Abstract
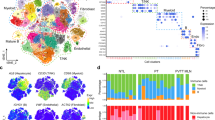
Hepatocellular carcinoma (HCC) is one of the most lethal malignancies worldwide, and its progression is closely linked to the establishment of an immunosuppressive tumor microenvironment. Myristoylated alanine-rich C kinase substrate (MARCKS) has been implicated in tumor biology; however, its role in regulating immune interactions in HCC remains poorly defined. Here, we performed an integrated multi-omics analysis combining bulk transcriptomics, single-cell RNA sequencing, and spatial transcriptomics to systematically investigate the expression pattern and functional relevance of MARCKS in HCC. We found that MARCKS was significantly upregulated in HCC tissues and that high MARCKS expression was associated with aggressive clinicopathological features and unfavorable prognosis. Single-cell and spatial analyses revealed that MARCKS expression was enriched in myeloid cell populations within the tumor microenvironment. Functional annotation and mIF(Multiple immunofluorescence) validation demonstrated that MARCKS expression was associated with enhanced JAK/STAT3 signaling and M2-like macrophage polarization. Consistently, MARCKS silencing in HCC cell lines reduced STAT3 phosphorylation, suppressed malignant phenotypes in vitro, inhibited tumor growth in vivo, and diminished the capacity of tumor-derived conditioned media to promote macrophage M2 polarization. Together, these findings identify MARCKS as a key regulator of the immunosuppressive tumor microenvironment in HCC and highlight its potential as a therapeutic target for overcoming immune evasion.

Similar content being viewed by others
Data availability
RNA-seq raw count data, clinical information, and somatic mutation (MAF) files for HCC were obtained from the TCGA-LIHC cohort via the Genomic Data Commons (https://portal.gdc.cancer.gov/projects/TCGA-LIHC; dbGaP: phs000178). Pan-cancer RNA-seq data integrating TCGA and GTEx were retrieved from the UCSC Xena Toil recompute dataset (https://xenabrowser.net/). The single-cell RNA-seq dataset GSE149614 was downloaded from GEO (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE149614), and the spatial transcriptomics dataset HRA000437 was obtained from NGDC (https://ngdc.cncb.ac.cn/gsa-human/browse/HRA000437).
Code availability
The custom scripts and codes used for data processing and analysis are available from the corresponding author upon reasonable request.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Llovet, J. M. et al. Locoregional therapies in the era of molecular and immune treatments for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 18, 293–313 (2021).
Sonbol, M. B. et al. Systemic therapy and sequencing options in advanced hepatocellular carcinoma: a systematic review and network meta-analysis. JAMA Oncol. 6, e204930 (2020).
Vogel, A. & Martinelli, E. ESMO Guidelines Committee. Updated treatment recommendations for hepatocellular carcinoma (HCC) from the ESMO Clinical Practice Guidelines. Ann. Oncol. 32, 801–805 (2021).
He, H. et al. Multi-dimensional single-cell characterization revealed suppressive immune microenvironment in AFP-positive hepatocellular carcinoma. Cell Discov. 9, 60 (2023).
Wang, Y. F. et al. Spatial maps of hepatocellular carcinoma transcriptomes reveal spatial expression patterns in tumor immune microenvironment. Theranostics 12, 4163–4180 (2022).
Chen, S. et al. Distinct single-cell immune ecosystems distinguish true and de novo HBV-related hepatocellular carcinoma recurrences. Gut 72, 1196–1210 (2023).
Shen, H. et al. Prognostic biomarker MITD1 and its correlation with immune infiltrates in hepatocellular carcinoma (HCC). Int. Immunopharmacol. 81, 106222 (2020).
Xiang, X., Wang, J., Lu, D. & Xu, X. Targeting tumor-associated macrophages to synergize tumor immunotherapy. Signal Transduct. Target. Ther. 6, 75 (2021).
Chen, D., Zhang, X., Li, Z. & Zhu, B. Metabolic regulatory crosstalk between tumor microenvironment and tumor-associated macrophages. Theranostics 11, 1016–1030 (2021).
Chu, X., Tian, Y. & Lv, C. Decoding the spatiotemporal heterogeneity of tumor-associated macrophages. Mol. Cancer 23, 150 (2024).
Li, C. et al. Tumor-associated macrophages: potential therapeutic strategies and future prospects in cancer. J. Immunother. Cancer 9, e001341 (2021).
Qian, J., Li, J., Ma, H. & Ji, W. Exosomal circ-ADRM1 promotes lung adenocarcinoma progression and induces macrophage M2 polarization through regulating MMP14 mRNA and protein. Anticancer Drugs 34, 333–343 (2023).
Ruf, B. et al. Tumor-associated macrophages trigger MAIT cell dysfunction at the HCC invasive margin. Cell 186, 3686–3705.e32 (2023).
Cheng, K. et al. Tumor-associated macrophages in liver cancer: from mechanisms to therapy. Cancer Commun. 42, 1112–1140 (2022).
El Amri, M., Fitzgerald, U. & Schlosser, G. MARCKS and MARCKS-like proteins in development and regeneration. J. Biomed. Sci. 25, 43 (2018).
Chen, Z. et al. The myristoylated alanine-rich C-kinase substrates (MARCKS): a membrane-anchored mediator of the cell function. Autoimmun. Rev. 20, 102942 (2021).
Beckmann, L. et al. MARCKS affects cell motility and response to BTK inhibitors in CLL. Blood 138, 544–556 (2021).
Bickeböller, M. et al. Functional characterization of the tumor-suppressor MARCKS in colorectal cancer and its association with survival. Oncogene 34, 1150–1159 (2015).
Huang, X. Z. et al. Single-cell sequencing of ascites fluid illustrates heterogeneity and therapy-induced evolution during gastric cancer peritoneal metastasis. Nat. Commun. 14, 822 (2023).
Zhang, L. et al. MARCKS inhibition cooperates with autophagy antagonists to potentiate the effect of standard therapy against drug-resistant multiple myeloma. Cancer Lett. 480, 29–38 (2020).
Reddy, S. P., Natarajan, V. & Dudek, A. Z. MARCKS is marked in combating lung cancer growth and acquired resistance. Am. J. Respir. Crit. Care Med. 190, 1084–1086 (2014).
Chen, C. H. et al. Upregulation of MARCKS in kidney cancer and its potential as a therapeutic target. Oncogene 36, 3588–3598 (2017).
Naboulsi, W. et al. Quantitative tissue proteomics analysis reveals versican as potential biomarker for early-stage hepatocellular carcinoma. J. Proteome Res. 15, 38–47 (2016).
Masaki, T. et al. Comparison study of the expressions of myristoylated alanine-rich C kinase substrate in hepatocellular carcinoma, liver cirrhosis, chronic hepatitis, and normal liver. Int. J. Oncol. 26, 661–671 (2005).
Lu, H. et al. MARCKS is a new prognostic biomarker in hepatocellular carcinoma. Int. J. Gen. Med. 16, 1603–1619 (2023).
Ren, X. et al. MARCKS on tumor-associated macrophages is correlated with immune infiltrates and poor prognosis in hepatocellular carcinoma. Cancer Invest 39, 756–768 (2021).
Vogel, A., Meyer, T., Sapisochin, G., Salem, R. & Saborowski, A. Hepatocellular carcinoma. Lancet 400, 1345–1362 (2022).
Donne, R. & Lujambio, A. The liver cancer immune microenvironment: therapeutic implications for hepatocellular carcinoma. Hepatology 77, 1773–1796 (2023).
Rimassa, L., Finn, R. S. & Sangro, B. Combination immunotherapy for hepatocellular carcinoma. J. Hepatol. 79, 506–515 (2023).
Greten, T. F. et al. Biomarkers for immunotherapy of hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 20, 780–798 (2023).
Wang, Z., Wang, Y., Gao, P. & Ding, J. Immune checkpoint inhibitor resistance in hepatocellular carcinoma. Cancer Lett. 555, 216038 (2023).
Gao, J., Wang, W. Q., Pei, Q., Lord, M. S. & Yu, H. J. Engineering nanomedicines through boosting immunogenic cell death for improved cancer immunotherapy. Acta Pharm. Sin. 41, 986–994 (2020).
Pitt, J. M. et al. Targeting the tumor microenvironment: removing obstruction to anticancer immune responses and immunotherapy. Ann. Oncol. 27, 1482–1492 (2016).
Mellman, I., Chen, D. S., Powles, T. & Turley, S. J. The cancer-immunity cycle: indication, genotype, and immunotype. Immunity 56, 2188–2205 (2023).
Ren, X. et al. Insights gained from single-cell analysis of immune cells in the tumor microenvironment. Annu. Rev. Immunol. 39, 583–609 (2021).
Fu, T. et al. Spatial architecture of the immune microenvironment orchestrates tumor immunity and therapeutic response. J. Hematol. Oncol 14, 98 (2021).
Wu, J. et al. A noninvasive approach to evaluate tumor immune microenvironment and predict outcomes in hepatocellular carcinoma. Phenomics 3, 549–564 (2023).
Shao, K. et al. Copper transporter gene ATP7A: a predictive biomarker for immunotherapy and targeted therapy in hepatocellular carcinoma. Int. Immunopharmacol. 114, 109518 (2023).
Li, Y. et al. CircTRIM1 encodes TRIM1-269aa to promote chemoresistance and metastasis of TNBC via enhancing CaM-dependent MARCKS translocation and PI3K/AKT/mTOR activation. Mol. Cancer 23, 102 (2024).
Yang, Y. et al. Targeting phospho-MARCKS overcomes drug resistance and induces antitumor activity in preclinical models of multiple myeloma. Leukemia 29, 715–726 (2015).
Niewerth, D. et al. Molecular basis of resistance to proteasome inhibitors in hematological malignancies. Drug Resist. Updates 18, 18–35 (2015).
Jarboe, J. S. et al. MARCKS regulates growth and radiation sensitivity and is a novel prognostic factor for glioma. Clin. Cancer Res. J. Am. Assoc. Cancer Res. 18, 3030–3041 (2012).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 82360465,82560579); Guangxi Natural Science Foundation (2024GXNSFDA010029, 2025GXNSFAA069704); Guangxi Key Laboratory of Longevity Science and Technology (Open Fund Project No.gxkllst-20241003; Guangxi Science and Technology Program under Grant No.AD25069077; the Special Fund for Clinical Discipline Construction of Guangxi Medical University (GXMULJZ202402); Innovation Project of Guangxi Graduate Education (No. YCSW2025252); The Guangxi Natural Science Foundation Youth Science Fund Project (2025GXNSFBA069037); Young and Middle-aged Scientific Research Capacity Enhancement Project (2024KY0128) and National Postdoctoral Researcher Funding Program (GZC20230583); The Open Project of Guangxi Key Laboratory of Enhanced Recovery after Surgery for Gastrointestinal Cancer (GXEKL202401); Youth Science Foundation of Guangxi Medical University (GXMUYSF202501).
Author information
Authors and Affiliations
Contributions
Haifei Qin, Qinchen Lu, and Baicheng Hu contributed equally to this work. Haifei Qin, Qinchen Lu, and Baicheng Hu designed and performed most of the experiments and analyses. Chenlu Lan, Honglong Lu, Donghua Gao, Chongjiu Qin, and Kai Peng assisted with data collection, bioinformatics analysis, and figure preparation. Xin Zhou, Yongguang Wei, Xiwen Liao, and Tao Peng provided technical support and contributed to data interpretation. Tao Peng, Liming Shang, and Guangzhi Zhu supervised the project, critically revised the manuscript for important intellectual content, and served as corresponding authors. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qin, H., Lu, Q., Hu, B. et al. Integrated multi-omics analysis reveals that MARCKS reprograms the immunosuppressive microenvironment to drive hepatocellular carcinoma progression. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01372-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01372-7