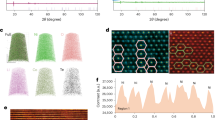
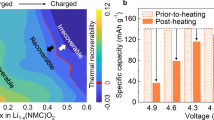
Abstract
The transition to sustainable energy storage demands lithium-ion batteries with high energy density and reduced reliance on critical metals such as nickel (Ni), yet current strategies to increase capacity have largely depended on raising Ni content, leading to escalating supply risks, rising costs and sustainability concerns. More critically, Ni-rich cathodes suffer from rapid electrochemical degradation driven by structural instability, creating an insurmountable trade-off between capacity and cycle life. Here we introduce a low-Ni chemistry cathode, Li(Li0.05Ni0.57Mn0.31Co0.07)O2, with a radial phase integration design that overcomes these limitations, enabling a remarkable Ni usage reduction (Ni < 0.6) while demonstrating high capacity (215 mAh g−1) and markedly improved cyclability (~97% retention over 400 cycles) compared to conventional high-Ni cathodes (Ni = 0.8). Advanced X-ray and electron microscopy analyses reveal that the designed cathode exhibits a highly reversible oxygen anionic redox, benefiting from a structurally stable surface and minimizing irreversible phase transitions. Moreover, the integrated structure substantially mitigates lattice strain and improves mechanical stability even under harsh conditions. This advance offers a general design principle for developing next-generation cathodes that combine resource efficiency with long-term electrochemical reliability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information, which is also available from the corresponding authors upon request. Source data are provided with this paper.
References
Nykvist, B. & Nilsson, M. Rapidly falling costs of battery packs for electric vehicles. Nat. Clim. Change 5, 329–332 (2015).
Kwade, A. et al. Current status and challenges for automotive battery production technologies. Nat. Energy 3, 290–300 (2018).
Borlaug, B. et al. Heavy-duty truck electrification and the impacts of depot charging on electricity distribution systems. Nat. Energy 6, 673–682 (2021).
Deng, J., Bae, C., Denlinger, A. & Miller, T. Electric vehicles batteries: requirements and challenges. Joule 4, 511–515 (2020).
Li, W., Erickson, E. M. & Manthiram, A. High-nickel layered oxide cathodes for lithium-based automotive batteries. Nat. Energy 5, 26–34 (2020).
Thackeray, M. M. & Amine, K. Layered Li–Ni–Mn–Co oxide cathodes. Nat. Energy 6, 933–933 (2021).
Bi, Y. et al. Reversible planar gliding and microcracking in a single-crystalline Ni-rich cathode. Science 370, 1313–1317 (2020).
Nell, K., Valenta, R. K., Forbes, G., Yahyaei, M. & Ilyas, H. M. A. Sustainable resource management: the end of nickel mining? Recycling 9, 102 (2024).
Mervine, E. M. et al. Biomass carbon emissions from nickel mining have significant implications for climate action. Nat. Commun. 16, 481 (2025).
Sun, J., Zhou, H. & Huang, Z. The future nickel metal supply for lithium-ion batteries. Green. Chem. 26, 6926 (2024).
Mudd, G. M. Global trends and environmental issues in nickel mining: sulfides versus laterites. Ore Geol. Rev. 38, 9–26 (2010).
Zhang, R. et al. Long-life lithium-ion batteries realized by low-Ni, Co-free cathode chemistry. Nat. Energy 8, 695–702 (2023).
Lu, J. et al. Microstructures of layered Ni-rich cathodes for lithium-ion batteries. Chem. Soc. Rev. 53, 4707–4740 (2024).
Zhang, H., Liu, H., Piper, L. F. J., Whittingham, M. S. & Zhou, G. Oxygen loss in layered oxide cathodes for Li-ion batteries: mechanisms, effects, and mitigation. Chem. Rev. 122, 5641–5681 (2022).
Liu, T. et al. Understanding Co roles towards developing Co-free Ni-rich cathodes for rechargeable batteries. Nat. Energy 6, 277–286 (2021).
Genreith-Schriever, A. R. et al. Oxygen hole formation controls stability in LiNiO2 cathodes. Joule 7, 1623–1640 (2023).
Wang, L., Liu, T., Wu, T. & Lu, J. Strain-retardant coherent perovskite phase stabilized Ni-rich cathode. Nature 611, 61–67 (2022).
Huang, W. et al. Unrecoverable lattice rotation governs structural degradation of single-crystalline cathodes. Science 384, 912–919 (2024).
Zhang, S. S. Problems and their origins of Ni-rich layered oxide cathode materials. Energy Storage Mater. 24, 247–254 (2020).
Ryu, H.-H., Park, K.-J., Yoon, C. S. & Sun, Y.-K. Capacity fading of Ni-rich Li[NixCoyMn1–x–y]O2 (0.6 ≤ x ≤ 0.95) cathodes for high-energy-density lithium-ion batteries: bulk or surface degradation? Chem. Mater. 30, 1155–1163 (2018).
Yin, S. et al. Fundamental and solutions of microcrack in Ni-rich layered oxide cathode materials of lithium-ion batteries. Nano Energy 83, 105854 (2021).
Park, N. Y. et al. Mechanism of doping with high-valence elements for developing Ni-rich cathode materials. Adv. Energy Mater. 13, 2301530 (2023).
Cui, Z., Li, X., Bai, X., Ren, X. & Ou, X. A comprehensive review of foreign-ion doping and recent achievements for nickel-rich cathode materials. Energy Storage Mater. 57, 14–43 (2023).
Xu, G.-L. et al. Building ultraconformal protective layers on both secondary and primary particles of layered lithium transition metal oxide cathodes. Nat. Energy 4, 484–494 (2019).
Yan, P. et al. Tailoring grain boundary structures and chemistry of Ni-rich layered cathodes for enhanced cycle stability of lithium-ion batteries. Nat. Energy 3, 600–605 (2018).
Sun, Y. K. et al. Nanostructured high-energy cathode materials for advanced lithium batteries. Nat. Mater. 11, 942–947 (2012).
Li, Y. et al. Synthesis of full concentration gradient cathode studied by high energy X-ray diffraction. Nano Energy 19, 522–531 (2016).
Hua, W. et al. Lithium-ion (de)intercalation mechanism in core-shell layered Li(Ni,Co,Mn)O2 cathode materials. Nano Energy 78, 105231 (2020).
Yang, J. et al. Improving the cycling performance of the layered Ni-rich oxide cathode by introducing low-content Li2MnO3. Electrochim. Acta 189, 101–110 (2016).
Wu, B. et al. In situ mitigating cation mixing of Ni-rich cathode at high voltage via Li2MnO3 injection. Energy Storage Mater. 53, 212–221 (2022).
The latest trends in lithium-ion batteries. BNamericas https://www.bnamericas.com/en/features/the-biggest-trends-in-lithium-ion-batteries (2019).
Marie, J.-J. et al. Trapped O2 and the origin of voltage fade in layered Li-rich cathodes. Nat. Mater. 23, 818–825 (2024).
He, W. et al. Challenges and recent advances in high capacity Li-rich cathode materials for high energy density lithium-ion batteries. Adv. Mater. 33, 2005937 (2021).
Jarvis, K. A., Deng, Z., Allard, L. F., Manthiram, A. & Ferreira, P. J. Atomic structure of a lithium-rich layered oxide material for lithium-ion batteries: evidence of a solid solution. Chem. Mater. 23, 3614–3621 (2011).
House, R. A. et al. Superstructure control of first-cycle voltage hysteresis in oxygen-redox cathodes. Nature 577, 502–508 (2020).
Huang, W. et al. Delocalized Li@Mn6 superstructure units enable layer stability of high-performance Mn-rich cathode materials. Chem 8, 2163–2178 (2022).
Luo, D. et al. A Li-rich layered oxide cathode with negligible voltage decay. Nat. Energy 8, 1078–1087 (2023).
Zhao, S. et al. Reaction mechanisms of layered lithium-rich cathode materials for high-energy lithium-ion batteries. Angew. Chem. Int. Ed. 60, 2208–2220 (2021).
Assat, G. et al. Fundamental interplay between anionic/cationic redox governing the kinetics and thermodynamics of lithium-rich cathodes. Nat. Commun. 8, 2219 (2017).
Gu, M. et al. Formation of the spinel phase in the layered composite cathode used in Li-ion batteries. ACS Nano 7, 760–767 (2013).
Zeng, L. et al. Voltage decay of Li-rich layered oxides: mechanism, modification strategies, and perspectives. Adv. Funct. Mater. 33, 2213260 (2023).
Yan, P. et al. Atomic resolution structural and chemical imaging revealing the sequential migration of Ni, Co, and Mn upon the battery cycling of layered cathode. Nano Lett. 17, 3946–3951 (2017).
Liu, X. et al. Origin and regulation of oxygen redox instability in high-voltage battery cathodes. Nat. Energy 7, 808–817 (2022).
Yan, P. et al. Injection of oxygen vacancies in the bulk lattice of layered cathodes. Nat. Nanotechnol. 14, 602–608 (2019).
Zhuo, Z. et al. Distinct oxygen redox activities in Li2MO3 (M = Mn, Ru, Ir). ACS Energy Lett. 6, 3417–3424 (2021).
Gao, X. et al. Clarifying the origin of molecular O2 in cathode oxides. Nat. Mater. 24, 743–752 (2025).
Yang, W. Extrinsic observation and intrinsic state. Nat. Mater. 24, 660–661 (2025).
Balasubramanian, M., Sun, X., Yang, X. & McBreen, J. In situ X-ray diffraction and X-ray absorption studies of high-rate lithium-ion batteries. J. Power Source 92, 1–8 (2001).
Zhang, H. et al. Layered oxide cathodes for Li-ion batteries: oxygen loss and vacancy evolution. Chem. Mater. 31, 7790–7798 (2019).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Rad. 12, 537–541 (2005).
Li, Q. et al. Could irradiation introduce oxidized oxygen signals in resonant inelastic X-ray scattering of battery electrodes? J. Phys. Chem. Lett. 12, 1138–1143 (2021).
Xiao, X. et al. TXM-Sandbox: an open-source software for transmission X-ray microscopy data analysis. J. Synchrotron Rad. 29, 266–275 (2022).
Acknowledgements
This work gratefully acknowledges support from the US Department of Energy (DOE), Office of Energy Efficiency and Renewable Energy, Vehicle Technologies Office. This work was performed, in part, at the Center for Nanoscale Materials, an Office of Science user facility, supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under contract no. DE-AC02-06CH11357. This research used resources of the Advanced Photon Source (11-ID-C), a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. This research used resources of the Advanced Light Source, which is a DOE Office of Science User Facility under contract no. DE-AC02-05CH11231. Use of the National Synchrotron Light Source II (3-ID, 7-BM, 18-ID and 28-ID) is supported by the US Department of Energy, an Office of Science user facility operated by Brookhaven National Laboratory under contract no. DE-SC0012704.
Author information
Authors and Affiliations
Contributions
W.H., T. Liu and K.A. conceived the idea and designed the experiments. W.H. synthesized all the materials. W.H., A.D., R.A. and S.T. conducted electrochemical measurements. Z.Z. and J.G. conducted the mRIXS. J.H., T.Z., H.A. and J. Wen carried out the TEM results. X.-M.L. carried out the ICP-OES. W.H., J. Wang, G.K. and T. Li performed synchrotron HEXRD and PDF. W.H. and L.M. performed XAS. W.H. and X.X. conducted the TXM. X.H. conducted the XRF. W.H., T. Liu and K.A. wrote the paper, and all authors edited the paper.
Corresponding authors
Ethics declarations
Competing interests
T. Liu and K.A. report one US non-provisional patent application filed by the Argonne National Laboratory, patent application no. 18/105,469. The patent is related to the composition and structure design reported in this work. The other authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Weibo Hua, Laura Simonelli and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–30, Table 1 and references 1 and 2.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data for Fig. 1a,d.
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2a–g.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3a–f.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4b–j.
Source Data Fig. 5 (download XLSX )
Statistical source data for Fig. 5d.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, W., Zhuo, Z., Dai, A. et al. Low-nickel cathode chemistry for sustainable and high-energy lithium-ion batteries. Nat Sustain 9, 317–327 (2026). https://doi.org/10.1038/s41893-025-01704-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41893-025-01704-8