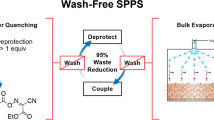
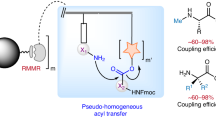
Abstract
Solid-phase peptide synthesis (SPPS) is a primary source of peptides for structure–function studies in development of potential clinical, agricultural, veterinary or cosmetic applications. Despite its efficiency and scalability, SPPS produces unsustainable volumes of toxic waste and non-degradable solid supports. With regulatory changes now in effect to reduce solvent use, there is a pressing need for sustainable, water-based synthesis methods. Due to the aqueous insolubility of the commonly employed Nα-Fmoc-amino acids—the primary building blocks in SPPS—establishing water-based peptide synthesis protocols is extremely challenging. Here we show that amine salts of, for example, N-methylmorpholine or N,N,N-triethanolamine and Nα-Fmoc protected amino acids are water soluble at high concentrations. Activation with a pre-formed water-soluble activating agent enables high quality SPPS on a hydrophilic and biodegradable poly-ε-lysine-based solid support in aqueous solution without characteristic side reactions. The presented methodology provides a simple avenue to sustainable, scalable water-based chemical synthesis of peptides.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within this Article and its Supplementary Information. Crystallographic data for the structures reported in this paper have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers 2402790–2402791. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/structures.
References
Otvos, L. Jr. & Wade, J. D. Current challenges in peptide-based drug discovery. Front. Chem. 2, 62 (2014).
Fosgerau, K. & Hoffmann, T. Peptide therapeutics: current status and future directions. Drug Discov. Today 20, 122–128 (2015).
Henninot, A., Collins, J. C. & Nuss, J. M. The current state of peptide drug discovery: back to the future?. J. Med. Chem. 61, 1382–1414 (2018).
Otvos, L. & Wade, J. D. Big peptide drugs in a small molecule world. Front. Chem. 11, 1302169 (2023).
Jaradat, D. M. M. Thirteen decades of peptide synthesis: key developments in solid phase peptide synthesis and amide bond formation utilized in peptide ligation. Amino Acids 50, 39–68 (2018).
Martin, V. et al. Greening the synthesis of peptide therapeutics: an industrial perspective. RSC Adv. 10, 42457–42492 (2020).
Sherwood, J., Albericio, F. & de la Torre, B. G. N,N-dimethyl formamide European restriction demands solvent substitution in research and development. ChemSusChem 17, e202301639 (2024).
MacMillan, D. S., Murray, J., Sneddon, H. F., Jamieson, C. & Watson, A. J. B. Evaluation of alternative solvents in common amide coupling reactions: replacement of dichloromethane and N-dimethylformamide. Green Chem. 15, 596–600 (2013).
Öhlander, A., Lüdtke, C., Sahakjan, A. & Johnsson, R. E. N-butylpyrrolidinone is an equally good solvent as N,N-dimethylformamide for microwave assisted solid phase peptide synthesis. J. Pept. Sci. 30, e3612 (2024).
Martin, V. et al. Harnessing polarity and viscosity to identify green binary solvent mixtures as viable alternatives to DMF in solid-phase peptide synthesis. Green Chem. 23, 3295–3311 (2021).
Li, W., O’Brien-Simpson, N. M., Hossain, M. A. & Wade, J. D. The 9-fluorenylmethoxycarbonyl (Fmoc) group in chemical peptide synthesis – its past, present and future. Aust. J. Chem. 73, 271–276 (2020).
Wade, J. D., Bedford, J., Sheppard, R. C. & Tregear, G. W. 1,8-Diazabicyclo[5.4.0]undec-7-ene as an Nα-deprotecting reagent in continuous flow solid phase peptide synthesis. Pept. Res. 4, 194–199 (1991).
Mthembu, S. N., Chakraborty, A., Schönleber, R., Albericio, F. & de la Torre, B. G. Morpholine, a strong contender for Fmoc removal in solid-phase peptide synthesis. J. Pept. Sci. 30, e3538 (2024).
Guzmán, F. et al. The tea-bag protocol for comparison of Fmoc removal reagents in solid-phase peptide synthesis. Amino Acids 52, 1201–1205 (2020).
Martelli, G. et al. Replacing piperidine in solid phase peptide synthesis: effective Fmoc removal by alternative bases. Green Chem. 23, 8096–8107 (2021).
Jaradat, D. M. M., Al Musaimi, O. & Albericio, F. Advances in solid-phase peptide synthesis in aqueous media (ASPPS). Green Chem. 24, 6360–6372 (2022).
Hojo, K., Maeda, M. & Kawasaki, K. Solid-phase peptide synthesis in water. Part 3: a water-soluble N-protecting group, 2-[phenyl(methyl)sulfonio]ethoxycarbonyl tetrafluoroborate, and its application to solid phase peptide synthesis in water. Tetrahedron 60, 1875–1886 (2004).
Hojo, K. et al. Peptide synthesis in water IV. Preparation of N-ethanesulfonylethoxycarbonyl (Esc) amino acids and their application to solid phase peptide synthesis. Chem. Pharm. Bull. 52, 422–427 (2004).
Hojo, K., Maeda, M. & Kawasaki, K. 2-(4-Sulfophenylsulfonyl)ethoxycarbonyl group: a new water-soluble N-protecting group and its application to solid phase peptide synthesis in water. Tetrahedron Lett. 45, 9293–9295 (2004).
Knauer, S. et al. Sustainable peptide synthesis enabled by a transient protecting group. Angew. Chem. Int. Ed. 59, 12984–12990 (2020).
Hojo, K. et al. Development of a method for environmentally friendly chemical peptide synthesis in water using water-dispersible amino acid nanoparticles. Chem. Cent. J. 5, 49 (2011).
Henderson, R. K., Hill, A. P., Redman, A. M. & Sneddon, H. F. Development of GSK’s acid and base selection guides. Green Chem. 17, 945–949 (2015).
Eissler, S. et al. Substitution determination of Fmoc-substituted resins at different wavelengths. J. Pept. Sci. 23, 757–762 (2017).
Williams, A. & Ibrahim, I. T. Carbodiimide chemistry: recent advances. Chem. Rev. 81, 589–636 (1981).
Bokhtia, R. M. et al. Efficient synthesis and computational studies of useful guanylating agents: 1H-Benzotriazole-1-carboximidamides. ChemistrySelect 5, 13963 (2020).
Palasek, S. A., Cox, Z. J. & Collins, L. M. Limiting racemization and aspartimide formation in microwave-enhanced Fmoc solid phase peptide synthesis. J. Pept. Sci. 13, 143–148 (2007).
Hojo, K. et al. Aqueous microwave-assisted solid-phase peptide synthesis using Fmoc strategy. III: racemization studies and water-based synthesis of histidine-containing peptides. Amino Acids 46, 2347–2354 (2014).
Meldal, M. Pega: a flow stable polyethylene glycol dimethylacrylamide copolymer for solid phase synthesis. Tetrahedron Lett. 33, 3077–3080 (1992).
Auzanneau, F. I., Meldal, M. & Bock, K. Synthesis, characterization and biocompatibility of PEGA resins. J. Pept. Sci. 1, 31–44 (1995).
Sparrow, J. T., Knieb-Cordonier, N. G., Obeyseskere, N. U. & McMurray, J. S. Large-pore polydimethylacrylamide resin for solid-phase peptide synthesis: applications in Fmoc chemistry. Pept. Res. 9, 297–304 (1996).
Cavalli, G. et al. Novel non-PEG derived polyethers as solid supports. 2. Solid-phase synthesis synthesis. J. Comb. Chem. 9, 1012–1027 (2007).
Akintayo, D. C., de la Torre, B. G., Li, Y. & Albericio, F. Amino-Li-resin - A fiber polyacrylamide resin for solid-phase peptide synthesis. Polymers 14, 928 (2022).
Lawrenson, S., North, M., Peigneguy, F. & Routledge, A. Greener solvents for solid-phase synthesis. Green Chem. 19, 952–962 (2017).
Wellings, D. A. Cross-linked poly-e-lysine particles. UK patent 1812195.4. (31.10.2018).
Beyermann, M., Bienert, M., Niedrich, H., Carpino, L. & Sadat-Aalaee, D. Rapid continuous peptide synthesis via FMOC amino acid chloride coupling and 4-(aminomethyl)piperidine deblocking. J. Org. Chem. 55, 721–728 (1990).
Carpino, L., Sadat-Aalaee, D. & Beyermann, M. Tris(2-aminoethyl)amine as a substitute for 4-(aminomethyl)piperidine in the FMOC/polyamine approach to rapid peptide synthesis. J. Org. Chem. 55, 1673–1675 (1990).
Takahashi, D., Inomata, T. & Fukui, T. AJIPHASE®: a highly efficient synthetic method for one-pot peptide elongation in the solution phase by an Fmoc strategy. Angew. Chem. Int. Ed. 56, 7803–7807 (2017).
Yano, S., Mori, T. & Kubota, H. Silylated tag-assisted peptide synthesis: continuous one-pot elongation for the production of difficult peptides under environmentally friendly conditions. Molecules 26, 3497 (2021).
Leggio, A., Liguori, A., Napoli, A., Siciliano, C. & Sindona, G. New strategies for an efficient removal of the 9-fluorenylmethoxycarbonyl (Fmoc) protecting group in the peptide synthesis. Eur. J. Org. Chem. 2000, 573–575 (2000).
Basavaprabhu, A., Muniyappa, K., Panguluri Rao, N., Veladi, P. & Sureshbabu, V. V. A simple and greener approach for the amide bond formation employing FeCl3 as a catalyst. New J. Chem. 39, 7746–7749 (2015).
Mergler, M., Dick, F., Sax, B., Weller, P. & Vorherr, T. The aspartimide problem in Fmoc-based SPPS. J. Pept. Sci. 9, 36–46 (2003).
Samson, D., Rentsch, D., Minuth, M., Meier, T. & Loidl, G. The aspartimide problem persists: fluorenylmethyloxycarbonyl-solid phase peptide synthesis (Fmoc-SPPS) chain termination due to formation of N-terminal piperazine-2,5-diones. J. Pept. Sci. 24, 33193 (2019).
Yang, Y. in Side Reactions in Peptide Synthesis 22–28 (Academic Press, 2015).
Mueller, L. K., Baumruck, A. C., Zhdanova, H. & Tietze, A. A. Challenges and perspectives in chemical synthesis of highly hydrophobic peptides. Front. Bioeng. Biotechnol. 8, 162 (2020).
Ferrazzano, L. et al. Sustainability in peptide chemistry: current synthesis and purification technologies and future challenges. Green Chem. 24, 975–1020 (2022).
Kekessie, I. et al. Process mass intensity (PMI): a holistic analysis of current peptide manufacturing processes informs sustainability in peptide synthesis. J. Org. Chem. 89, 4261–4281 (2024).
Pawlas, J. & Rasmussen, J. H. Circular aqueous Fmoc/t-Bu solid-phase peptide synthesis. ChemSusChem 14, 32313236 (2021).
Kumar, A., Sharma, A., de la Torre, B. G. & Albericio, F. Rhodiasolv PolarClean—a greener alternative in solid-phase peptide synthesis. Green Chem. Lett. Rev. 14, 545–550 (2021).
Hyde, C., Johnson, T. & Sheppard, R. C. Internal aggregation during solid phase peptide synthesis. Dimethyl sulfoxide as a powerful dissociating solvent. J. Chem. Soc. Chem. Comm. https://doi.org/10.1039/C39920001573 (1992).
Liu, Z., Mohseni, M., Sauvé, S. & Barbeau, B. Segmented regeneration of ion exchange resins used for natural organic matter removal. Sep. Purif. Technol. 303, 122271 (2022).
Groom, C. R., Bruno, I. J., Lightfoot, M. P. & Ward, S. C. The cambridge structural database. Acta Cryst. 17, 171–179 (2016).
Acknowledgements
The electron diffraction experiments were supported by Novo Nordisk Foundation Research Infrastructure grant number NNF220C0074439 (A.L.). M.M. was supported by NNF22SA0081975 and Villum Foundation 00056577. This work was also partly supported by an Australian Research Council Linkage grant (LP180101006) to J.D.W., M.A.H. and D.A.W. Studies at the Florey Institute of Neuroscience and Mental Health were also supported by the Victorian Government’s Operational Infrastructure Support Program. D.A.W., M.M. and J.D.W. gratefully acknowledge R. C. Sheppard (deceased) and E. Atherton, who made seminal contributions to the development of Fmoc-based SPPS and provided formative postdoctoral and professional opportunities that greatly influenced and enabled the authors’ enduring contributions to the field.
Author information
Authors and Affiliations
Contributions
This study was conceived by D.A.W., M.M. and J.D.W., who designed the research and analysed the data. Experiments were prepared and performed by D.A.W., J.G, I.T., C.H., W.L., F.L., M.A.H. and J.D.W. (peptide chemistry), M.M. (peptide chemistry, NMR) and A.L. (crystallography). The manuscript was drafted by D.A.W., A.L., M.M. and J.W.D. All authors contributed to the discussion and finalization of the paper.
Corresponding authors
Ethics declarations
Competing interests
D.A.W. is the Managing Director of SpheriTech Ltd. J.D.W. is an unpaid member of the Scientific Advisory Board of SpheriTech Ltd. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks William Lubell, Beatriz G. de la Torre, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary methods, Figs. 1–11, Tables 1–5 and references.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wellings, D.A., Greenwood, J., Thomas, I. et al. Water-based coupling of amino acids for sustainable solid-phase peptide synthesis. Nat Sustain (2026). https://doi.org/10.1038/s41893-025-01761-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41893-025-01761-z