Abstract
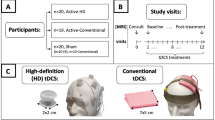
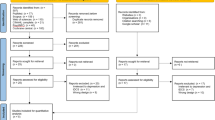
High-definition transcranial alternating current stimulation (HD-tACS) is an emerging neuromodulatory intervention for major depressive disorder (MDD). Here, we evaluate the efficacy and safety of a portable, low-intensity (1.6 mA), 10 Hz HD-tACS protocol targeting the bilateral dorsolateral prefrontal cortex (DLPFC) in adults with MDD. This randomized clinical trial was conducted including 120 adult MDD participants. Participants underwent 20 sessions (5 sessions/week for 4 weeks) of either active 10 Hz HD-tACS (1.6 mA) or sham stimulation a ratio of 1:1 and were followed for an additional 4 weeks. The primary outcome was the percentage reduction in 17-item Hamilton Depression Rating Scale scores at week 4. The active group showed significantly greater improvement in depression than the sham group (48% ± 14% vs 24% ± 13%, p < 0.001), sustained through the 4-week follow-up period. Additionally, significant improvements in the active group were also found in anxiety, sleep efficiency, quality of life components of physical, vitality, social functioning, and mental health. Adverse events including two cases of new-onset hypomania occurred, but no serious adverse events were observed. Thus, this trial supported the efficacy and good tolerability of 10 Hz HD-tACS for patients with MDD. Further research requires more focus on the underlying neural mechanisms and the optimized stimulation protocols. Trial Registration: China Clinical Trial Registry Identifier: ChiCTR2500103570. ClinicalTrials.gov Identifier: NCT06891326.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author Bomin Sun and Xin Lv. Raw data are protected and are not publicly available due to data privacy laws. Access to the data is restricted to psychiatry or neurology specialists, with a response timeframe of one month and a validity period of six months. The datasets will be distributed by email upon reasonable request.
Code availability
Statistical analysis in this study was performed using SPSS (version 27.0, IBM Corp.) and R (version 4.3.0, R Foundation for Statistical Computing). SPSS is a commercial software, while R is an open-source platform publicly available from its official website (https://www.r-project.org/). The specific R scripts, data analysis pipelines, and custom functions used to generate the reported results are available from the corresponding author Bomin Sun and Xin Lv for non-commercial, academic research purposes.
References
World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates. Geneva, Switzerland: WHO Document Production Services; 2017.
Rush, A. J. et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am. J. Psychiatry 163, 1905–1917 (2006).
Kovich, H., Kim, W. & Quaste, A. M. Pharmacologic treatment of depression. Am. Fam. Phys. 107, 173–181 (2023).
Qi, H. et al. Treatment rate for major depressive disorder in China: a meta-analysis of epidemiological studies. Psychiatr. Q 90, 883–895 (2019).
Hollon, S. D. et al. Presenting characteristics of depressed outpatients as a function of recurrence: preliminary findings from the STAR*D clinical trial. J. Psychiatr. Re.s 40, 59–69 (2006).
Baig-Ward, K. M., Jha, M. K. & Trivedi, M. H. The individual and societal burden of treatment-resistant depression: an overview. Psychiatr. Clin. North Am. 46, 211–226 (2023).
Woodham, R. D. et al. Home-based transcranial direct current stimulation treatment for major depressive disorder: a fully remote phase 2 randomized sham-controlled trial. Nat. Med. 31, 87–95 (2025).
Brunoni, A. R. et al. Trial of electrical direct-current therapy versus escitalopram for depression. N. Engl. J. Med. 376, 2523–2533 (2017).
Fregni, F. et al. Evidence-based guidelines and secondary meta-analysis for the use of transcranial direct current stimulation in neurological and psychiatric disorders. Int. J. Neuropsychopharmacol. 24, 256–313 (2021).
McGirr, A. & Berlim, M. T. Clinical usefulness of therapeutic neuromodulation for major depression: a systematic meta-review of recent meta-analyses. Psychiatr. Clin. North Am. 41, 485–503 (2018).
Razza, L. B. et al. A systematic review and meta-analysis on the effects of transcranial direct current stimulation in depressive episodes. Depress Anxiety 37, 594–608 (2020).
Elyamany, O., Leicht, G., Herrmann, C. S. & Mulert, C. Transcranial alternating current stimulation (tACS): from basic mechanisms towards first applications in psychiatry. Eur. Arch. Psychiatry Clin. Neurosci. 271, 135–156 (2021).
Vöröslakos M. Direct effects of transcranial electric stimulation on brain circuits in rats and humans. Nat. Commun. Published online 2018.
Herrmann, C. S., Rach, S., Neuling, T. & Strüber, D. Transcranial alternating current stimulation: a review of the underlying mechanisms and modulation of cognitive processes. Front Hum. Neurosci. 7, 279 (2013).
Heise, K. F. et al. Evaluation of a modified high-definition electrode montage for transcranial alternating current stimulation (tACS) of pre-central areas. Brain Stimul. 9, 700–704 (2016).
Grover, S., Nguyen, J. A., Viswanathan, V. & Reinhart, R. M. G. High-frequency neuromodulation improves obsessive-compulsive behavior. Nat. Med 27, 232–238 (2021).
Riddle, J., Rubinow, D. R. & Frohlich, F. A case study of weekly tACS for the treatment of major depressive disorder. Brain Stimul. 13, 576–577 (2020).
Wang, H. et al. Transcranial alternating current stimulation for treating depression: a randomized controlled trial. Brain 145, 83–91 (2022).
Alexander, M. L. et al. Double-blind, randomized pilot clinical trial targeting alpha oscillations with transcranial alternating current stimulation (tACS) for the treatment of major depressive disorder (MDD). Transl. Psychiatry 9, 106 (2019).
Smart, O. L., Tiruvadi, V. R. & Mayberg, H. S. Multimodal approaches to define network oscillations in depression. Biol. Psychiatry 77, 1061–1070 (2015).
Lefaucheur, J. P. et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin. Neurophysiol. 128, 56–92 (2017).
O’Reardon, J. P. et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol. Psychiatry 62, 1208–1216 (2007).
Leuchter, A. F., Cook, I. A., Hunter, A. M., Cai, C. & Horvath, S. Resting-state quantitative electroencephalography reveals increased neurophysiologic connectivity in depression. PLoS One 7, e32508 (2012).
Newson, J. J. & Thiagarajan, T. C. EEG frequency bands in psychiatric disorders: a review of resting state studies. Front Hum. Neurosci. 12, 521 (2018).
Olbrich, S., Tränkner, A., Chittka, T., Hegerl, U. & Schönknecht, P. Functional connectivity in major depression: Increased phase synchronization between frontal cortical EEG-source estimates. Psychiatry Res.: Neuroimaging 222, 91–99 (2014).
Olbrich, S. & Arns, M. EEG biomarkers in major depressive disorder: discriminative power and prediction of treatment response. Int Rev. Psychiatry 25, 604–618 (2013).
van der Vinne, N., Vollebregt, M. A., van Putten, M. J. A. M. & Arns, M. Frontal alpha asymmetry as a diagnostic marker in depression: Fact or fiction? A meta-analysis. Neuroimage Clin. 16, 79–87 (2017).
Kaiser, A. K., Gnjezda, M. T., Knasmüller, S. & Aichhorn, W. Electroencephalogram alpha asymmetry in patients with depressive disorders: current perspectives. Neuropsychiatr. Dis. Treat. 14, 1493–1504 (2018).
Koutsomitros, T., Evagorou, O., Schuhmann, T., Zamar, A. & Sack, A. T. Advances in transcranial magnetic stimulation (TMS) and its applications in resistant depression. Psychiatriki 32, 90–98 (2021).
Ren, C. et al. Transcranial electrical stimulation in treatment of depression: a systematic review and meta-analysis. JAMA Netw. Open 8, e2516459 (2025).
Nikolin, S. et al. Time-course of the tDCS antidepressant effect: An individual participant data meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 125, 110752 (2023).
Thomas, L. J. et al. Cognitive behavioural therapy as an adjunct to pharmacotherapy for treatment resistant depression in primary care: The CoBalT randomised controlled trial protocol. Contemp. Clin. Trials 33, 312–319 (2012).
Strüber, D., Rach, S., Trautmann-Lengsfeld, S. A., Engel, A. K. & Herrmann, C. S. Antiphasic 40 Hz oscillatory current stimulation affects bistable motion perception. Brain Topogr. 27, 158–171 (2014).
Santarnecchi, E. et al. Frequency-dependent enhancement of fluid intelligence induced by transcranial oscillatory potentials. Curr. Biol. 23, 1449–1453 (2013).
Voss, U. et al. Induction of self awareness in dreams through frontal low current stimulation of gamma activity. Nat. Neurosci. 17, 810–812 (2014).
Wilkening, A., Kurzeck, A., Dechantsreiter, E., Padberg, F. & Palm, U. Transcranial alternating current stimulation for the treatment of major depression during pregnancy. Psychiatry Res 279, 399–400 (2019).
Helfrich, R. F. et al. Selective modulation of interhemispheric functional connectivity by HD-tACS shapes perception. PLoS Biol. 12, e1002031 (2014).
Wagner, S. et al. Investigation of tDCS volume conduction effects in a highly realistic head model. J. Neural Eng. 11, 016002 (2014).
Nitsche, M. A. & Bikson, M. Extending the parameter range for tDCS: Safety and tolerability of 4 mA stimulation. Brain Stimul. 10, 541–542 (2017).
Antal, A. & Herrmann, C. S. Transcranial alternating current and random noise stimulation: possible mechanisms. Neural Plast. 2016, 3616807 (2016).
Dmochowski, J. P., Datta, A., Bikson, M., Su, Y. & Parra, L. C. Optimized multi-electrode stimulation increases focality and intensity at target. J. Neural Eng. 8, 046011 (2011).
Neuling, T., Rach, S. & Herrmann, C. S. Orchestrating neuronal networks: sustained after-effects of transcranial alternating current stimulation depend upon brain states. Front Hum. Neurosci. 7, 161 (2013).
Vossen, A., Gross, J. & Thut, G. Alpha power increase after transcranial alternating current stimulation at alpha frequency (α-tACS) reflects plastic changes rather than entrainment. Brain Stimul. 8, 499–508 (2015).
Lee, M., Hong, J. K., Lee, Y. & Yoon, I. Y. Transcranial alternating current stimulation in subjects with insomnia symptoms: A randomized, double-blind and controlled study. J. Psychiatr. Res 186, 129–136 (2025).
Wang, H. X. et al. Effect of transcranial alternating current stimulation for the treatment of chronic insomnia: a randomized, double-blind, parallel-group, placebo-controlled clinical trial. Psychother. Psychosom. 89, 38–47 (2020).
Zhu, X., Ren, Y., Tan, S. & Ma, X. Efficacy of transcranial alternating current stimulation in treating chronic insomnia and the impact of age on its effectiveness: A multisite randomized, double-blind, parallel-group, placebo-controlled study. J. Psychiatr. Res 170, 253–261 (2024).
Saturnino, G. B., Madsen, K. H. & Thielscher, A. Electric field simulations for transcranial brain stimulation using FEM: an efficient implementation and error analysis. J. Neural Eng. 16, 066032 (2019).
Chow S. C., Shao J., Wang H., Lokhnygina Y. Sample Size Calculations in Clinical Research. 3rd Edition. Chapman and Hall/CRC; 2017. https://doi.org/10.1201/9781315183084.
Acknowledgements
This study was funded by the National Natural Science Foundation of China (32571273, 82271515, 82401813, T2293730, T2293734), the Scientific and technological innovation action plan of Shanghai (KY20211478), the Shanghai Municipal Science and Technology Major Project (2021SHZDZX), the Nursing Development Program of Shanghai Jiao Tong University School of Medicine (SJTUHLXK2022), 2024 Shanghai Ruijin Hospital Nursing Research Fund (RJHK-2024-001), and 2024 Shanghai Nursing Association Research Fund (2024MS-B13).
Author information
Authors and Affiliations
Contributions
L.X. and W.Y.H. designed this study and developed the methodology; Y.K.H., W.Y.H., G.H.H., L.D.B., M.Y., S.Z.Y., H.P., P.Y.X. performed the experiments and collected the data. L.X., W.Y.H., and N.C.B. conducted the statistical analysis. L.X., S.B.M., W.J.J., C.Y., S.L., Q.X. supervised the project; W.Y.H., S.B.M. acquired the funding; All authors participated in writing, reviewing, and editing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, Y., Ning, C., Shi, L. et al. A multicenter randomized clinical trial of portable transcranial alternating current stimulation for major depressive disorder. npj Digit. Med. (2026). https://doi.org/10.1038/s41746-026-02575-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-026-02575-9