Abstract
Exercise and mindfulness-based interventions have growing evidence for managing fatigue and comorbid symptoms; however, packaging them in a cohesive digital way for patients undergoing cancer treatment has not been evaluated. We conducted a randomized controlled trial to assess the impact of a 12 week digital integrative medicine program, Integrative Medicine at Home (IM@Home), versus enhanced usual care on fatigue severity (primary outcome), comorbid symptoms and acute healthcare utilization (secondary outcomes), in 200 patients with solid tumors experiencing fatigue during treatment. Fatigue severity decreased more in IM@Home than in the control (1.99 vs. 1.51 points; p = 0.04). IM@Home participants also had reduced symptom distress (p = 0.003), anxiety (p = 0.03), and depression (p = 0.02). Acute healthcare utilization was lower with IM@Home, with fewer emergency department visits (rate ratio 0.49; p = 0.04), hospitalizations (4% vs. 12.9%; p = 0.03), and shorter hospital stays (4.25 vs. 10 days; p < 0.001). These promising findings should be confirmed in phase III clinical trials. “Study registered at clinicaltrials.gov (NCT05053230) on 09-20-2021”.
Similar content being viewed by others
Introduction
Patients with cancer on active treatment experience a substantial burden from symptoms such as fatigue, anxiety and depression arising from both their disease and the toxicities of treatment1. These symptoms often cluster together, leading to disruptions in treatment and acute care utilization such as emergency department (ED) visits and unplanned hospitalizations2,3. Uncontrolled symptoms for oncology patients on active treatment have been associated with increased acute care utilization4. In particular, fatigue is among the most common presenting symptoms to the ED in patients with cancer and is often associated with other conditions such as nausea, emesis, and dyspnea; it has been associated with worse outcomes for patients with advanced cancer on treatment5,6,7. Recognizing the need to advance the well-being of individuals living with cancer, improved symptom management is widely acknowledged as a key component of quality, patient-centered oncologic care, and it is a primary goal of the Cancer Moonshot initiative8 and National Cancer Institute (NCI) National Cancer Plan9. To date, efforts have focused on digital symptom assessment, while data is more limited regarding the utility of digital medicine interventions10,11,12.
There is growing evidence that integrative medicine interventions, such as mind-body therapies (e.g., mindfulness-based interventions, yoga, and tai chi) and exercise can improve symptom management for patients with cancer13,14,15. Mind-body therapies, which focus on the interconnectedness of the mind, body, and behavior, have demonstrated effectiveness in addressing various cancer-related symptoms (e.g., fatigue, nausea/vomiting)14,16, anxiety and depressive symptoms17, and overall quality of life16,17. Exercise is also recognized as one of the most effective treatments for cancer-related fatigue18. Acknowledging the potential benefits, national practice guidelines from the American Society of Clinical Oncology (ASCO), Society for Integrative Oncology, and National Comprehensive Cancer Network (NCCN) now endorse the integration of mind-body therapies and exercise for symptom management during active cancer care19,20. Despite these recommendations, access to these treatments remains limited for many patients with cancer due to barriers at the patient level (e.g., time, transportation) and system level (e.g., provider resources, staffing, space)21,22. A digital care model enables patients to participate in these therapies from home, potentially overcoming some of these barriers.
While research is very limited on digital integrative medicine programs, a growing body of research in psycho-oncology has demonstrated that digital programs based on evidence-based interventions such as acceptance and commitment therapy (ACT) or cognitive behavioral therapy (CBT) (e.g., STAMP + CBT23, iCanADAPT24,25) can be feasible and effective in addressing psychological distress and expand access to evidence-based mental health services20. Patients with cancer have varying levels of fatigue and functions as well as motivation and preferences for physical activity and integrative medicine programing26,27,28. There is a need to develop a multi-component intervention that offers evidence-based options, addresses the complex and multifaceted symptom burden experienced by oncology patients, and reflects the clinical reality.
In response to the challenges of the COVID-19 pandemic and the need for effective symptom mitigation and patient support strategies, we developed a digital, synchronous (live) mind-body and exercise intervention, Integrative Medicine @ Home (IM@Home). We previously demonstrated that the program was feasible, scalable, and garnered high patient satisfaction29. We then conducted a single-blind, basket randomized controlled trial (RCT) to evaluate the effect of IM@Home on fatigue, comorbid symptoms, and acute healthcare utilization. We hypothesized that during the 12-week intervention patients in IM@Home would experience greater improvements in fatigue (primary outcome) and comorbid symptoms and reduced acute healthcare utilization compared to the control group.
Results
Patient population
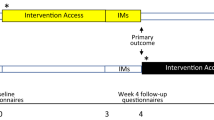
From October 2021 through March 2023, we screened 609 patients, of whom 409 were excluded (see Fig. 1). Of the 200 patients enrolled, 99 were randomized to IM@Home and 101 to the enhanced usual care (EUC) control group. For the patient reported outcomes (PRO) measures models, 192 patients were analyzed (95 IM@Home, 97 EUC), as 8 (4 per arm) provided no PRO data at any of the timepoints. All 200 patients were included in the analysis of acute healthcare utilization.
This figure presents a consort diagram detailing the participant flow through the IMPROVE trial, including screening, randomization, allocation, follow-up, and analysis.
The mean age at enrollment was 59.9 years (range, 29 to 90 years); 181 patients (90.5%) were female, 155 (77.5%) white, 18 (9.0%) Black, 15 (7.5%) Asian, and 178 (89.0%) non-Hispanic/Latino. By cancer subtype, 73 patients (36.5%) had breast, 49 (24.5%) thoracic, 43 (21.5%) gynecologic, 25 (12.5%) head/neck, and 10 (5.0%) had melanoma. Other demographic and clinical characteristics are described in Table 1.
Engagement with the Intervention
Of 99 patients in the IM@Home group, 88 (88.9%) registered for classes. The majority of participants chose to register for 3 classes a week (31, 35.2%), followed by 4 classes per week (21, 23.9%), 2 classes per week (15, 17.0%), 5 classes per week (13, 14.8%), 6 classes per week (6, 6.8%), and 1 class per week (2, 2.3%). The most popular class types at registration were yoga (69, 78.4%) and fitness (63, 71.6%), followed by meditation (52, 59.1%), tai chi/qigong (50, 56.8%), music therapy (20, 22.7%), and dance (15, 17.0%). On average, patients attended 3 classes per week (range, 1 to 18 classes per week).
Of patients in the EUC group, 81 (80.2%) accessed on-demand resources at least once; on average, patients accessed recordings 3 times per week (range, 1 to 10 times per week).
Effect of IM@Home on fatigue and co-morbid symptoms
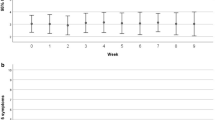
From baseline to week 12, IM@Home participants’ fatigue severity score (primary outcome) decreased by a mean of 1.99 points (95% confidence interval [CI], −2.29 to −1.70) as compared with those of EUC participants, whose scores decreased by a mean of 1.51 points (95% CI, −1.81 to −1.21); between arm difference at week 12 was 0.48 points (95% CI, −0.87 to −0.09) (Table 2). The multivariate Wald test of the arm-by-time interaction coefficients was statistically significant (p = 0.04) favoring the intervention group (Fig. 2).
This figure presents the change in fatigue severity scores, measured by the BFI, for participants in both the IM@Home and EUC groups. The orange dashed line represents IM@Home and the blue solid line represents EUC.
Relative to EUC participants, based on multivariate Wald tests of the arm-by-time interaction coefficients, IM@Home participants also had promising reductions in symptom distress scores (p = 0.003), anxiety (p = 0.03) and depression (p = 0.02). There were no significant differences in insomnia or PROMIS physical and mental health scores between arms over time (see Table 2).
Effect of IM@Home on acute healthcare utilization
While the proportion of patients with ED visits was similar between the study arms (9 [9.1%] IM@Home vs. 12 [11.9%] EUC; p = 0.45), among patients who had an ED visit, the mean number of ED visits per patient was almost half for the IM@Home vs. EUC arm (rate ratio 0.49; 5% CI, 0.23 to 0.95; p = 0.04; Fig. 3a). During the study period, fewer patients in the IM@Home group were hospitalized (4 [4.0%] vs. 13 [12.9%]; p = 0.03), and those inpatients spent fewer days in the hospital (4.25 vs. 10.00 days per patient; rate ratio 0.42; 95% CI, 0.25 to 0.68; p < 0.001; Fig. 3b).
This figure presents acute healthcare utilization for participants receiving the IM@Home intervention vs. EUC over 12 weeks. a shows ED visits for both groups over the study period. b shows unplanned hospitalizations for both groups over the study period. The orange color represents the IM@Home group and the blue color represents the EUC group.
No adverse events were reported as related to the interventions in either group.
Discussion
Patients with cancer experience substantial symptom burden leading to unplanned acute healthcare utilization during active cancer therapy. This is the first randomized trial demonstrating that a digital synchronous intervention package of mind-body and exercise programing, IM@Home, improved fatigue and comorbid symptoms such as anxiety and depression. IM@Home also showed reductions in the number of ED visits and hospitalizations. This demonstrates the potential of a digital medicine intervention integrated into oncological treatment to improve the patient experience by reducing symptom burden and acute healthcare utilization.
Our study contributes to the limited research demonstrating that digital medicine interventions can improve fatigue and co-morbid symptoms. Most digital interventions focus on mental health outcomes only and have shown promise in alleviating anxiety, depression, and improving well-being among people with chronic conditions, including those impacted by cancer11,12. However, their implementation in standard cancer care is scarce11,12. One randomized controlled trial of a web-based tailored intervention for cancer survivors demonstrated its effectiveness in improving social functioning and reducing fatigue and depression in some participants, though results varied30. Current research also emphasizes the importance of tailoring digital health interventions (i.e., content, design) to accommodate participants’ diverse needs, preferences, and functional capabilities11,31,32. Our study is novel as we evaluated a package of several evidence-based interventions including exercise, yoga, tai chi, and meditation rather than studying individual interventions. We aimed to translate prior research findings into a digital care delivery method and facilitate the delivery of guideline-concordant treatment in a convenient and scalable way.
Our study also contributes novel findings of the potential of digital medicine on acute health care utilization. In a scoping review of digital health interventions in cancer care, no RCT was found to focus on evaluating acute care utilization as an outcome33. Mo et al.7 found that high baseline fatigue leads to greater high grade treatment related toxicities among people living with advanced cancer. Here, we demonstrated that engaging patients undergoing active cancer treatment using digitally delivered and evidence-based non-pharmacological symptom interventions has potential to reduce ED visits, hospitalization, and length of hospital stays. If these preliminary data are confirmed, it can lead to substantial cost savings for health systems and society. Our prior qualitative data suggest our intervention could help patients with cancer maintain healthy behaviors, reduce psychological distress, and enhance social connections29. These potential mechanisms should be further investigated to increase our understanding of how best to develop and refine digital interventions that not only improve patient outcomes but also reduce the overall cost of cancer care. Additionally, more research is needed to evaluate whether specific types of interventions (e.g., exercise, yoga) may contribute to better outcomes for specific types of patients (e.g., older patients, those with functional limitations). Furthermore, we need to evaluate whether non-digital components, such as tailored recommendations and follow-up calls, may enhance engagement and adherence to the digital intervention, therefore improving overall outcomes.
Further, our study demonstrates that a PRO-basket design can be an efficient way to investigate digital health interventions for symptom management. Traditional basket trials accelerate drug development for different diseases sharing a common biological therapeutic target34. In symptom science research, targeted outcomes (e.g., fatigue) are often measured by PROs and interventions can apply across cancer types, making the basket design highly appropriate but, to our knowledge, underutilized. The Integrative Medicine for Patient-reported Outcomes, Values and Experience (IMPROVE) PRO-basket trial adapted this master protocol concept, defining the therapeutic target as a specific PRO – fatigue. The PRO-basket design enables the estimation of recruitment rates as well as preliminary effects of the intervention for different disease type in specific care delivery settings (e.g., medical oncology, radiation oncology). These preliminary data will help plan future trials to more definitively test a specific hypothesis supported by preliminary results. This type of design will reduce the regulatory burden, time, and cost of multiple trials, therefore, facilitating the more rapid development of effective digital health interventions for specific targets that can be integrated in care delivery settings to improve outcomes.
Our study has some limitations. The intervention was delivered to patients at a single center, potentially limiting the generalizability of the results. However, the patients were recruited from the Memorial Sloan Kettering Cancer Center (MSK) main campus in Manhattan as well as from MSK Regional Care Network sites in New York State and New Jersey. In addition, the patient population was mainly female, which was driven by the cancer diagnoses included in the study (breast and gynecologic), white, and had higher levels of education. Future research needs to include more diverse populations. Although our recruitment efforts suggest that technology access was not a major barrier, future studies should consider providing loaner devices with internet data plans for those who might otherwise be unable to participate as research expands to under-resourced populations. Our EUC control included evidence-based content and may have decreased the observed benefit of our intervention relative to usual care. Additionally, since our mind-body digital intervention consisted of a selection of evidence-based therapies, it is difficult to determine which were the “active ingredients” leading to the benefit. Assessment of each intervention separately would be helpful in prioritizing which therapies are of most benefit to this patient population. However, the diverse nature of the intervention itself may have enhanced utility and patient engagement with the intervention. Further, the co-morbid symptoms and acute health care outcomes are secondary. We did not adjust for multiple comparison. These findings need to be interpreted with caution and require confirmation in follow up studies. Lastly, the long-term effectiveness of digital mind-body therapy requires further evaluation as patients were followed for 12 weeks.
In conclusion, the recently released National Cancer Plan has set among its goals optimizing care delivery for patients living with cancer and engaging patients with cancer in their care to improve their treatment experience35. Digital IM therapies are one way to accomplish both these goals as they have the potential to improve symptom control and reduce acute care visits through increasing patients’ engagement with their care. Despite promising results, future adequately powered trials are needed to confirm the effect of digital health interventions for improving patient and health system delivery outcomes for people undergoing active cancer treatment.
Methods
Trial design
The IMPROVE trial was a single-center pragmatic, single-blind, two-arm, parallel-group PRO-basket RCT among patients with cancer on active treatment who reported moderate or greater fatigue. The study was approved by the Institutional Review Board at MSK (IRB# 21-369); and registered at clinicaltrials.gov (NCT05053230). Full details of the trial protocol can be found in the Supplementary Information. All participants provided written informed consent.
Patient eligibility
Patients were eligible for the trial if they were: aged 18 years or older; English speaking; diagnosed with breast, thoracic, gynecologic, or head/neck cancers or melanoma; actively receiving systemic or radiation treatments; experiencing moderate or greater fatigue (rated 4 or higher on a 0-10 numerical rating scale36,37) over the past week; had a Karnofsky Performance Scale38 score ≥60; and life expectancy >6 months. Patients were ineligible if they had evidence of cognitive impairment or were participating in concurrent trials of IM interventions.
Randomization and masking
Eligible patients were randomized to the IM@Home intervention or control condition, with a 1:1 allocation ratio through MSK’s Clinical Research Database, a secure computer system ensuring full allocation concealment, and using random permuted block stratified by tumor type. Participants masking was not possible due to the nature of the intervention. The study statisticians and outcome assessment coordinator were masked.
Trial procedures
Patients were on the study for 12 weeks and completed weekly PROs through the MSK Engage platform, a secure tool for collecting and visualizing patient-generated health data that is nested within the MSK Patient Portal4. Patients completed the assessment using devices with Internet access or over the phone with an outcomes assessor (blinded to treatment group). To minimize missing data, the outcomes assessor checked surveys after completion; for patients who missed a study assessment, the outcomes assessor re-sent the survey via Research Electronic Data Capture (REDCap) database or called patients to complete the assessment.
Assessment and outcomes
The primary outcome was the fatigue severity as measured by the Brief Fatigue Inventory (BFI). BFI is a reliable and valid measure of cancer-related fatigue (Cronbach’s α, 0.82 to 0.97)39,40. It contains 3 fatigue severity items and 6 fatigue interference items scored on a Likert scale from 0–10, where 1–3 indicates ‘mild’ fatigue severity/interference; 4–6, ‘moderate;’ and 7–10, ‘severe.’ A composite fatigue severity score can be found by averaging the 9 item scores. The worst fatigue rating in the past week and the fatigue interference subscale scores were used as secondary fatigue outcomes.
Secondary outcomes were symptom distress, insomnia, anxiety, depression, quality of life, and acute healthcare utilization. Symptom distress was measured by the Edmonton Symptom Assessment System (ESAS)41,42, a tool using an 11-point Likert scale (0–10) for symptom intensity, where a ≥1-point change signifies clinically meaningful improvement or deterioration in symptoms, with higher scores indicating greater symptom burden43. Insomnia severity was measured by the Insomnia Severity Index (ISI), a 7-item tool with each rated on a 5-point Likert response scale (e.g., 0=no problem; 4=very severe problem), yielding a total score ranging from 0 to 28 with higher scores representing more severe insomnia symptoms44,45,46. Anxiety and depression were measured by the Hospital Anxiety and Depression Scale (HADS)47, a 14-item tool with 7 items each for depression and anxiety subscales, as scored on a 4-point Likert scale (0–3). Quality of life was measured by the Patient Reported Outcomes Measurement Information System (PROMIS®) Scale - Global Health, a 10-item Likert scale (questions 1–8 and 10 on a 5-point scale (1–5) and question 9 on a 11-point scale (0–10)) instrument yielding two scores, Physical Health and Mental Health, where higher scores indicate better health48,49. We measured acute healthcare utilization by quantifying the frequency of ED visits and unplanned hospital admissions as extracted from MSK’s electronic health record (EHR) and from patients’ self-report, which included visits both within and outside the MSK system. EHR and self-reported visits were further reviewed by study oncologists to confirm accuracy and minimize risk of missing data on visits/admissions.
Over the 12-week intervention, patients completed the BFI and ESAS at baseline and weekly and completed the ISI, HADS, and PROMIS-10 Global Health at baseline and weeks 4, 8, and 12 post-randomization.
Interventions
The IM@Home group received a digital program featuring 23 mind-body and exercise classes delivered synchronously via Zoom29,50. Upon enrollment, patients received detailed information regarding the class schedule and registration instructions. Upon randomization, the member of the research team contacted patients within 1–3 days via phone call to inform them about their group assignment, introduce the IM@Home platform including assessing their technical skills and providing guidance on using Zoom as needed, present the class schedule, assess their needs, and help them choose and register for classes. Immediately after the first call, the class links and on-demand resources were sent through a portal message or e-mail as per patient preference. The following week after registration, a research team member checked in with patients to review their class attendance and reported symptoms and offered tailored class recommendations based on their symptoms. Starting from week 2, the research team member monitored patients’ class attendance and survey completion weekly. Patients who regularly attended classes and submitted weekly surveys were classified as “low touch” and did not require further follow-up unless requested. Those who missed classes and surveys for two consecutive weeks were classified as “medium touch” and received a check-in call. During these calls, questions and concerns were addressed, and additional class recommendations were offered as needed. Patients who neither attended classes nor submitted surveys despite follow-up calls were classified as “high touch.” They received a follow-up phone call to discuss concerns and potentially modify their current class registration. If a patient could not be reached, the research team member would alert the care team and the study PI. Patients could attend as many classes as desired, with options for both movement-based (e.g., fitness, yoga, tai chi) and more mind-focused (e.g., meditation, music therapy) classes. Each class duration was 30 to 60 minutes, with optional participation by audio, video, and group chat. Classes were led by licensed MSK Integrative Medicine Service clinicians (e.g., certified fitness trainers, yoga instructors, mindfulness instructors, and licensed music therapists) with expertise in conducting sessions in the oncology setting.
The Enhanced Usual Care (EUC) group received standard care and access to 17 pre-recorded, on-demand audio or video recordings for meditation, guided imagery, and relaxation. This control condition was chosen to control for patients’ expectation of receiving an integrative medicine intervention, while isolating the active components, such as supervised live classes, tailored recommendations, and community support, in the IM@Home intervention. Upon randomization, a member of the research team contacted EUC patients within 1–3 days via phone call to review study procedures and answer any questions regarding accessing the on-demand resources. Upon study completion, patients received complimentary 3-month access to the IM@Home program.
Statistical analysis
Sample size and power
Our sample size was selected to provide sufficient statistical power to detect a clinically relevant effect size between the IM@Home and EUC arms on our primary outcome measure, the BFI fatigue severity score. Although we used a repeated-measures model to compare the change in BFI scores from baseline to post-baseline between arms, we conservatively estimated our smallest detectable effect size using a two-sample t-test. With a target sample size of 200 patients (100 per arm) we estimated a priori that we would have 80% power to detect a BFI treatment effect size (Cohen’s d) of 0.40 between arms based on prior meta-analysis51 at a given post-baseline follow-up assessment using a two-sided two-sample t-test at a significance threshold of p < 0.05.
Statistical plan
Descriptive statistics were used to summarize patients’ characteristics and reported as frequencies and percentages for categorical variables and as means and standard deviations for continuous variables. For the analysis of the primary outcome (BFI fatigue severity score) and continuous secondary outcomes (ESAS, HADS, ISI, and PROMIS scores), we used constrained linear mixed models (LMMs) in which we constrained the treatment arms to have a common baseline mean52, reflecting the pre-randomization timing of the baseline assessment. Each model included the randomization stratification variable (cancer type), treatment arm, time (categorical, per groupings described below), and the arm-by-time interaction as fixed effects and patient-specific random intercepts. Results are presented as model-estimated means and 95% CIs at each timepoint by arm and by model-estimated mean differences and 95% CIs between arms. The model-estimated mean differences between arms at each follow-up time are also presented as standardized treatment effect sizes (with 95% CIs), similar to Cohen’s d, calculated by dividing the model-estimated mean differences by the pooled baseline standard deviation of the outcome measure53. Inferences regarding differences between arms were based on (1) the multivariate Wald test to evaluate the overall significance of the arm-by-time interaction across all time periods and (2) the individual model coefficients from the arm-by-time interaction to evaluate arm differences at each time period. The LMM analysis method accounts for within-subject correlations from repeated measurements in the same subjects and allows unbiased estimation of between-group differences without necessitating exclusion of participants with missing follow-up data, assuming data are missing-at-random (MAR)54,55. Therefore, all randomized patients with ≥1 patient-reported outcome (PRO) assessment (n = 192) were included in the models, and we pre-specified our linear mixed modeling approach as our preferred method of appropriately addressing missing follow-up data in the analysis. Sensitivity analyses that controlled for additional variables associated with missing follow-up data supported the plausibility of the MAR assumption. Given symptom variability week-to-week and to improve accuracy due to missing weekly assessments, we grouped the weekly BFI and ESAS scores into 4 time periods: baseline/week 0, weeks 1–4, weeks 5–8, and weeks 9–12. All other PROs were measured at weeks 0, 4, 8, and 12 and analyzed as such. We did not adjust the CIs for multiple testing of secondary outcomes.
We modified the original analysis plan for the primary and secondary PRO endpoints from the generalized estimating equations (GEE) models to LMMs because when we plotted the data descriptively the effects between intervention and control separate over time and indicated the effect of IM@Home was cumulative. The LMM analysis allowed for more nuanced evaluation of treatment effects over time while maintaining statistical rigor52,56.
Separate logistic regressions were used to test differences between the study arms in the proportions of patients with ED visits and hospitalizations. Among patients experiencing these events, Poisson regression was used to test differences between the arms in the rates of ED visits and hospitalization days per patient. All analyses were performed in R version 4.2.2.28.
Data availability
Original data will be provided by the corresponding author upon reasonable request.
References
Daly, B. et al. Misery Loves Company: Presenting Symptom Clusters to Urgent Care by Patients Receiving Antineoplastic Therapy. JCO Oncol. Pr. 14, e484–e495 (2018).
Adam, R. et al. Treatment burden in individuals living with and beyond cancer: A systematic review of qualitative literature. PLoS One 18, e0286308 (2023).
Henry, D. H. et al. Symptoms and treatment burden associated with cancer treatment: results from a cross-sectional national survey in the U.S. Support Care Cancer 16, 791–801 (2008).
Daly, B. et al. Analysis of a Remote Monitoring Program for Symptoms Among Adults With Cancer Receiving Antineoplastic Therapy. JAMA Netw. Open 5, e221078 (2022).
Alishahi Tabriz, A. et al. Trends and Characteristics of Potentially Preventable Emergency Department Visits Among Patients With Cancer in the US. JAMA Netw. Open 6, e2250423 (2023).
Carnio, S., Di Stefano, R. F. & Novello, S. Fatigue in lung cancer patients: symptom burden and management of challenges. Lung Cancer (Auckl.) 7, 73–82 (2016).
Mo, J. et al. Association of Fatigue and Outcomes in Advanced Cancer: An Analysis of Four SWOG Treatment Trials. JCO Oncol. Pr. 17, e1246–e1257 (2021).
The White House. Fact sheet: President Biden reignites cancer moonshot to end cancer as we know it, https://www.whitehouse.gov/briefing-room/statements-releases/2022/02/02/fact-sheet-president-biden-reignites-cancer-moonshot-to-end-cancer-as-we-know-it/ (2022).
National Plan Unveiled to Combat Cancer. Cancer Dis. 13, OF1. (2023)
Zon, R. T. et al. Telehealth in Oncology: ASCO Standards and Practice Recommendations. JCO Oncol. Pr. 17, 546–564 (2021).
Shah, A. et al. Digital Health Interventions for Depression and Anxiety Among People With Chronic Conditions: Scoping Review. J. Med. Internet Res. 24, e38030 (2022).
Elkefi, S., Trapani, D. & Ryan, S. The role of digital health in supporting cancer patients’ mental health and psychological well-being for a better quality of life: A systematic literature review. Int J. Med. Inf. 176, 105065 (2023).
Deleemans, J. M. et al. Recent Progress in Mind–Body Therapies in Cancer Care. Curr. Oncol. Rep. 25, 293–307 (2023).
Duan, L., Xu, Y. & Li, M. Effects of Mind-Body Exercise in Cancer Survivors: A Systematic Review and Meta-Analysis. Evid. Based Complement Altern. Med. 2020, 7607161 (2020).
Bower, J. E. et al. Management of Fatigue in Adult Survivors of Cancer: ASCO–Society for Integrative Oncology Guideline Update. J. Clin. Oncol. 42, 2456–2487 (2024).
Wayne, P. M. et al. Tai Chi and Qigong for cancer-related symptoms and quality of life: a systematic review and meta-analysis. J. Cancer Surviv. 12, 256–267 (2018).
Carlson, L. E. et al. Mind-Body Therapies in Cancer: What Is the Latest Evidence? Curr. Oncol. Rep. 19, 67 (2017).
Bower, J. E. et al. Screening, assessment, and management of fatigue in adult survivors of cancer: an American Society of Clinical oncology clinical practice guideline adaptation. J. Clin. Oncol. 32, 1840–1850 (2014).
Mao, J. J. et al. Integrative Medicine for Pain Management in Oncology: Society for Integrative Oncology-ASCO Guideline. J. Clin. Oncol. 40, 3998–4024 (2022).
Carlson, L. E. et al. Integrative Oncology Care of Symptoms of Anxiety and Depression in Adults With Cancer: Society for Integrative Oncology–ASCO Guideline. J. Clin. Oncol. 41, 4562–4591 (2023).
Bauml, J. M. et al. Do attitudes and beliefs regarding complementary and alternative medicine impact its use among patients with cancer? A cross-sectional survey. Cancer 121, 2431–2438 (2015).
Desai, K., Liou, K., Liang, K., Seluzicki, C. & Mao, J. J. Availability of Integrative Medicine therapies at National Cancer Institute Designated Comprehensive Cancer Centers and Community Hospitals. J. Alter. Complement Med. 27, 1011–1101 (2021).
Azizoddin, D. R. et al. Development and pre-pilot testing of STAMP + CBT: an mHealth app combining pain cognitive behavioral therapy and opioid support for patients with advanced cancer and pain. Support Care Cancer 32, 123 (2024).
Davies, F. et al. Uptake of an online psychological therapy program (iCanADAPT-Early) when implemented within a clinical pathway in cancer care centres. Psychooncology 31, 338–341 (2022).
Murphy, M. J. et al. Randomised controlled trial of internet-delivered cognitive behaviour therapy for clinical depression and/or anxiety in cancer survivors (iCanADAPT Early). Psychooncology 29, 76–85 (2020).
Romero, S. A. D. et al. Barriers to physical activity: a study of academic and community cancer survivors with pain. J. Cancer Surviv. 12, 744–752 (2018).
Desai, K. et al. Understanding interest, barriers, and preferences related to yoga practice among cancer survivors. Support Care Cancer 29, 5313–5321 (2021).
Romero, S. A. D. et al. The association between fatigue and pain symptoms and decreased physical activity after cancer. Support Care Cancer 26, 3423–3430 (2018).
Emard, N. et al. Virtual Mind-Body Programming for Patients With Cancer During the COVID-19 Pandemic: Qualitative Study. JMIR Cancer 7, e27384 (2021).
Willems, R. A., Mesters, I., Lechner, L., Kanera, I. M. & Bolman, C. A. W. Long-term effectiveness and moderators of a web-based tailored intervention for cancer survivors on social and emotional functioning, depression, and fatigue: randomized controlled trial. J. Cancer Surviv. 11, 691–703 (2017).
Lee, K. et al. Digital Health Interventions for Adult Patients With Cancer Evaluated in Randomized Controlled Trials: Scoping Review. J. Med Internet Res. 25, e38333 (2023).
Aapro, M. et al. Digital health for optimal supportive care in oncology: benefits, limits, and future perspectives. Support Care Cancer 28, 4589–4612 (2020).
Shaffer, K. M. et al. Digital health and telehealth in cancer care: a scoping review of reviews. Lancet Digit Health 5, e316–e327 (2023).
Woodcock, J. & LaVange, L. M. Master Protocols to Study Multiple Therapies, Multiple Diseases, or Both. N. Engl. J. Med. 377, 62–70 (2017).
U.S. Department of Health & Human Services & National Cancer Institute. (National Cancer Institute, 2023).
Temel, J. S., Pirl, W. F., Recklitis, C. J., Cashavelly, B. & Lynch, T. J. Feasibility and Validity of a One-Item Fatigue Screen in a Thoracic Oncology Clinic. J. Thorac. Oncol. 1, 454–459 (2006).
Butt, Z. et al. Use of a single-item screening tool to detect clinically significant fatigue, pain, distress, and anorexia in ambulatory cancer practice. J. Pain. Symptom Manag. 35, 20–30 (2008).
Schag, C. C., Heinrich, R. L. & Ganz, P. A. Karnofsky performance status revisited: reliability, validity, and guidelines. J. Clin. Oncol. 2, 187–193 (1984).
Mendoza, T. R. et al. The rapid assessment of fatigue severity in cancer patients: use of the brief fatigue inventory (English). Cancer 85, 1186–1196 (1999).
Mendoza, T. R. et al. Assessment of fatigue in cancer patients and community dwellers: validation study of the Filipino version of the brief fatigue inventory. Oncology 79, 112–117 (2010).
Bruera, E., Kuehn, N., Miller, M. J., Selmser, P. & Macmillan, K. The Edmonton Symptom Assessment System (ESAS): a simple method for the assessment of palliative care patients. J. Palliat. Care 7, 6–9 (1991).
Hui, D. et al. Minimal clinically important differences in the Edmonton Symptom Assessment Scale in cancer patients: A prospective, multicenter study. Cancer 121, 3027–3035 (2015).
Zimmermann, C. et al. Nurse and physician inter-rater agreement of three performance status measures in palliative care outpatients. Support Care Cancer 18, 609–616 (2010).
Bastien, C. H., Vallieres, A. & Morin, C. M. Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep. Med. 2, 297–307 (2001).
Morin, C. M., Belleville, G., Belanger, L. & Ivers, H. The Insomnia Severity Index: psychometric indicators to detect insomnia cases and evaluate treatment response. Sleep 34, 601–608 (2011).
Savard, M. H., Savard, J., Simard, S. & Ivers, H. Empirical validation of the Insomnia Severity Index in cancer patients. Psychooncology 14, 429–441 (2005).
Zigmond, A. S. & Snaith, R. P. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 67, 361–370 (1983).
Hays, R. D., Bjorner, J. B., Revicki, D. A., Spritzer, K. L. & Cella, D. Development of physical and mental health summary scores from the patient-reported outcomes measurement information system (PROMIS) global items. Qual. Life Res. 18, 873–880 (2009).
Revicki, D. A. et al. Predicting EuroQol (EQ- 5D) scores from the patient-reported outcomes measurement information system (PROMIS) global items and domain item banks in a United States sample. Qual. Life Res. 18, 783–791 (2009).
Trevino, K. M. et al. Rapid deployment of virtual mind-body interventions during the COVID-19 outbreak: feasibility, acceptability, and implications for future care. Support Care Cancer 29, 543–546 (2021).
Tomlinson, D., Diorio, C., Beyene, J. & Sung, L. Effect of Exercise on Cancer-Related Fatigue: A Meta-analysis. Am. J. Phys. Med Rehabil. 93, 675–686 (2014).
Liu, G. F., Lu, K., Mogg, R., Mallick, M. & Mehrotra, D. V. Should baseline be a covariate or dependent variable in analyses of change from baseline in clinical trials? Stat. Med. 28, 2509–2530 (2009).
Feingold, A. A Regression Framework for Effect Size Assessments in Longitudinal Modeling of Group Differences. Rev. Gen. Psychol. 17, 111–121 (2013).
Mallinckrod, C. H., Lane, P. W., Schnell, D., Peng, Y. & Mancuso, J. P. Recommendations for the Primary Analysis of Continuous Endpoints in Longitudinal Clinical Trials. Drug Inf. J. 42, 303–319 (2008).
Laird, N. M. & Ware, J. H. Random-effects models for longitudinal data. Biometrics 38, 963–974 (1982).
Diggle, P., Heagerty, P., Liang, K.-Y. & Zeger, S. Analysis of Longitudinal Data. (Oxford University Press, Oxford, 2002).
Acknowledgements
This work was supported in part by the JDJ Foundation, AKTIV Against Cancer, a National Institutes of Health (NIH) / National Cancer Institute (NCI) Cancer Center Support Grant (P30 CA008748), and the Translational and Integrative Medicine Research Fund at Memorial Sloan Kettering Cancer Center. Dr. Daly is supported by a grant from the Emerson Collective and an NCI 1P50CA271357-01. Dr. Gillespie is supported by an NCI Career Development Award No. K08 CA252640.
Author information
Authors and Affiliations
Contributions
J.J.M.: conceptualization, methodology, investigation, data analysis & interpretation, writing – original draft, writing – reviewing & editing, supervision, funding acquisition. K.B.: investigation, trial management, writing – original draft, writing – reviewing & editing. E.G.: conceptualization, methodology, investigation, writing – reviewing & editing. A.G.: conceptualization, methodology, investigation, writing – reviewing & editing. T(KW)H: conceptualization, methodology, investigation, writing – reviewing & editing. R.B.: methodology, formal analysis, writing – reviewing & editing. K.P.: conceptualization, methodology, writing – reviewing & editing. M.P.: conceptualization, methodology, investigation, writing – reviewing & editing. B.D.: conceptualization, methodology, investigation, writing – original draft, writing – reviewing & editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
J.J.M.: Research Funding: Tibet Cheezheng Tibetan Medicine Co. Ltd., Zhongke Health International LLC. K.B.: None meeting criteria for this time period. EG: Other Relationship: eContour. A.G.: Consulting or Advisory Role: Merck; Research Funding: Lilly, Mereo BioPharma. KW(T)H: Stock and Other Ownership Interests: TeamX Medical Group; Patents, Royalties, Other Intellectual Property: LookUpTrials (patent pending). R.B.: None meeting criteria for this time period. K.P.: Stock and Other Ownership Interests: 23andMe, Adicet Bio, Chinook Therapeutics, Codexis, T2 Biosystems, Vincerx Pharma; Research Funding: American Association for Cancer Research (AACR). M.A.P.: Consulting or Advisory Role: Bristol-Myers Squibb, Cancer Expert Now, Chugai Pharma, Eisai, Erasca Inc, Intellisphere, Merck, MJH Associates, Nektar, Novartis, Pfizer, WebMD; Research Funding: Array BioPharma, Bristol-Myers Squibb, Infinity Pharmaceuticals, Merck, Novartis, Rgenix. B.D.: Stock and Other Ownership Interests: CVS Health, Lilly, Roche/Genentech; Consulting or Advisory Role: Varian Medical Systems and I-MAB Biopharma.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mao, J.J., Bryl, K., Gillespie, E.F. et al. Randomized clinical trial of a digital integrative medicine intervention among patients undergoing active cancer treatment. npj Digit. Med. 8, 29 (2025). https://doi.org/10.1038/s41746-024-01387-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41746-024-01387-z