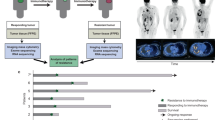
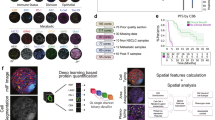
Abstract
Although previous studies have linked body composition to immunotherapy efficacy, comprehensive multidimensional analyses with biological explanations remain lacking. This study integrated eight independent cohorts comprising 2,132 non-small cell lung cancer (NSCLC) patients, including five immune checkpoint inhibitor prognostic cohorts (n = 1,919), two bulk RNA-seq cohorts (n = 190), and one prospective single-cell RNA-seq cohort (n = 23). Using deep learning algorithms, we automatically extracted 92 body composition parameters from computed tomography images. The AI-based segmentation system demonstrated high consistency with manual measurements (intraclass correlation coefficient >0.87) with significantly improved efficiency. In male patients, higher intermuscular fat volume (IMFV) and 14 other indicators were independent predictors of overall survival; in female patients, T12 subcutaneous fat density and 6 other indicators showed potential associations with survival. Male patients with high IMFV exhibited significant upregulation of interferon-related pathways in CD8 + T cells and NK cells, along with lower exhaustion scores, while female patients with high T12 subcutaneous fat density showed macrophage polarization toward the M1 phenotype. This study underscores the importance of multidimensional body composition in NSCLC patient management, demonstrating that specific parameters are not only closely related to survival outcomes but also exhibit unique gender differences and location variations, providing new insights for optimizing immunotherapy strategies.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Sung, H. et al. Global cancer statistics 2020: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Theelen, W. S. M. E. et al. Effect of pembrolizumab after stereotactic body radiotherapy vs pembrolizumab alone on tumor response in patients with advanced non-small cell lung cancer: Results of the PEMBRO-RT phase 2 randomized clinical trial. JAMA Oncol. 5, 1276–1282 (2019).
Guo, G. et al. Immune cell concentrations among the primary tumor microenvironment in colorectal cancer patients predicted by clinicopathologic characteristics and blood indexes. J. Immunother. Cancer 7, 179 (2019).
Nakao S. et al. Intratumoral expression of IL-7 and IL-12 using an oncolytic virus increases systemic sensitivity to immune checkpoint blockade. Sci Transl Med. 12https://doi.org/10.1126/scitranslmed.aax7992 (2020).
Wattenberg, M. M. & Beatty, G. L. Overcoming immunotherapeutic resistance by targeting the cancer inflammation cycle. Semin Cancer Biol. 65, 38–50 (2020).
Chaunzwa, T. L. et al. Body composition in advanced non-small cell lung cancer treated with immunotherapy. JAMA Oncol. 10, 773–783 (2024).
Fleming, C. A. et al. Body composition, inflammation, and 5-year outcomes in colon cancer. JAMA Netw. Open 4, e2115274 (2021).
Berardi, G. et al. Association of sarcopenia and body composition with short-term outcomes after liver resection for malignant tumors. JAMA Surg. 155, e203336 (2020).
Bader, J. E. et al. Obesity induces PD-1 on macrophages to suppress anti-tumour immunity. Nature. 630, 968–975 (2024).
Wang, Z. et al. Paradoxical effects of obesity on T cell function during tumor progression and PD-1 checkpoint blockade. Nat. Med. 25, 141–151 (2019).
Gazourian, L. et al. Quantitative pectoralis muscle area is associated with the development of lung cancer in a large lung cancer screening cohort. Lung. 198, 847–853 (2020).
Cury, S. S. et al. Low muscle mass in lung cancer is associated with an inflammatory and immunosuppressive tumor microenvironment. J. Transl. Med 21, 116 (2023).
Cortellini A. et al. Prognostic effect of body mass index in patients with advanced NSCLC treated with chemoimmunotherapy combinations. J. Immunother Cancer. 10https://doi.org/10.1136/jitc-2021-004374 (2022).
Blanc-Durand, P. et al. Prognostic value of anthropometric measures extracted from whole-body CT using deep learning in patients with non-small-cell lung cancer. Eur. Radio. 30, 3528–3537 (2020).
Takenaka, Y., Oya, R., Takemoto, N. & Inohara, H. Predictive impact of sarcopenia in solid cancers treated with immune checkpoint inhibitors: a meta-analysis. J. Cachexia Sarcopenia Muscle 12, 1122–1135 (2021).
Stern, J. H., Rutkowski, J. M. & Scherer, P. E. Adiponectin, leptin, and fatty acids in the maintenance of metabolic homeostasis through adipose tissue crosstalk. Cell Metab. 23, 770–784 (2016).
Lettieri-Barbato, D. & Aquilano, K. Aging and immunometabolic adaptations to thermogenesis. Ageing Res. Rev. 63, 101143 (2020).
Bekkelund, S. I. & Jorde, R. Alanine aminotransferase and body composition in obese men and women. Dis. Markers. 2019, 1695874 (2019).
Kichenadasse, G. et al. Association between body mass index and overall survival with immune checkpoint inhibitor therapy for advanced non-small cell lung cancer. JAMA Oncol. 6, 512–518 (2020).
Park, J., Euhus, D. M. & Scherer, P. E. Paracrine and endocrine effects of adipose tissue on cancer development and progression. Endocr. Rev. 32, 550–570 (2011).
Zhang, M., Wei, T., Zhang, X. & Guo, D. Targeting lipid metabolism reprogramming of immunocytes in response to the tumor microenvironment stressor: A potential approach for tumor therapy. Front Immunol. 13, 937406 (2022).
Kim, T. K., Vandsemb, E. N., Herbst, R. S. & Chen, L. Adaptive immune resistance at the tumour site: mechanisms and therapeutic opportunities. Nat. Rev. Drug Discov. 21, 529–540 (2022).
Nirschl C. J. et al. IFNγ-dependent tissue-immune homeostasis is co-opted in the tumor microenvironment. Cell. 170https://doi.org/10.1016/j.cell.2017.06.016 (2017).
Sun, D. et al. Classification of tumor immune microenvironment according to programmed death-ligand 1 expression and immune infiltration predicts response to immunotherapy plus chemotherapy in advanced patients with NSCLC. J. Thorac. Oncol. 18, 869–881 (2023).
Huang Z. et al. Effects of sex and aging on the immune cell landscape as assessed by single-cell transcriptomic analysis. Proc. Natl. Acad. Sci. USA. 118https://doi.org/10.1073/pnas.2023216118 (2021).
Oliva M. et al. The impact of sex on gene expression across human tissues. Science. 369https://doi.org/10.1126/science.aba3066 (2020).
Björntorp, P. The regulation of adipose tissue distribution in humans. Int J. Obes. Relat. Metab. Disord. 20, 291–302 (1996).
Fanciulli, G., Delitala, A. & Delitala, G. Growth hormone, menopause and ageing: no definite evidence for ‘rejuvenation’ with growth hormone. Hum. Reprod. Update 15, 341–358 (2009).
Sharma, S., Gibbons, A. & Saphire, E. O. Sex differences in tissue-specific immunity and immunology. Science 389, 599–603 (2025).
Xiao, T. et al. Hallmarks of sex bias in immuno-oncology: mechanisms and therapeutic implications. Nat. Rev. Cancer 24, 338–355 (2024).
Young A. C. et al. Impact of body composition on outcomes from anti-PD1 +/- anti-CTLA-4 treatment in melanoma. J Immunother Cancer. 8https://doi.org/10.1136/jitc-2020-000821 (2020).
Gómez-Banoy N. et al. Body mass index and adiposity influence responses to immune checkpoint inhibition in endometrial cancer. J Clin Invest. 134https://doi.org/10.1172/JCI180516 (2024).
Yu, J. et al. Paradoxical effect of myosteatosis on the immune checkpoint inhibitor response in metastatic renal cell carcinoma. J. Cachexia Sarcopenia Muscle 16, e13758 (2025).
Collins, G. S., Reitsma, J. B., Altman, D. G. & Moons, K. G. M. Transparent Reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD statement. Ann. Intern Med. 162, 55–63 (2015).
Wasserthal, J. et al. TotalSegmentator: Robust segmentation of 104 anatomic structures in CT images. Radio. Artif. Intell. 5, e230024 (2023).
Gong, B. et al. A combined model based on bone mineral density for noninvasive prediction of prognosis in non-small cell lung cancer patients receiving immune checkpoint inhibitors: A multicenter retrospective study. MedComm (2020) 6, e70398 (2025).
Acknowledgements
This work was supported by the Noncommunicable Chronic Diseases-National Science and Technology Major Project (2024ZD0522800/2024ZD0522806), the key project of Hubei provincial Natural Science Foundation (2023BCB014), the National Nature Science Foundation of China (No. 82172034, No. 82472058). The funders of this study had no role in study design, data collection, data analysis, data interpretation, or writing of this report.
Author information
Authors and Affiliations
Contributions
Y.G., B.G., and J.L. conceived the project. Y.G., B.G., J.L., Y.P., Q.S., and S.P. performed investigation. L.W., Y.C., X.L., P.M., Q.W., Q.S., and S.P. assisted in study design and data curation. Y.G., B.G., and J.L. performed data analysis. L.W. collected human samples. Y.G., B.G., and J.L. wrote the original draft of the manuscript. C.Z. and L.Y. were responsible for project management and supervision. L.W., Y.C., X.L., P.M., Q.W., Q.S., S.P., C.Z., and L.Y. reviewed and edited the manuscript. All authors have approved and reviewed the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, Y., Gong, B., Lou, J. et al. AI-driven body composition atlas reveals its association with NSCLC immunotherapy outcome and molecular background: a multicenter study. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01382-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01382-5