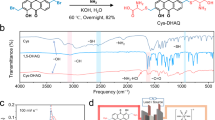
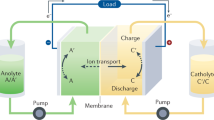
Abstract
The growing global capacity for renewable energy generation necessitates the deployment of energy storage technologies with a combination of low cost, good performance and scalability. With these advantages, aqueous organic flow batteries have the potential to be the system of choice because they could store energy from organic redox-active molecules. Here we report naphthalene derivatives as organic redox-active molecules that exhibit high solubility (~1.5 M) and a stable redox-active framework with no obvious capacity decay over 40 days (50 Ah l−1) in an air atmosphere in flow batteries. We report a battery that runs smoothly even under continuous airflow without obvious capacity decay for ~22 days (more than 600 cycles). A series of spectral analyses and theoretical calculations reveal that the dimethylamine scaffolds improve the water solubility and protect the active centre, ensuring the stability of the molecules during the charge and discharge process. Owing to the success in kilogramme-scale molecular synthesis, pilot-scale stack expansion with notable cycling stability over 270 cycles (~27 days) is attained. The cost benefit evidenced by technoeconomic analysis together with the stability even under open-air conditions indicates the practical value of the present molecular system in grid-scale energy storage.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated and/or analysed from this study are available from the corresponding authors upon reasonable request. Source data are provided with this paper.
References
Zhao, Z. et al. Development of flow battery technologies using the principles of sustainable chemistry. Chem. Soc. Rev. 52, 6031–6074 (2023).
Cameron, J. M. et al. Molecular redox species for next-generation batteries. Chem. Soc. Rev. 50, 5863–5883 (2021).
Zhang, C., Yuan, Z. & Li, X. Designing better flow batteries: an overview on fifty years’ research. ACS Energy Lett. 9, 3456–3473 (2024).
Huskinson, B. et al. A metal-free organic-inorganic aqueous flow battery. Nature 505, 195–198 (2014).
Janoschka, T. et al. An aqueous, polymer-based redox-flow battery using non-corrosive, safe, and low-cost materials. Nature 527, 78–81 (2015).
Wang, J. et al. Conjugated sulfonamides as a class of organic lithium-ion positive electrodes. Nat. Mater. 20, 665–673 (2021).
Feng, R. et al. Reversible ketone hydrogenation and dehydrogenation for aqueous organic redox flow batteries. Science 372, 836–840 (2021).
Hollas, A. et al. A biomimetic high-capacity phenazine-based anolyte for aqueous organic redox flow batteries. Nat. Energy 3, 508–514 (2018).
Zhang, C. et al. Phenothiazine-based organic catholyte for high-capacity and long-life aqueous redox flow batteries. Adv. Mater. 31, 1901052 (2019).
Nguyen, T. P. et al. Polypeptide organic radical batteries. Nature 593, 61–66 (2021).
Janoschka, T., Martin, N., Hager, M. D. & Schubert, U. S. An aqueous redox-flow battery with high capacity and power: the TEMPTMA/MV system. Angew. Chem. Int. Ed. 55, 14427–14430 (2016).
Liu, Y. et al. A long-lifetime all-organic aqueous flow battery utilizing TMAP-TEMPO radical. Chem 5, 1861–1870 (2019).
Feng, R. et al. Proton-regulated alcohol oxidation for high-capacity ketone-based flow battery anolyte. Joule 7, 1609–1622 (2023).
Li, X. et al. Symmetry-breaking design of an organic iron complex catholyte for a long cyclability aqueous organic redox flow battery. Nat. Energy 6, 873–881 (2021).
Luo, J., Hu, B., Hu, M., Zhao, Y. & Liu, T. L. Status and prospects of organic redox flow batteries toward sustainable energy storage. ACS Energy Lett. 4, 2220–2240 (2019).
Kwabi, D. G., Ji, Y. & Aziz, M. J. Electrolyte lifetime in aqueous organic redox flow batteries: a critical review. Chem. Rev. 120, 6467–6489 (2020).
Singh, V., Kim, S., Kang, J. & Byon, H. R. Aqueous organic redox flow batteries. Nano Res. 12, 1988–2001 (2019).
Liu, W. Q. et al. A high potential biphenol derivative cathode: toward a highly stable air-insensitive aqueous organic flow battery. Sci. Bull. 66, 457–463 (2021).
Wedege, K., Dražević, E., Konya, D. & Bentien, A. Organic redox species in aqueous flow batteries: redox potentials, chemical stability and solubility. Sci. Rep. 6, 39101 (2016).
Kwabi, D. G. et al. Alkaline quinone flow battery with long lifetime at pH 12. Joule 2, 1894–1906 (2018).
Wang, C. et al. Molecular design of fused-ring phenazine derivatives for long-cycling alkaline redox flow batteries. ACS Energy Lett. 5, 411–417 (2020).
Pang, S., Wang, X., Wang, P. & Ji, Y. Biomimetic amino acid functionalized phenazine flow batteries with long lifetime at near-neutral pH. Angew. Chem. Int. Ed. 60, 5289–5298 (2021).
Zhang, C. & Li, X. Perspective on organic flow batteries for large-scale energy storage. Curr. Opin. Electrochem. 30, 100836 (2021).
Carrington, M. E. et al. Associative pyridinium electrolytes for air-tolerant redox flow batteries. Nature 623, 949–955 (2023).
Clark, C. D., Debad, J. D., Yonemoto, E. H., Mallouk, T. E. & Bard, A. J. Effect of oxygen on linked Ru(bpy)32+−Viologen species and methylviologen: a reinterpretation of the electrogenerated chemiluminescence. J. Am. Chem. Soc. 119, 10525–10531 (1997).
Levey, G. T. & Ebbesen, W. Methyl viologen radical reactions with several oxidizing agents. J. Phys. Chem. 87, 829–832 (1983).
Zotti, G., Schiavon, G., Zecchin, S. & Favretto, D. Dioxygen-decomposition of ferrocenium molecules in acetonitrile: the nature of the electrode-fouling films during ferrocene electrochemistry. J. Electroanal. Chem. 456, 217–221 (1998).
Zhao, E. W. et al. Coupled in situ NMR and EPR studies reveal the electron transfer rate and electrolyte decomposition in redox flow batteries. J. Am. Chem. Soc. 143, 1885–1895 (2021).
Symons, P. Quinones for redox flow batteries. Curr. Opin. Electrochem. 29, 100759 (2021).
Lu, T. & Chen, Q. Interaction region indicator: a simple real space function clearly revealing both chemical bonds and weak interactions. Chem.–Methods 1, 231–239 (2021).
Dai, Q. et al. High-performance PBI membranes for flow batteries: from the transport mechanism to the pilot plant. Energy Environ. Sci. 15, 1594–1600 (2022).
Frisch, M. J. et al. Gaussian 16 Rev. A.03 (Gaussian, Inc., 2016). https://gaussian.com/citation/
Grimme, S. Accurate description of van der Waals complexes by density functional theory including empirical corrections. J. Comput. Chem. 25, 1463–1473 (2004).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. 113, 6378–6396 (2009).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Acknowledgements
We acknowledge financial support from the National Key R&D Programme of China (grant no. 2022YFB2405000), the National Natural Science Foundation of China (grant nos 22279133 and 22393964), the International Partnership Programme of the Chinese Academy of Sciences (grant no. 121421KYSB20210028) and the CAS Strategic Leading Science & Technology Program (A) (grant no. XDA0400201). We thank X. Ai for his help with the in situ NMR measurement and discussions.
Author information
Authors and Affiliations
Contributions
Z.Z. and C.Z. conceived the idea and designed the experiments. C.Z., S.L. and X.L. supervised the project. Z.Z. conducted the experiments and data analysis. M.Z. and C.Z. helped with battery performance tests. T.L. contributed to the DFT simulation. Z.Z., T.L., C.Z. and X.L. organized and wrote the manuscript. All authors contributed to the discussion and revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Qing Chen, Minjoon Park, Xiongwei Wu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–6, 23–29 and 39 showing spectra analysis, Figs. 7–13, 15–22, 34–37 and 42–44 showing electrochemical or battery analysis, Figs. 14, 30 and 40 presenting digital images, Figs. 31–33 and 38 presenting the theoretical calculations, Fig. 41 showing the cost analysis, Tables 1 and 2 detailing electrochemical or battery properties/performance, Tables 3 and 4 giving cost analysis and Notes 1 and 2 presenting structural stability and battery performance.
Source data
Source Data Fig. 1 (download XLSX )
Data underlying Fig. 1.
Source Data Fig. 2 (download ZIP )
Data underlying Fig. 2.
Source Data Fig. 3 (download XLSX )
Data underlying Fig. 3.
Source Data Fig. 4 (download XLSX )
Data underlying Fig. 4.
Source Data Fig. 5 (download XLSX )
Data underlying Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Z., Li, T., Zhang, C. et al. Air-stable naphthalene derivative-based electrolytes for sustainable aqueous flow batteries. Nat Sustain 7, 1273–1282 (2024). https://doi.org/10.1038/s41893-024-01415-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41893-024-01415-6
This article is cited by
-
An open decoupled cell design achieving electricity generation and amplification through waste-to-energy conversion
Nature Communications (2026)
-
Self-charging organic flow batteries based on multivalent metal negative electrodes
Nature Communications (2025)