Abstract
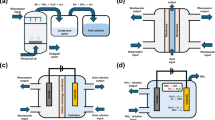
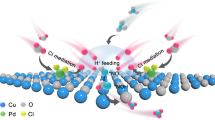
Recovering ammonia (NH3) from ammonium (NH4+)-containing wastewater simultaneously achieves resource recovery and wastewater treatment. Given that NH3 recovery involves a reversible NH4+ hydrolysis reaction, traditional strategy requires alkaline reactants for promoting the reaction forward and energy-intensive heating for recovering NH3, resulting in substantial cost and energy consumption. Here we propose a solar-driven NH3 recovery strategy enabled by floatable amino-grafted (‒NH2) MXene (Ti3C2)-based sponge that possesses local alkaline environment and interfacial heat on water surface. Both H+ trapping via ‒NH2 groups and NH3 evaporating via interfacial solar heating thermodynamically facilitate efficient and sustainable NH3 recovery. Taking ammonia chloride (NH4Cl) wastewater for example, a NH3 recovery rate of 0.6 mol m−2 h−1 with a purity of 99.8% is obtained under 5 sun without extra reagents and energy consumption, and the recovered NH3 can be directly used as nitrogen fertilizer. Besides, the amino-grafted MXene-based sponge is also capable of being fully regenerated to its initial performance under 15 sun, and hydrochloric acid, a valuable by-product, can be obtained during this process. Life-cycle and techno-economic assessments highlight the advantages of solar-driven NH3 recovery in terms of environmental benefits and economic potential.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in this article and its Supplementary Information. Source data are provided with this paper.
References
Service, R. F. New recipe produces ammonia from air, water, and sunlight. Science 345, 610–610 (2014).
Rosca, V., Duca, M., de Groot, M. T. & Koper, M. T. M. Nitrogen cycle electrocatalysis. Chem. Rev. 109, 2209–2244 (2009).
Wang, K. F. et al. Intentional corrosion-induced reconstruction of defective NiFe layered double hydroxide boosts electrocatalytic nitrate reduction to ammonia. Nat. Water 1, 1068–1078 (2023).
Comer, B. M. et al. Prospects and challenges for solar fertilizers. Joule 3, 1578–1605 (2019).
Li, Y. et al. Bipolar membrane electrodialysis for generation of hydrochloric acid and ammonia from simulated ammonium chloride wastewater. Water Res. 89, 201–209 (2016).
Chang, I. S. & Chung, C. M. Pollution prevention for manufacturing of ammonium chloride-an experimental study of wastewater recycling. Desalination 127, 145–153 (2000).
Li, J. Y. et al. Subnanometric alkaline-earth oxide clusters for sustainable nitrate to ammonia photosynthesis. Nat. Commun. 13, 1098 (2012).
Huang, H., Xiao, X. & Yan, B. Complex treatment of the ammonium nitrogen wastewater from rare-earth separation plant. Desalin. Water Treat. 8, 109–117 (2012).
Buelens, L. C., Galvita, V. V., Poelman, H., Detavernier, C. & Marin, G. B. Super-dry reforming of methane intensifies CO2 utilization via Le Chatelier’s principle. Science 354, 449–452 (2016).
Liu, B., Hu, F. & Shi, B. F. Recent advances on ester synthesis via transition-metal catalyzed C–H functionalization. ACS Catal. 5, 1863–1881 (2015).
Li, X. Q. et al. Graphene oxide-based efficient and scalable solar desalination under one sun with a confined 2D water path. Proc. Natl Acad. Sci. USA 113, 13953–13958 (2016).
Xu, N. et al. Going beyond efficiency for solar evaporation. Nat. Water 1, 494–501 (2023).
Chen, C., Kuang, Y. & Hu, L. Challenges and opportunities for solar evaporation. Joule 3, 683–718 (2019).
Li, J. et al. Interfacial solar steam generation enables fast-responsive, energy-efficient, and low-cost off-grid sterilization. Adv. Mater. 30, 1805159 (2018).
Li, Y. et al. 3D-printed, all-in-one evaporator for high-efficiency solar steam generation under 1 sun illumination. Adv. Mater. 29, 1700981 (2017).
Zhou, L. et al. Quantifying hot carrier and thermal contributions in plasmonic photocatalysis. Science 362, 69–72 (2018).
Zhang, Q. et al. Vertically aligned Janus MXene-based aerogels for solar desalination with high efficiency and salt resistance. ACS Nano 13, 13196–13207 (2019).
He, S. et al. Nature-inspired salt resistant bimodal porous solar evaporator for efficient and stable water desalination. Energy Environ. Sci. 12, 1558–1567 (2019).
Zhao, F. et al. Highly efficient solar vapour generation via hierarchically nanostructured gels. Nat. Nanotechnol. 13, 486–495 (2018).
Zielinski, M. S. et al. Hollow mesoporous plasmonic nanoshells for enhanced solar vapor generation. Nano Lett. 16, 2159–2167 (2016).
Li, X. Q. et al. Storage and recycling of interfacial solar steam enthalpy. Joule 2, 2477–2484 (2018).
Wang, Z. X. et al. Versatile coating with multifunctional performance for solar steam generation. Nano Energy 74, 104886 (2020).
Ai, F. R. et al. Amino-functionalized Ti3C2 MXene quantum qots as photoluminescent sensors for diagnosing histidine in human serum. ACS Appl. Nano Mater. 4, 8192–8199 (2021).
Wu, Q. et al. Ultrasensitive and selective determination of carcinoembryonic antigen using multifunctional ultrathin amino-functionalized Ti3C2–MXene nanosheets. Anal. Chem. 92, 3354–3360 (2020).
Haddadi, S. A. et al. Amino-functionalized MXene nanosheets doped with Ce(III) as potent nanocontainers toward self-healing epoxy nanocomposite coating for corrosion protection of mild steel. ACS Appl. Mater. Interfaces 13, 42074–42093 (2021).
Zhang, G. L. et al. Synthesis of amino-functionalized Ti3C2Tx MXene by alkalization-grafting modification for efficient lead adsorption. Chem. Commun. 56, 11283–11286 (2020).
Yang, X. H. et al. Insights into the role of cation vacancy for significantly enhanced electrochemical nitrogen reduction. Appl. Catal. B 264, 118477 (2020).
Xu, N. et al. A water lily-inspired hierarchical design for stable and efficient solar evaporation of high-salinity brine. Sci. Adv. 5, eaaw7013 (2019).
Xu, W. C. et al. Flexible and salt resistant Janus absorbers by electrospinning for stable and efficient solar desalination. Adv. Energy Mater. 8, 1702884 (2018).
Zhou, L. et al. 3D self-assembly of aluminium nanoparticles for plasmon-enhanced solar desalination. Nat. Photon. 10, 393–398 (2016).
Surwade, S. P. et al. Water desalination using nanoporous single-layer graphene. Nat. Nanotechnol. 10, 459–464 (2015).
Zhou, X. Y., Zhao, F., Guo, Y. H., Rosenberger, B. & Yu, G. H. Architecting highly hydratable polymer networks to tune the water state for solar water purification. Sci. Adv. 5, eaaw5484 (2019).
Ahmed, I., Khan, N. A., Yoon, J. W., Chang, J.-S. & Jhung, S. H. Protonated MIL-125-NH2: remarkable adsorbent for the removal of quinoline and indole from liquid fuel. ACS Appl. Mater. Interfaces 9, 20938–20946 (2017).
Rapti, S. A. et al. Selective capture of hexavalent chromium from an anion-exchange column of metal organic resin-alginic acid composite. Chem. Sci. 7, 2427–2436 (2016).
Luo, S. et al. Protonated NH2-MIL-125 via HCl vapor to introduce the moiety with charge and ample hydrogen as a novel bifunctional photocatalyst: enhanced photocatalytic H2 production and NO purification. Chem. Eng. J. 432, 134244 (2022).
Soloveichik, G. Electrochemical synthesis of ammonia as a potential alternative to the Haber–Bosch process. Nat. Catal. 2, 377–380 (2019).
Vinardell, S., Nicolas, P., Sastre, A. M., Cortina, J. L. & Valderrama, C. Sustainability assessment of green ammonia production to promote industrial decarbonization in Spain. ACS Sustain. Chem. Eng. 11, 15975–15983 (2023).
Yao, P. C. et al. Greener and higher conversion of esterification via interfacial photothermal catalysis. Nat. Sustain. 5, 348–356 (2022).
Wankat, P. C. Separation Process Engineering (Pearson Education, 2006).
Acknowledgements
This work is jointly supported by the National Natural Science Foundation of China (grant nos. 52322211 to N.X., 52525202 to J.Z., 92262305 to J.Z., 52302225 to X.W. and 52202251 to H.Z.), National Key R&D Program of China (grant nos. 2024YFF0506000 to J.Z. and 2022YFB3804902 to N.X.), the Fundamental Research Funds for the Central Universities (grant no. 20250107 to N.X.), GeoX’ Interdisciplinary Project of Frontiers Science Center for Critical Earth Material Cycling (grant no. 20250107 to N.X.), Postdoctoral Fellowship Program of CPSF (grant no. 2022M721556 to Q.Z.) and Jiangsu Funding Program for Excellent Postdoctoral Talent (grant nos. 2023ZB416 to Q.Z. and 2023ZB732 to N.C.). This work is also supported by the New Cornerstone Science Foundation through the XPLORER PRIZE, Nanjing U35 Basic Strengthening Program. We acknowledge the microfabrication center at the National Laboratory of Solid State Microstructures (NLSSM) for technical support.
Author information
Authors and Affiliations
Contributions
J.Z., N.X. and Q.Z. conceived of and designed the project. Q.Z., X.W., H.Z. and X.Z. performed the material preparation and characterizations. Q.Z. and T.W. contributed to the recovery experiments. T.W., M.F., N.C. and H.C. performed the calculations. Q.Z., T.W., M.F., N.X. and J.Z. wrote the paper. All the authors discussed the results and commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–27, Notes 1–3 and Tables 1–8.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Statistical source data for Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Q., Wei, T., Fei, M. et al. Solar-driven efficient and selective ammonia recovery from ammonium-containing wastewater. Nat Sustain 8, 1058–1067 (2025). https://doi.org/10.1038/s41893-025-01609-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41893-025-01609-6
This article is cited by
-
Solar-driven ammonia recovery from wastewater using MXene-based sponges
Nature Reviews Materials (2025)