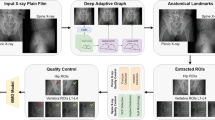
Abstract
Early identification of abnormal bone mineral density (BMD) through opportunistic screening is critical for preventing osteoporotic fractures. We validated an AI model in 2384 asymptomatic adults (57.7% female; mean age 43.6 years) undergoing health examinations in Taiwan. Using DXA as the reference, the model identified 255 suspected abnormal BMD cases, with 94 (3.9%) DXA-confirmed positive. Population-level performance was robust, yielding an AUC of 0.95 (95% CI 0.93–0.99) and sensitivity of 79.7% (95% CI 71.3–86.5%). Although BMI distributions paralleled East Asian regional trends, intersectional subgroup analyses remain exploratory due to small event counts. Decision curve analysis indicated superior net benefit for AI-based referral over “refer all” or “refer none” strategies, particularly for women with normal BMI (18.5–23 kg/m²). This AI tool offers precise triage for Asian health examination populations, though further validation in multi-center cohorts is required to confirm broad generalizability.
Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are de-identified, but due to patient data confidentiality and Institutional Review Board requirements, they are not publicly available. They can be obtained from the corresponding author upon reasonable request.
Code availability
The custom code used for this study is proprietary to Acer Medical Inc. and cannot be made publicly available due to company confidentiality and intellectual property restrictions. The code was implemented using Python (version 3.12) and C# (version 7.3). Specific variables and parameters used for model training and statistical analysis are described in the “Methods” section and Supplementary Information, providing sufficient information for independent reproduction of the results.
References
World Health Organization (WHO). Prevention and Management of Osteoporosis: Report of a WHO Scientific Group (World Health Organization, Geneva, 2003).
Kanis, J. A. et al. The diagnosis of osteoporosis. J. Bone Miner. Res. 9, 1137–1141 (1994).
Ferizi, U., Honig, S. & Chang, G. Artificial intelligence, osteoporosis and fragility fractures. Curr. Opin. Rheumatol. 31, 368–375 (2019).
Dimai, H. P. New horizons: artificial intelligence tools for managing osteoporosis. J. Clin. Endocrinol. Metab. 108, 775–783 (2023).
Gatineau, G. et al. Development and reporting of artificial intelligence in osteoporosis management. J. Bone Miner. Res. 39, 1553–1573 (2024).
Amani, F. et al. Diagnostic accuracy of deep learning in prediction of osteoporosis: a systematic review and meta-analysis. BMC Musculoskelet. Disord. 25, 991 (2024).
He, Y., Lin, J., Zhu, S., Zhu, J. & Xu, Z. Deep learning in the radiologic diagnosis of osteoporosis: a literature review. J. Int. Med. Res. 52, 3000605241244754 (2024).
Paderno, A. et al. Artificial intelligence-enhanced opportunistic screening of osteoporosis in CT scan: a scoping review. Osteoporos. Int. 35, 1681–1692 (2024).
Yu, X., Ye, C. & Xiang, L. Application of artificial neural network in the diagnostic system of osteoporosis. Neurocomputing 214, 376–381 (2016).
Iliou, T., Anagnostopoulos, C. N., Stephanakis, I. M. & Anastassopoulos, G. A novel data preprocessing method for boosting neural network performance: a case study in osteoporosis prediction. Inf. Sci. 380, 92–100 (2017).
Nam, K. H. et al. Machine learning model to predict osteoporotic spine on CT. J. Korean Neurosurg. Soc. 62, 442–449 (2019).
Pickhardt, P. J. et al. Opportunistic screening for osteoporosis using abdominal computed tomography scans obtained for other indications. Ann. Intern. Med. 158, 588–595 (2013).
Pickhardt, P. J. et al. Simultaneous screening for osteoporosis at CT colonography: bone mineral density assessment using MDCT attenuation techniques compared with the DXA reference standard. J. Bone Miner. Res. 26, 2194–2203 (2011).
Cosman, F. et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos. Int. 25, 2359–2381 (2014).
International Society for Clinical Densitometry (ISCD). 2023 ISCD Official Positions - Adult. ISCD.org. https://iscd.org/learn/official-positions/adult-positions/ (2023).
US Preventive Services Task Force (USPSTF). Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. https://doi.org/10.1001/jama.2024.27154 (2025).
Yang, J. et al. Criteria for osteoporosis diagnosis: a systematic review and meta-analysis of osteoporosis diagnostic studies with DXA and QCT. eClinicalMedicine 83, 103244 (2025).
Ong, W. et al. Artificial intelligence applications for osteoporosis classification using computed tomography. Bioengineering 10, 1364 (2023).
Hong, N. et al. Deep-learning-based detection of vertebral fracture and osteoporosis using lateral spine X-ray radiography. J. Bone Miner. Res. 38, 887–895 (2023).
Lin, C. et al. Osteoporotic precise screening using chest radiography and artificial neural network: the OPSCAN randomized controlled trial. Radiology 311, e231937 (2024).
Wang, F. et al. Lumbar bone mineral density estimation from chest X-ray images: anatomy-aware attentive multi-ROI modeling. IEEE Trans. Med. Imaging 42, 257–267 (2023).
Ou Yang, W. Y. et al. Development of machine learning models for prediction of osteoporosis from clinical health examination data. Int. J. Environ. Res. Public Health 18, 7635 (2021).
Shim, J. G., Kim, D. W. & Ryu, K. H. Application of machine learning approaches for osteoporosis risk prediction in postmenopausal women. Arch. Osteoporos. 15, 169 (2020).
Bui, H. M. et al. Predicting the risk of osteoporosis in older Vietnamese women using machine learning approaches. Sci. Rep. 12, 20160 (2022).
Tu, J. et al. Using machine learning techniques to predict the risk of osteoporosis based on nationwide chronic disease data. Sci. Rep. 14, 5245 (2024).
Liu, L. et al. A hierarchical opportunistic screening model for osteoporosis using machine learning applied to clinical data and CT images. BMC Bioinform. 23, 63 (2022).
Tseng, S. C. et al. Clinical validation of a deep learning-based software for lumbar bone mineral density and T-score prediction from chest X-rays. Diagnostics 14, 1208 (2024).
World Health Organization. The Asia-Pacific Perspective: Redefining Obesity and Its Treatment (Health Communications Australia, 2000).
Wright, N. C. et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J. Bone Miner. Res. 29, 2520–2526 (2014).
Bliuc, D. et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA 301, 513–521 (2009).
Chandran, M. et al. Prevalence of osteoporosis and incidence of related fractures in developed economies in the Asia Pacific region: a systematic review. Osteoporosis. Int. 34, 1037–1053 (2023).
Liu, Y. et al. The effect of overweight or obesity on osteoporosis: a systematic review and meta-analysis. Clin. Nutr. 42, 2457–2467 (2023).
Taiwan Health Promotion Administration. National Nutrition and Health Survey in Taiwan 2017–2020 (NAHSIT). Health Promotion Administration, Ministry of Health and Welfare, Taipei, Taiwan (2021).
Ministry of Health, Labour and Welfare. National Health and Nutrition Survey 2022. Ministry of Health, Labour and Welfare, Tokyo, Japan (2022).
Korea Disease Control and Prevention Agency. Korea National Health and Nutrition Examination Survey (KNHANES) 2019–2021. Korea Disease Control and Prevention Agency, Cheongju, Korea (2022).
Biro, J. M. et al. Opportunities and risks of artificial intelligence in patient portal messaging in primary care. npj Digit. Med. 8, 222 (2025).
Chandran, M. et al. Development of the Asia Pacific Consortium on Osteoporosis (APCO) Framework: clinical standards of care for the screening, diagnosis, and management of osteoporosis in the Asia-Pacific region. Osteoporos. Int. 32, 1249–1275 (2021).
International Osteoporosis Foundation (IOF). The global call to action to close the osteoporosis care gap. https://www.osteoporosis.foundation. Accessed Oct 2025.
World Health Organization (WHO). Global Strategy on Digital Health 2020-2025. World Health Organization, Geneva. https://www.who.int/docs/default-source/documents/global-strategy-on-digital-health-2020-2025.pdf. Accessed Oct 2025.
Vickers, A. J. & Elkin, E. B. Decision curve analysis: a novel method for evaluating prediction models. Med. Decis. Making 26, 565–574 (2006).
Acknowledgements
This research was funded by St. Paul’s Hospital (grant number SPMRP-U1-8002), awarded to S.-H.C., and partially supported by Acer Medical Inc., with funding awarded to K.-H.C. to support the technical development and external validation of the AI system. We acknowledge Acer Medical Inc. for technical assistance during model deployment, and Taichung Veterans General Hospital for providing the internal dataset used to pre-train the AI model prior to external validation. The funders had no role in the study design, data collection, analysis, interpretation, manuscript preparation, or the decision to publish.
Author information
Authors and Affiliations
Contributions
S.-H.C. and R.-E.C. conceptualized and designed the study, led the clinical data analysis, and drafted the manuscript. S.-H.C. additionally contributed to data collection, analysis, and interpretation. C.-E.L. provided technical supervision and contributed to model conceptualization. D.-J.Y. was responsible for data curation, software development, and model validation. M.-L.W. and P.Y. coordinated data extraction and de-identification. K.-H.C. oversaw technical development and supervised external validation. All authors reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
K.-H.C. received research funding from Acer Medical Inc. to support the technical development of the AI model published in the previous work (Reference 23). C.-E.L. is an employee of Acer Medical Inc. D.-J.Y. is an employee of Acer Inc. All other authors declare no competing interests. The study was independently designed and conducted by the clinical team. Commercial collaborators had no role in the study design, data collection, analysis, interpretation, or manuscript preparation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, SH., Chang, RE., Lien, CE. et al. Advancing diagnostic equity through artificial intelligence chest radiograph screening for osteoporosis in Asian populations. npj Digit. Med. (2026). https://doi.org/10.1038/s41746-026-02484-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-026-02484-x