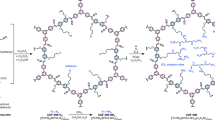
Abstract
Capturing CO2 directly from ambient air is necessary for managing carbon levels and supporting long-term climate sustainability. However, the slow adsorption and desorption kinetics of current direct air capture sorbents remain a major limitation, whereas faster kinetics allow for quicker CO2 uptake and greater air throughput—both are essential for enhancing system efficiency. In this work, we present a covalent organic framework (COF) with both fast kinetics and high CO2 uptake. The COF (termed COF-1000) exhibited a CO2 capacity of 1.31 mmol g−1 under dry conditions at 400 ppm CO2, reaching half of its capacity within 8.1 min. Under humid conditions (75% relative humidity), water further enhanced both uptake and kinetics, leading to a remarkable CO2 capacity of 2.19 mmol g−1 with a reduced half-capacity time of 6.8 min. The exceptionally fast kinetics observed for COF-1000 were further demonstrated by using outdoor air as the CO2 source, where 50 adsorption–desorption cycles were conducted within 3 days, yielding a CO2 uptake of 22.1 mmol g−1 d−1, a value exceeding the current state-of-the-art materials. These results highlight COF-1000’s potential to enable efficient, scalable direct air capture and promote sustainable carbon mitigation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main text or Supplementary Information. Source data are provided with this paper.
References
Lackner, K., Ziock, H.-J. & Grimes, P. Carbon dioxide extraction from air: is it an option? In 24th Annual Technical Conference on Coal Utilization and Fuel Systems (Clearwater, 1999).
Lackner, K. S. et al. The urgency of the development of CO2 capture from ambient air. Proc. Natl Acad. Sci. USA 109, 13156–13162 (2012).
Sanz-Pérez, E. S., Murdock, C. R., Didas, S. A. & Jones, C. W. Direct capture of CO2 from ambient air. Chem. Rev. 116, 11840–11876 (2016).
Shi, X. et al. Sorbents for the direct capture of CO2 from ambient air. Angew. Chem. Int. Ed. 59, 6984–7006 (2020).
Panda, D., Kulkarni, V. & Singh, S. K. Evaluation of amine-based solid adsorbents for direct air capture: a critical review. React. Chem. Eng. 8, 10–40 (2023).
Kuwahara, Y. et al. Enhanced CO2 adsorption over polymeric amines supported on heteroatom-incorporated SBA-15 silica: impact of heteroatom type and loading on sorbent structure and adsorption performance. Chem. Eur. J. 18, 16649–16664 (2012).
Choi, S., Drese, J. H., Eisenberger, P. M. & Jones, C. W. Application of amine-tethered solid sorbents for direct CO2 capture from the ambient air. Environ. Sci. Technol. 45, 2420–2427 (2011).
Abhilash, K. A. S., Deepthi, T., Sadhana, R. A. & Benny, K. G. Functionalized polysilsesquioxane-based hybrid silica solid amine sorbents for the regenerative removal of CO2 from air. ACS Appl. Mater. Interfaces 7, 17969–17976 (2015).
Deutz, S. & Bardow, A. Life-cycle assessment of an industrial direct air capture process based on temperature–vacuum swing adsorption. Nat. Energy 6, 203–213 (2021).
Shekhah, O. et al. Made-to-order metal-organic frameworks for trace carbon dioxide removal and air capture. Nat. Commun. 5, 4228 (2014).
Lyu, H. et al. Carbon dioxide capture chemistry of amino acid functionalized metal-organic frameworks in humid flue gas. J. Am. Chem. Soc. 144, 2387–2396 (2022).
Chen, O. I. F. et al. Water-enhanced direct air capture of carbon dioxide in metal-organic frameworks. J. Am. Chem. Soc. 146, 2835–2844 (2024).
He, H. et al. Porous polymers prepared via high internal phase emulsion polymerization for reversible CO2 capture. Polymer 55, 385–394 (2014).
Song, J. et al. Quaternized chitosan/PVA aerogels for reversible CO2 capture from ambient air. Ind. Eng. Chem. Res. 57, 4941–4948 (2018).
Hou, C., Wu, Y., Wang, T., Wang, X. & Gao, X. Preparation of quaternized bamboo cellulose and its implication in direct air capture of CO2. Energy. Fuels 33, 1745–1752 (2019).
He, H. et al. Reversible CO2 capture with porous polymers using the humidity swing. Energy Environ. Sci. 6, 488–493 (2013).
Wang, X. et al. Development of high capacity moisture-swing DAC sorbent for direct air capture of CO2. Sep. Purif. Technol. 324, 124489 (2023).
Lyu, H., Li, H., Hanikel, N., Wang, K. & Yaghi, O. M. Covalent organic frameworks for carbon dioxide capture from air. J. Am. Chem. Soc. 144, 12989–12995 (2022).
Zhou, Z. et al. Carbon dioxide capture from open air using covalent organic frameworks. Nature 635, 96–101 (2024).
Li, H. et al. Bonding of polyethylenimine in covalent organic frameworks for CO2 capture from air. J. Am. Chem. Soc. 146, 35486–35492 (2024).
Yaghi, O. M., Kalmutzki, M. J. & Diercks, C. S. Introduction to Reticular Chemistry (Wiley, 2019); https://doi.org/10.1002/9783527821099
Forse, A. C. et al. Influence of pore size on carbon dioxide diffusion in two isoreticular metal–organic frameworks. Chem. Mater. 32, 3570–3576 (2020).
Brunauer, S., Emmett, P. H. & Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60, 309–319 (1938).
Wang, Y. et al. Carbon capture in polymer-based electrolytes. Sci. Adv. 10, eadk2350 (2024).
Mao, H. et al. A scalable solid-state nanoporous network with atomic-level interaction design for carbon dioxide capture. Sci. Adv. 8, eabo6849 (2022).
Al Mohtar, A. & Pinto, M. L. Water effect on CO2 adsorption kinetics in amine-functionalized silicas assessed using the density functional theory. J. Phys. Chem. C 126, 5159–5166 (2022).
He, H. et al. Carbon black functionalized with hyperbranched polymers: synthesis, characterization, and application in reversible CO2 capture. J. Mater. Chem. A 1, 6810–6821 (2013).
Gebald, C., Wurzbacher, J. A., Tingaut, P., Zimmermann, T. & Steinfeld, A. Amine-based nanofibrillated cellulose as adsorbent for CO2 capture from air. Environ. Sci. Technol. 45, 9101–9108 (2011).
Choi, S., Gray, M. L. & Jones, C. W. Amine-tethered solid adsorbents coupling high adsorption capacity and regenerability for CO2 capture from ambient air. ChemSusChem 4, 628–635 (2011).
Masuda, S., Osaka, Y., Tsujiguchi, T. & Kodama, A. High-purity CO2 recovery following two-stage temperature swing adsorption using an internally heated and cooled adsorber. Sep. Purif. Technol. 309, 123062 (2023).
Acknowledgements
We thank A. Alawadhi for his help in collecting water sorption data. We also thank H. Li and Y. Xie for their help in collecting gas chromatography-mass spectrometry (GC-MS) data. We thank Y. Shi and Z. Rong from the Yaghi Research Group for their valuable discussions. We acknowledge H. Celik, R. Giovine and Pines Magnetic Resonance Center’s Core NMR Facility (PMRC Core) for spectroscopic assistance. We also thank M. Kang and the UC Berkeley Electron Microscope Laboratory for access and assistance in scanning electron microscopy (SEM) data collection. This research was supported by the King Abdulaziz City for Science and Technology (Center of Excellence for Nanomaterials and Clean Energy Applications), ATOCO Inc. and the Bakar Institute of Digital Materials for the Planet. Z.Z. acknowledges the support from Kavli Energy NanoScience Institute (ENSI) Graduate Student Fellowship. Z.Z. and O.M.Y. acknowledge the interest and support of Fifth Generation Inc. (Love, Tito’s). The NMR instrument used in this work was in part supported by the National Institutes of Health (NIH) under grant S10OD024998. The SEM instrument used in this work is supported by NIH under grant S10OD030258-01.
Author information
Authors and Affiliations
Contributions
Z.Z. and O.M.Y. conceived of the idea and led the experimental efforts. Z.Z. and T.M. developed synthetic methodologies and conducted PXRD, thermogravimetric analysis (TGA), FT-IR and gas sorption experiments. Z.Z. conducted NMR and breakthrough experiments. Z.Z., T.M., H.Z. and N.S.S. synthesized the COFs. T.M. and H.Z. collected SEM images. Z.Z. and O.M.Y. prepared the initial draft and finalized it. All authors contributed to revising the paper.
Corresponding author
Ethics declarations
Competing interests
COF-1000, COF-998 and their related materials have been filed as international patent application (number PCT/US2024/049400). O.M.Y. and Z.Z. are the inventors of this patent. O.M.Y. is a co-founder of ATOCO Inc., aiming at commercializing related technologies. The other authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Dan Zhao and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Ideal chemical structure of COF-1000.
COF-1000 possesses an hcb topology with polyamine functional groups incorporated within the pores.
Extended Data Fig. 2 CO2 uptake of COF-1000 under humid simulated air (400 ppm of CO2 with 50% RH) at 25 °C, obtained from 200 rapid temperature-swing adsorption-desorption breakthrough cycles.
Simulated air (400 ppm of CO2 balanced in 4/1 N2/O2 with 50% RH) was used in the adsorption process. Adsorption time: 30 min; desorption time: 40 min; desorption temperature: 60 °C.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–42, discussion and Tables 1–7.
Source data
Source Data Figs. 2–5 and Extended Data Fig. 2 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, Z., Ma, T., Zhang, H. et al. Fast and selective CO2 capture from outdoor air by covalent organic frameworks. Nat Sustain 9, 431–438 (2026). https://doi.org/10.1038/s41893-025-01735-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41893-025-01735-1