled Phase II Trial (ProCAID). J. Clin. Oncol. 39, 190–201 (2021).
AstraZeneca, TRUQAP® (capivasertib) combination in PTEN-deficient metastatic hormone-sensitive prostate cancer demonstrated statistically significant and clinically meaningful improvement in radiographic progression-free survival in CAPItello-281 Phase III trial. AstraZeneca Media. (2024).
Sweeney, C. et al. Ipatasertib plus abiraterone and prednisolone in metastatic castration-resistant prostate cancer (IPATential150): a multicentre, randomised, double-blind, phase 3 trial. Lancet 398, 131–142 (2021).
de Bono, J. S. et al. Final Overall Survival and Molecular Data Associated with Clinical Outcomes in Patients Receiving Ipatasertib and Abiraterone in the Phase 3 IPATential150 Trial. Eur. Urol. 87, 672–682 (2025).
Michael Thomas Schweizer, K.P., Atish Dipankar Choudhury, Emiliano Calvo, Richard C. Frank, Li Liu, Rajendar K. Mittapalli, Jessica Tougias, Claudia Andreu-Vieyra, Timothy Geoffrey Bowler, Neelesh Soman, and Benjamin Garmezy, Phase 1 trial of mevrometostat (PF-06821497), a potent and selective inhibitor of enhancer of zeste homolog 2 (EZH2), in castration-resistant prostate cancer (CRPC). in 2024 ASCO Annual MeetingJ. Clin. Oncol. (2024).
Aggarwal, R. et al. Safety and Efficacy of Tarlatamab in Patients with Neuroendocrine Prostate Cancer: Results from the Phase 1b DeLLpro-300 Study. Clin. Cancer Res. 31, 3854–3863 (2025).
Dorff, T. B. et al. PSCA-CAR T cell therapy in metastatic castration-resistant prostate cancer: a phase 1 trial. Nat. Med. 30, 1636–1644 (2024).
Narayan, V. et al. PSMA-targeting TGFbeta-insensitive armored CAR T cells in metastatic castration-resistant prostate cancer: a phase 1 trial. Nat. Med. 28, 724–734 (2022).
Le, D. T. et al. Pembrolizumab for previously treated, microsatellite instability-high/mismatch repair-deficient advanced colorectal cancer: final analysis of KEYNOTE-164. Eur. J. Cancer 186, 185–195 (2023).
Graham, L. S. et al. Mismatch repair deficiency in metastatic prostate cancer: Response to PD-1 blockade and standard therapies. PLoS One 15, e0233260 (2020).
Wu, Y. M. et al. Inactivation of CDK12 Delineates a Distinct Immunogenic Class of Advanced Prostate Cancer. Cell 173, 1770–1782.e14 (2018).
Higa, K. et al. Efficacy of pembrolizumab in MSI-high and BRCA-positive castration-resistant prostate cancer. Cancer Genet. 296-297, 41–44 (2025).
Peyraud, F. & Italiano, A. Combined PARP inhibition and immune checkpoint therapy in solid tumors. Cancers 12, 1502 (2020).
Rescigno, P. et al. PERSEUS1: An Open-label, Investigator-initiated, Single arm, Phase 2 Trial Testing the Efficacy of Pembrolizumab in Patients with Metastatic Castration-resistant Prostate Cancer with Mismatch Repair Deficiency and Other Immune-sensitive Molecular Subtypes. Eur. Urol. Oncol. 8, 1059–1069 (2025).
Sharma, P. et al. Nivolumab Plus Ipilimumab for Metastatic Castration-Resistant Prostate Cancer: Preliminary Analysis of Patients in the CheckMate 650 Trial. Cancer Cell 38, 489–499.e3 (2020).
Leone, G. et al. Nivolumab and Ipilimumab for Metastatic Castration-Resistant Prostate Cancer With an Immunogenic Signature: The Multicenter, Two-Cohort, Phase II NEPTUNES Study. J. Clin. Oncol. 43, 3070–3080 (2025).
You, G. et al. B7-H3x4-1BB bispecific antibody augments antitumor immunity by enhancing terminally differentiated CD8(+) tumor-infiltrating lymphocytes. Sci. Adv. 7, eaax3160 (2021).
Aggarwal, C. et al. Dual checkpoint targeting of B7-H3 and PD-1 with enoblituzumab and pembrolizumab in advanced solid tumors: interim results from a multicenter phase I/II trial. J. Immunother. Cancer 10, e004424 (2022).
Shi, W. et al. Immune checkpoint B7-H3 is a therapeutic vulnerability in prostate cancer harboring PTEN and TP53 deficiencies. Sci. Transl. Med. 15, eadf6724 (2023).
Sartor, O. et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 385, 1091–1103 (2021).
Fallah, J. et al. FDA Approval Summary: Lutetium Lu 177 Vipivotide Tetraxetan for Patients with Metastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res 29, 1651–1657 (2023).
Boixareu, C. et al. Targeting the tumour cell surface in advanced prostate cancer. Nat. Rev. Urol. 22, 569–589 (2025).
Chen, B. et al. Antibody-drug conjugates in cancer therapy: current landscape, challenges, and future directions. Mol. Cancer 24, 279 (2025).
Giugliano, F. et al. Bystander effect of antibody-drug conjugates: fact or fiction? Curr. Oncol. Rep. 24, 809–817 (2022).
Petrylak, D. P. et al. Phase 1 study of PSMA ADC, an antibody-drug conjugate targeting prostate-specific membrane antigen, in chemotherapy-refractory prostate cancer. Prostate 79, 604–613 (2019).
Petrylak, D. P. et al. PSMA ADC monotherapy in patients with progressive metastatic castration-resistant prostate cancer following abiraterone and/or enzalutamide: Efficacy and safety in open-label single-arm phase 2 study. Prostate 80, 99–108 (2020).
Carrasquillo, J. A. et al. Imaging Patients with Metastatic Castration-Resistant Prostate Cancer Using (89)Zr-DFO-MSTP2109A Anti-STEAP1 Antibody. J. Nucl. Med 60, 1517–1523 (2019).
Danila, D. C. et al. Phase I Study of DSTP3086S, an Antibody-Drug Conjugate Targeting Six-Transmembrane Epithelial Antigen of Prostate 1, in Metastatic Castration-Resistant Prostate Cancer. J. Clin. Oncol. 37, 3518–3527 (2019).
Bardia, A. et al. Sacituzumab govitecan, a Trop-2-directed antibody-drug conjugate, for patients with epithelial cancer: final safety and efficacy results from the phase I/II IMMU-132-01 basket trial. Ann. Oncol. 32, 746–756 (2021).
Sardinha, M. et al. Antibody-Drug Conjugates in Prostate Cancer: A Systematic Review. Cureus 15, e34490 (2023).
Doi, T. et al. 453O DS-7300 (B7-H3 DXd antibody-drug conjugate [ADC]) shows durable antitumor activity in advanced solid tumors: Extended follow-up of a phase I/II study. Ann. Oncol. 33, S744–S745 (2022).
de Bono, J. S. et al. 1654P TAMARACK: Randomized Phase II trial of the B7-H3 targeting antibody drug conjugate (ADC) vobramitamab duocarmazine (vobra duo) in metastatic castration-resistant prostate cancer (mCRPC). Ann. Oncol. 35, S996–S997 (2024).
Corn, P. G. et al. Cabazitaxel plus carboplatin for the treatment of men with metastatic castration-resistant prostate cancers: a randomised, open-label, phase 1-2 trial. Lancet Oncol. 20, 1432–1443 (2019).
Li, H. et al. PARP inhibitor resistance: the underlying mechanisms and clinical implications. Mol. Cancer 19, 107 (2020).
Lin, K. K. et al. BRCA Reversion Mutations in Circulating Tumor DNA Predict Primary and Acquired Resistance to the PARP Inhibitor Rucaparib in High-Grade Ovarian Carcinoma. Cancer Discov. 9, 210–219 (2019).
Hofstad, M. et al. Dual inhibition of ATR and DNA-PKcs radiosensitizes ATM-mutant prostate cancer. Oncogene 44, 1746–1760 (2025).
Rafiei, S. et al. ATM Loss Confers Greater Sensitivity to ATR Inhibition Than PARP Inhibition in Prostate Cancer. Cancer Res. 80, 2094–2100 (2020).
Fatteh, M. et al. Poly (ADP-ribose) Polymerase Inhibitor Resistance Driven by Emergence of Polyclonal Mutations With Convergent Evolution: A Molecular Tumor Board Discussion. JCO Precis. Oncol. 8, e2400254 (2024).
Harvey-Jones, E. et al. Longitudinal profiling identifies co-occurring BRCA1/2 reversions, TP53BP1, RIF1 and PAXIP1 mutations in PARP inhibitor-resistant advanced breast cancer. Ann. Oncol. 35, 364–380 (2024).
Derlin, T. et al. PSMA-heterogeneity in metastatic castration-resistant prostate cancer: Circulating tumor cells, metastatic tumor burden, and response to targeted radioligand therapy. Prostate 83, 1076–1088 (2023).
Current, K. et al. Investigating PSMA-Targeted Radioligand Therapy Efficacy as a Function of Cellular PSMA Levels and Intratumoral PSMA Heterogeneity. Clin. Cancer Res. 26, 2946–2955 (2020).
Bakht, M. K. & Beltran, H. Biological determinants of PSMA expression, regulation and heterogeneity in prostate cancer. Nat. Rev. Urol. 22, 26–45 (2025).
Beltran, H. et al. The Role of Lineage Plasticity in Prostate Cancer Therapy Resistance. Clin. Cancer Res 25, 6916–6924 (2019).
Thang, S. P. et al. Poor Outcomes for Patients with Metastatic Castration-resistant Prostate Cancer with Low Prostate-specific Membrane Antigen (PSMA) Expression Deemed Ineligible for (177)Lu-labelled PSMA Radioligand Therapy. Eur. Urol. Oncol. 2, 670–676 (2019).
Beshiri, M. et al. Prostate organoids: emerging experimental tools for translational research. J. Clin. Invest.133, e169616 (2023).
Wong, E. Y., Chu, T. N. & Ladi-Seyedian, S. S. Genomics and Artificial Intelligence: Prostate Cancer. Urol. Clin. North Am. 51, 27–33 (2024).
Yan, L., Su, P. & Sun, X. Role of multi‑omics in advancing the understanding and treatment of prostate cancer (Review). Mol. Med. Rep. 31, 130 (2025).
Yu, X. et al. Single-cell omics traces the heterogeneity of prostate cancer cells and the tumor microenvironment. Cell Mol. Biol. Lett. 28, 38 (2023).
Mei, S. et al. Single-cell and spatial transcriptomics reveal a tumor-associated macrophage subpopulation that mediates prostate cancer progression and metastasis. Mol. Cancer Res. 23, 653–665 (2025).
Figiel, S. et al. Spatial transcriptomic analysis of virtual prostate biopsy reveals confounding effect of tissue heterogeneity on genomic signatures. Mol. Cancer 22, 162 (2023).
Casanova-Salas, I. et al. Quantitative and qualitative analysis of blood-based liquid biopsies to inform clinical decision-making in prostate cancer. Eur. Urol. 79, 762–771 (2021).
Zhang, X. et al. Genetic and epigenetic features of neuroendocrine prostate cancer and their emerging applications. Int Rev. Cell Mol. Biol. 383, 41–66 (2024).
Bevere, M. et al. An overview of circulating biomarkers in neuroendocrine neoplasms: a clinical guide. Diagnostics 13, 2820 (2023).
Zhao, S. G. et al. A clinical-grade liquid biomarker detects neuroendocrine differentiation in prostate cancer. J. Clin. Invest. 132, e161858 (2022).
Crocetto, F. et al. Liquid biopsy in prostate cancer management-current challenges and future perspectives. Cancers 14, 3272 (2022).
Acknowledgements
All figures in this manuscript were created with BioRender.com. We thank fellow clinicians, researchers, and all other members of the GU Cancer Working Group at the UMMC CCRI for their insightful discussion on the components presented in this manuscript. We would also like to acknowledge the funding support from the Department of Defense (HT9425-23-1-0452; W81XWH-22-1-0913) and the Cancer Center and Research Institute.
Author information
Authors and Affiliations
Contributions
Ajay P. Singh and John C. Henegan conceived and designed the manuscript. John A. Ligon conducted the literature review, performed data analysis, and prepared the first draft. Shashi Anand, Seema Singh, Jawed Siddiqui, John C. Henegan, and Ajay P. Singh reviewed the draft and suggested revisions, including the addition of relevant literature. Shashi Anand and Seema Singh contributed to the graphic design. John A. Ligon and Shashi Anand prepared the final revised version of the manuscript. All authors reviewed and approved the final manuscript for submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ligon, J.A., Anand, S., Singh, S. et al. Genomic landscape and precision therapy in prostate cancer: current status and future directions. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01368-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01368-3