Abstract
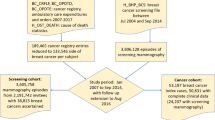
Thirty percent of interval breast cancers, diagnosed between routine screening mammograms, have a poorer prognosis than screen-detected cancers. Deep learning algorithms can estimate short-term risk from negative mammograms to guide supplemental imaging or screening intervals, but comparative validation on complete national screening data is lacking. We retrospectively evaluated four risk algorithms (Mirai, iCAD, Transpara, and Google) using 112,621 negative mammograms from two UK NHS Breast Screening Programme sites with different mammography systems (Philips, GE) over one screening round (2014–2017) with five-year follow-up, including 1225 future cancers. There was a distinct ranking in discriminative ability; overall AUCs ranged 0.65–0.72, only one algorithm significantly differed between systems. For interval cancers, AUCs ranged 0.67–0.77. Within the highest 4.0% of risk scores, top algorithms identified ~20% of future cancers, including ~27% of interval cancers, doubling at the 14.0% threshold. These differences highlight the need for multi-algorithm prospective trials and potential fine-tuning to improve generalisation across unseen systems.
Similar content being viewed by others
Data availability
Study data from the UK NHS Breast Screening Programme were pseudonymised. Access is restricted, and the data cannot be publicly shared. However, researchers may request access through the relevant NHS data governance and research ethics procedures, subject to review and appropriate data sharing agreements. Requests should be made to the corresponding author and will be considered on a case-by-case basis.
Code availability
The deep learning algorithms evaluated in this study were provided for research purposes by their respective commercial companies and cannot be shared by the authors. Researchers wishing to access these algorithms should contact the respective companies directly. Mirai has been made available by Yala et al. on GitHub: https://github.com/yala/Mirai. Analysis code for this study has been made available on GitHub: https://github.com/RandomForestJosh/breast_cancer_risk_prediction.
References
Marmot, M. G. et al. The benefits and harms of breast cancer screening: an independent review. Br. J. Cancer 108, 2205–2240 (2013).
Schumann, L., Hadwiger, M., Eisemann, N. & Katalinic, A. Lead-time corrected effect on breast cancer survival in germany by mode of detection. Cancers 16, 1326 (2024).
Dibden, A. et al. Reduction in interval cancer rates following the introduction of two-view mammography in the UK breast screening programme. Br. J. Cancer 110, 560–564 (2014).
Henderson, L. M. et al. Breast Cancer Characteristics Associated With Digital Versus Film-Screen Mammography for Screen-Detected and Interval Cancers. Am. J. Roentgenol. 205, 676–684 (2015).
Kerlikowske, K. et al. Advanced Breast Cancer Definitions by Staging System Examined in the Breast Cancer Surveillance Consortium. JNCI J. Natl. Cancer Inst. 113, 909–916 (2021).
Törnberg, S. et al. A pooled analysis of interval cancer rates in six European countries. Eur. J. Cancer Prev. 19, 87–93 (2010).
Bennett, R. L., Sellars, S. J. & Moss, S. M. Interval cancers in the NHS breast cancer screening programme in England, Wales and Northern Ireland. Br. J. Cancer 104, 571–577 (2011).
Yala, A. et al. Optimizing risk-based breast cancer screening policies with reinforcement learning. Nat. Med. 28, 136–143 (2022).
Kerlikowske, K. et al. Cumulative Advanced Breast Cancer Risk Prediction Model Developed in a Screening Mammography Population. JNCI J. Natl. Cancer Inst. 114, 676–685 (2022).
Van Winkel, S. L. et al. Using AI to Select Women with Intermediate Breast Cancer Risk for Breast Screening with MRI. Radiology 314, e233067 (2025).
Gilbert, F. J. et al. Comparison of supplemental breast cancer imaging techniques—interim results from the BRAID randomised controlled trial. Lancet 405, 1935–1944 (2025).
Lang, K., Hofvind, S., Rodriguez-Ruiz, A. & Andersson, I. Can artificial intelligence reduce the interval cancer rate in mammography screening? Eur. Radiol. 31, 5940–5947 (2021).
Eriksson, M. et al. European validation of an image-derived AI-based short-term risk model for individualized breast cancer screening—a nested case-control study. Lancet Reg. Health - Eur. 37, 100798 (2024).
Yala, A. et al. Multi-Institutional Validation of a Mammography-Based Breast Cancer Risk Model. J. Clin. Oncol. 40, 1732–1740 (2022).
Rothwell, J. W. D. et al. Evaluation of a Mammography-based Deep Learning Model for Breast Cancer Risk Prediction in a Triennial Screening Program. Radiology 317, e250391 (2025).
Hussain, S. et al. Breast cancer risk prediction using machine learning: a systematic review. Front. Oncol. 14, 1343627 (2024).
Lehman, C. et al. Deep Learning vs Traditional Breast Cancer Risk Models to Support Risk-Based Mammography Screening. J. Natl. Cancer Inst. https://doi.org/10.1093/jnci/djac142 (2022).
Hill, H., Roadevin, C., Duffy, S., Mandrik, O. & Brentnall, A. Cost-Effectiveness of AI for Risk-Stratified Breast Cancer Screening. JAMA Netw. Open 7, e2431715 (2024).
Isautier, J. M. J. et al. Clinical guidelines for the management of mammographic density: a systematic review of breast screening guidelines worldwide. JNCI Cancer Spectr. 8, pkae103 (2024).
Bakker, M. F. et al. Supplemental MRI Screening for Women with Extremely Dense Breast Tissue. N. Engl. J. Med. 381, 2091–2102 (2019).
Salim, M. et al. AI-based selection of individuals for supplemental MRI in population-based breast cancer screening: the randomized ScreenTrustMRI trial. Nat. Med. https://doi.org/10.1038/s41591-024-03093-5 (2024).
Halligan, S., Altman, D. G. & Mallett, S. Disadvantages of using the area under the receiver operating characteristic curve to assess imaging tests: A discussion and proposal for an alternative approach. Eur. Radiol. 25, 932–939 (2015).
Gastounioti, A. et al. External Validation of a Mammography-Derived AI-Based Risk Model in a U.S. Breast Cancer Screening Cohort of White and Black Women. Cancers 14, 4803 (2022).
Lauritzen, A. D. et al. Assessing Breast Cancer Risk by Combining AI for Lesion Detection and Mammographic Texture. Radiology 308, e230227 (2023).
Kerlikowske, K. et al. Identifying women with dense breasts at high risk for interval cancer: a cohort study. Ann. Intern. Med. 162, 673–681 (2015).
Arasu, V. A. et al. Comparison of Mammography AI Algorithms with a Clinical Risk Model for 5-year Breast Cancer Risk Prediction: An Observational Study. Radiology 307, e222733 (2023).
NHS England. Protocols for the surveillance of women at higher risk of developing breast cancer. GOV.UK https://www.gov.uk/government/publications/breast-screening-higher-risk-women-surveillance-protocols/protocols-for-surveillance-of-women-at-higher-risk-of-developing-breast-cancer (2025).
iCAD, Inc. ProFound AI Breast Cancer Health Suite. https://www.icadmed.com/breast-health/.
Choosing Transpara. ScreenPoint Medical BV https://screenpoint-medical.com/choosing-transpara/.
Yala, A. et al. Toward robust mammography-based models for breast cancer risk. Sci. Transl. Med. 13, eaba4373 (2021).
Yala, A. yala/OncoServe_Public. (2025).
McKinney, S. M. et al. International evaluation of an AI system for breast cancer screening. Nature 577, (2020).
NHS England. NHS Breast screening programme screening standards valid for data collected from 1 April 2021. GOV.UK https://www.gov.uk/government/publications/breast-screening-consolidated-programme-standards/nhs-breast-screening-programme-screening-standards-valid-for-data-collected-from-1-april-2021 (2024).
Grabler, P., Sighoko, D., Wang, L., Allgood, K. & Ansell, D. Recall and Cancer Detection Rates for Screening Mammography: Finding the Sweet Spot. Am. J. Roentgenol. 208, 208–213 (2017).
Acknowledgements
We would like to thank all the women who contributed their deidentified data to CC-MEDIA for research purposes, and consequently, this research. We would also like to thank the associated commercial companies for providing research access to their risk prediction algorithms, and Yala et al. for making the Mirai code base freely available under the MIT license. This research was supported by the Future Dreams Breast Cancer Charity, the National Institute for Health and Care Research (NIHR) Cambridge Biomedical Research Centre (NIHR203312*), and the Cancer Research UK early detection program grant (C543/A26884). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
Author information
Authors and Affiliations
Contributions
J.R., N.R.P., F.KT., and F.J.G. conceptualized the study. J.R., N.R.P., F.KT., S.E.H., B.K., and A.J. collected and curated screening data. J.R., N.R.P., J.D.K., R.T.B., and S.E.H. coordinated the technical setup of algorithms and the research environment in which they processed data. J.R. conducted failure analyses. J.R., N.R.P., and Y.H. coordinated and conducted analyses. Y.H. provided statistical guidance and supervised the implementation of analyses. J.R. assembled all figures. J.R. wrote the manuscript. All authors reviewed and contributed to manuscript editing.
Corresponding author
Ethics declarations
Competing interests
J.R., N.R.P., S.E.H. and F.J.G. research agreements with Merantix, ScreenPoint Medical, Lunit, iCAD, Google, Therapixel and Volpara. S.E.H. Radiology Artificial Intelligence Trainee Editorial Board member. J.R. and F.KT. were supported by a Future Dreams breast cancer charity grant awarded to F.J.G. F.KT. consulting for Genesis Care. J.D.K. research support from the NIHR Cambridge Biomedical Research Centre and the Wellcome Trust; grants from Cancer Research UK, AstraZeneca, and GE HealthCare. F.J.G., recipient of the Cancer Research UK Early Detection Programme grant; consulting for Alphabet and Kheiron; honoraria for lectures from GE HealthCare; participation on an advisory board for Bayer; past president (2020–2022) of the European Society of Breast Imaging; current Clinical Radiology AI Lead Advisor at the Royal College of Radiologists; contrast media for unrelated trial from Bayer. Y.H., R.T.B., B.K. and A.J. have no competing interests to declare.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rothwell, J., Payne, N., Kilburn-Toppin, F. et al. Performance of breast cancer risk prediction algorithms across mammography systems in the UK screening programme. npj Digit. Med. (2026). https://doi.org/10.1038/s41746-026-02507-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-026-02507-7