Abstract
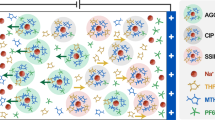
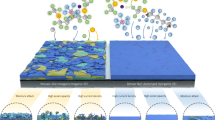
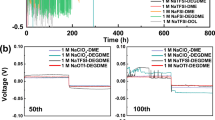
Abundant sodium (Na) batteries are a sustainable alternative to resource-constrained lithium-ion batteries, offering huge cost advantages. However, developing high-voltage anode-free sodium metal batteries (SMBs) to narrow the energy density gap with lithium-ion batteries is hindered by a critical challenge: existing electrolytes cannot simultaneously achieve ultra-high Na coulombic efficiency and anodic stability. Here we present a rationally designed non-fluorinated electrolyte (1.0 M NaPF6 in 1,2-diethoxyethane/1,2-di-tert-butoxyethane) to address this key limitation, achieving Na coulombic efficiency of >99.95% and anodic stability of >4.8 V. For coin cells (2.0 mAh cm−2, N/P = 1.7), our electrolyte design enables 4.0 V Na | |Na3V2(PO4)3 (NVP) at 5 C and 4.3 V Na | |NaNi0.6Mn0.2Co0.2O2 (NMC622) at 0.3 C for 5,000 and 500 cycles with a capacity retention >80%. Remarkably, the 50 mAh anode-free pouch cells 4.0 V Al | |NVP and 4.3 V Al | |NMC622 also achieve 500 and 300 cycles (retention >75%) with a specific energy of >360 Wh kg(electrode)−1. This work focuses on electrolyte optimization and conceptual advances, whereas critical aspects such as safety, large-scale manufacturability and practical feasibility of SMBs require further investigation. The electrolyte design using non-fluorinated solvents enhances the anodic stability without sacrificing Na efficiency, laying groundwork for advancing low-cost, high-energy SMBs and supporting the transition to sustainable battery technologies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information files. Source data are provided with this paper.
Code availability
Gaussian and VASP are commercial codes, RDKIT and CREST are freely available at https://www.rdkit.org and https://crest-lab.github.io/crest-docs/, and a MD simulation code is provided in Supplementary Materials. A specific version of the simulation code is attached as supporting archive to this paper together with the force field files and initial configurations. DFT calculations were performed using Gaussian 16 C.02 package, and VASP 5.4.4. rdkit and Crest were used to sample DME, DBE, TTE conformers.
References
Vaalma, C. et al. A cost and resource analysis of sodium-ion batteries. Nat. Rev. Mater. 3, 18013 (2018).
Choi, J. W. & Aurbach, D. Promise and reality of post-lithium-ion batteries with high energy densities. Nat. Rev. Mater. 1, 16013 (2016).
Usiskin, R. et al. Fundamentals, status and promise of sodium-based batteries. Nat. Rev. Mater. 6, 1020–1035 (2021).
Zhao, Y. et al. Recent developments and insights into the understanding of Na metal anodes for Na-metal batteries. Energy Environ. Sci. 11, 2673–2695 (2018).
Zhao, C. et al. Rational design of layered oxide materials for sodium-ion batteries. Science 370, 708–711 (2020).
Song, J. et al. Controlling surface phase transition and chemical reactivity of O3-layered metal oxide cathodes for high-performance Na-ion batteries. ACS Energy Lett. 5, 1718–1725 (2020).
Jin, Y. et al. Low-solvation electrolytes for high-voltage sodium-ion batteries. Nat. Energy 7, 718–725 (2022).
Ni, Q. et al. Anode-free rechargeable sodium-metal batteries. Batteries 8, 272 (2022).
Yang, T. et al. Anode-free sodium metal batteries as rising stars for lithium-ion alternatives. iScience 26, 105982 (2023).
Sun, B. et al. Design strategies to enable the efficient use of sodium metal anodes in high-energy batteries. Adv. Mater. 32, e1903891 (2020).
Zheng, X. et al. Critical effects of electrolyte recipes for Li and Na metal batteries. Chem 7, 2312–2346 (2021).
Chen, J. et al. High energy density Na-metal batteries enabled by a tailored carbonate-based electrolyte. Energy Environ. Sci. 15, 3360–3368 (2022).
Chen, X. et al. Ion-solvent chemistry-inspired cation-additive strategy to stabilize electrolytes for sodium-metal batteries. Chem 6, 2242–2256 (2020).
Zheng, X. et al. Bridging the immiscibility of an all-fluoride fire extinguishant with highly-fluorinated electrolytes toward safe sodium metal batteries. Energy Environ. Sci. 13, 1788–1798 (2020).
Zheng, X. et al. Knocking down the kinetic barriers towards fast-charging and low-temperature sodium metal batteries. Energy Environ. Sci. 14, 4936–4947 (2021).
Chen, F. et al. Cationic polymer-in-salt electrolytes for fast metal ion conduction and solid-state battery applications. Nat. Mater. 21, 1175–1182 (2022).
Seh, Z. W. et al. A highly reversible room-temperature sodium metal anode. ACS Cent. Sci. 1, 449–455 (2015).
Hobold, G. M. et al. Moving beyond 99.9% coulombic efficiency for lithium anodes in liquid electrolytes. Nat. Energy 6, 951–960 (2021).
Li, S. et al. Revisiting ether electrolytes for high-voltage sodium-ion batteries. Energy Storage Mater. 73, 103815 (2024).
Zou, Y. et al. Electrolyte solvation engineering stabilizing anode-free sodium metal battery with 4.0 V-class layered oxide cathode. Adv. Mater. 36, 2410261 (2024).
Zhang, Y. Y. et al. Refined electrolyte and interfacial chemistry toward realization of high-energy anode-free rechargeable sodium batteries. J. Am. Chem. Soc. 145, 25643–25652 (2023).
Qian, J. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Cao, X. et al. Monolithic solid–electrolyte interphases formed in fluorinated orthoformate-based electrolytes minimize Li depletion and pulverization. Nat. Energy 4, 796–805 (2019).
Cao, R. et al. Enabling room temperature sodium metal batteries. Nano Energy 30, 825–830 (2016).
Zheng, J. et al. Extremely stable sodium metal batteries enabled by localized high-concentration electrolytes. ACS Energy Lett. 3, 315–321 (2018).
Bhide, A. et al. Electrochemical stability of non-aqueous electrolytes for sodium-ion batteries and their compatibility with Na0.7CoO2. Phys. Chem. Chem. Phys. 16, 1987–1998 (2014).
Morales, D. et al. Ion transport and association study of glyme-based electrolytes with lithium and sodium salts. Electrochim. Acta 304, 239–245 (2019).
Wang, C. et al. Extending the low-temperature operation of sodium metal batteries combining linear and cyclic ether-based electrolyte solutions. Nat. Commun. 13, 4934 (2022).
Wang, C. et al. Robust anode-free sodium metal batteries enabled by artificial sodium formate interface. Adv. Energy Mater. 13, 2204125 (2023).
Zhuang, R. et al. Fluorinated porous frameworks enable robust anode-less sodium metal batteries. Sci. Adv. 9, eadh8060 (2023).
Wang, S. et al. Unraveling the solvent effect on solid-electrolyte interphase formation for sodium metal batteries. Angew. Chem. Int. Ed. 62, e202313447 (2023).
Lu, Z. et al. Building a beyond concentrated electrolyte for high-voltage anode-free rechargeable sodium batteries. Angew. Chem. Int. Ed. 61, e202200410 (2022).
Li, Y. et al. Interfacial engineering to achieve an energy density of over 200 Wh kg−1 in sodium batteries. Nat. Energy 7, 511–519 (2022).
Sun, W. et al. Insights into the oxidation kinetics of a cetane improver–1,2-dimethoxyethane (1,2-DME) with experimental and modeling methods. Proc. Combust. Inst. 37, 555–564 (2019).
Di Tommaso, S. et al. Oxidation mechanism of aliphatic ethers: theoretical insights on the main reaction channels. J. Phys. Chem. A 116, 9010–9019 (2012).
Yu, Z. et al. Molecular design for electrolyte solvents enabling energy-dense and long-cycling lithium metal batteries. Nat. Energy 5, 526–533 (2020).
Huang, Z. et al. A stable lithium–oxygen battery electrolyte based on fully methylated cyclic ether. Angew. Chem. Int. Ed. 58, 2345–2349 (2019).
Li, A. M. et al. Methylation enables the use of fluorine-free ether electrolytes in high-voltage lithium metal batteries. Nat. Chem. 16, 922–929 (2024).
Bai, Y. et al. Energy and environmental aspects in recycling lithium-ion batteries: concept of battery identity global passport. Mater. Today 41, 304–315 (2020).
Bartoli, G. et al. Alcohols and di-tert-butyl dicarbonate: how the nature of the Lewis acid catalyst may address the reaction to the synthesis of tert-butyl ethers. J. Org. Chem. 71, 9580–9588 (2006).
Carboni, M. et al. 1, 2-Dimethoxyethane degradation thermodynamics in Li−O2 redox environments. Chem. Eur. J. 22, 17188–17203 (2016).
Adams, B. D. et al. Towards a stable organic electrolyte for the lithium-oxygen battery. Adv. Energy Mater. 5, 1400867 (2015).
Zhou, L. et al. Electrolyte engineering enables high stability and capacity alloying anodes for sodium and potassium ion batteries. ACS Energy Lett. 5, 766–776 (2020).
Han, S. D. et al. Solvate structures and computational/spectroscopic characterization of LiPF6 electrolytes. J. Phys. Chem. C. 119, 8492–8500 (2015).
Kitashita, K. et al. Crystal structures of some cubic hexafluorophosphates at ambient temperatures. J. Fluor. Chem. 101, 173–179 (2000).
Cresce, A. V. et al. Solvation behavior of carbonate-based electrolytes in sodium ion batteries. Phys. Chem. Chem. Phys. 19, 574–586 (2017).
Peng, J. et al. Multinuclear magnetic resonance investigation of cation-anion and anion-solvent interactions in carbonate electrolytes. J. Power Sources 399, 215–222 (2018).
Shin, W. & Manthiram, A. A facile potential hold method for fostering an inorganic solid-electrolyte interphase for anode-free lithium-metal batteries. Angew. Chem. Int. Ed. 61, e202115909 (2022).
Zhang, C. et al. A dual-layered artificial solid electrolyte interphase formed by controlled electrochemical reduction of LiTFSI/DME-LiNO3 for dendrite-free lithium metal anode. Electrochim. Acta 306, 407–419 (2019).
Heng, S. et al. Fluoro-ether as a bifunctional interphase electrolyte additive with graphite/LiNi0.5Co0.2Mn0.3O2 full cell. ACS Appl. Energy Mater. 2, 6404–6416 (2019).
Choudhury, S. et al. Designing solid-liquid interphases for sodium batteries. Nat. Commun. 8, 898 (2017).
Song, J. et al. Interphases in sodium-ion batteries. Adv. Energy Mater. 8, 1703082 (2018).
Pang, Y. et al. Electrolyte/electrode interfaces in all-solid-state lithium batteries: a review. Electrochem. Energy Rev. 4, 169–193 (2021).
Zhang, J. et al. Advances in electrode/electrolyte interphase for sodium-ion batteries from half cells to full cells. Cell. Rep. Phys. Sci. 3, 100868 (2022).
Lee, J. et al. Ultraconcentrated sodium bis(fluorosulfonyl)imide-based electrolytes for high-performance sodium metal batteries. ACS Appl. Mater. Interfaces 9, 3723–3732 (2017).
Zhang, G. et al. A monofluoride ether-based electrolyte solution for fast-charging and low-temperature non-aqueous lithium metal batteries. Nat. Commun. 14, 1081 (2023).
Liu, T. et al. Recycled micro-sized silicon anode for high-voltage lithium-ion batteries. Nat. Sustain. 7, 1057–1066 (2024).
Zeng, X. et al. Research progress on Na3V2(PO4)3 cathode material of sodium ion battery. Front. Chem. 8, 635 (2020).
Delmas, C. et al. The layered oxides in lithium and sodium-ion batteries: a solid-state chemistry approach. Adv. Energy Mater. 11, 2001201 (2021).
Zuo, W. et al. Microstrain screening towards defect-less layered transition metal oxide cathodes. Nat. Nanotechnol. 19, 1644–1653 (2024).
Li, A. M. et al. Salt-in-presalt electrolyte solutions for high-potential non-aqueous sodium metal batteries. Nat. Nanotechnol. 20, 388–396 (2025).
Tang, Y. et al. Sustainable layered cathode with suppressed phase transition for long-life sodium-ion batteries. Nat. Sustain. 7, 348–359 (2024).
Gao, A. et al. Topologically protected oxygen redox in a layered manganese oxide cathode for sustainable batteries. Nat. Sustain. 5, 214–224 (2022).
Yuan, X. G. et al. A universal strategy toward air-stable and high-rate O3 layered oxide cathodes for Na-ion batteries. Adv. Funct. Mater. 32, 2111466 (2022).
Xu, G. L. et al. Challenges in developing electrodes, electrolytes, and diagnostics tools to understand and advance sodium-ion batteries. Adv. Energy Mater. 8, 1702403 (2018).
Huang, Q. et al. Fading mechanisms and voltage hysteresis in FeF2–NiF2 solid solution cathodes for lithium and lithium-ion batteries. Small 15, 1804670 (2019).
Østergaard, T. M. et al. Oxidation of ethylene carbonate on Li metal oxide surfaces. J. Phys. Chem. C. 122, 10442–10449 (2018).
Xiao, B. et al. Protonation stimulates the layered to rock salt phase transition of Ni-rich sodium cathodes. Adv. Mater. 36, 2308380 (2024).
Alvarado, J. et al. Bisalt ether electrolytes: a pathway towards lithium metal batteries with Ni-rich cathodes. Energy Environ. Sci. 12, 780–794 (2019).
Yao, A. et al. Critically assessing sodium-ion technology roadmaps and scenarios for techno-economic competitiveness against lithium-ion batteries. Nat. Energy 10, 404–416 (2025).
Bursch, M. et al. Best-practice DFT protocols for basic molecular computational chemistry. Angew. Chem. Int. Ed. 61, e202205735 (2022).
Gaussian 16 Rev. C.01 (Gaussian, 2016).
Han, S. D. et al. Electrolyte solvation and ionic association. V. acetonitrile-lithium bis(fluorosulfonyl)imide (LiFSI) mixtures. J. Electrochem. Soc. 161, A2042–A2053 (2014).
Borodin, O. Polarizable force field development and molecular dynamics simulations of ionic liquids. J. Phys. Chem. B 113, 11463–11478 (2009).
Fang, C. et al. Elucidating the molecular origins of the transference number in battery electrolytes using computer simulations. JACS Au 3, 306–315 (2023).
Acknowledgements
This work was supported by the US Department of Energy (DOE) under award number DE-EE0008202 (C.W.) at the University of Maryland (UMD) and subcontract number 725470 with Pacific Northwest National Laboratory (PNNL). Modelling work was supported by U.S. Army Combat Capabilities Development Command Army Research Laboratory (DEVCOM ARL) and Center for Research in Extreme Batteries (CREB) (O.B. and T.P.P.). The work at Brookhaven National Laboratory is supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technology Office of US DOE through the Advanced Battery Materials Research (BMR) Program under contract number DE-SC0012704 (E.H. and X.-Q.Y.). This research used 28-ID-2 beamline of the National Synchrotron Light Source II, US DOE Office of Science User Facilities, operated for the DOE Office of Science by Brookhaven National Laboratory under contract number DE-SC0012704 (E.H. and X.-Q.Y.).
Author information
Authors and Affiliations
Contributions
A.-M.L. and C.W. conceived the idea for the project and wrote the paper. O.B. and T.P.P. conducted and analysed the DFT calculations and MD simulations, and participated in the paper revision and mechanism identification. Z.W. assisted with DFT analysis. N.Z. helped with Raman data collection. F.O. and X.L. helped with the NMC622 cathode preparation and discussion. S.T., E.H. and X.-Q.Y. helped with synchrotron data collection and analysis. All authors participated in discussion of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Jang Wook Choi, Atsuo Yamada and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–8, Tables 1–4, Figs. 1–41 and References.
Supplementary Code 1 (download ZIP )
The customized simulation codes and force field files with initial configurations for this study.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2b,c.
Source Data Fig. 6 (download XLSX )
Statistical source data for Fig. 6a.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, AM., Pollard, T.P., Wang, Z. et al. Non-fluorinated electrolyte for high-voltage anode-free sodium metal battery. Nat Sustain 9, 306–316 (2026). https://doi.org/10.1038/s41893-025-01710-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41893-025-01710-w