Abstract
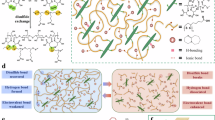
Bio-based adhesives offer inherent advantages over conventional petrochemical-derived systems, including renewable sourcing, reduced environmental impact and potential degradability. However, most bio-based adhesives suffer from poor adhesion strength, limited substrate compatibility and a lack of chemical recyclability. Here we present a bio-derived multiblock poly(ester amide) adhesive that leverages microphase segregation between different segments to reconcile mechanical robustness with strong interfacial bonding. Notably, this multiblock architecture is accessed through a one-pot, selective acceptorless dehydrogenative polymerization, obviating the need for multistep synthesis. The materials exhibit excellent adhesion across a range of substrates including metals, glass and wet wood surpassing commercial benchmarks, while also demonstrating thermal stability, tunable mechanical properties and closed-loop chemical recyclability even in the presence of other commodity plastics. Furthermore, the adhesive strength of these materials could be tuned for various potential applications through control over the chemical composition of the polymer. By integrating renewable feedstocks, high-performance functionality and efficient chemical circularity within a single platform, this work provides a viable pathway toward more sustainable adhesive technologies and contributes to advancing circular materials manufacturing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Full experimental details and the data supporting the findings of this study are available within the article and its Supplementary Information. Source data are provided with this paper.
References
Cui, C. & Liu, W. Recent advances in wet adhesives: adhesion mechanism, design principle and applications. Prog. Polym. Sci. 116, 101388 (2021).
Imam, S. H., Bilbao-Sainz, C., Chiou, B., Glenn, G. M. & Orts, W. J. Biobased adhesives, gums, emulsions, and binders: current trends and future prospects. J. Adhes. Sci. Technol. 27, 1972–1997 (2013).
Petrie, E. M. Handbook of Adhesive and Sealants 2nd edn (McGraw-Hill, 2007).
Heinrich, L. A. Future opportunities for bio-based adhesives – advantages beyond renewability. Green Chem. 21, 1866–1888 (2019).
Adhesives and Sealants Market Size, Share & Trends Analysis Report, 2024–2030 (Grand View Research, accessed 17 June 2025); https://www.grandviewresearch.com/industry-analysis/adhesives-and-sealants-market
Pocius, A. V. Adhesion and Adhesives Technology: An Introduction (Carl Hanser Verlag, 2021).
Engels, T. in Thermosets (ed Guo, Q.) Ch. 8 (Woodhead, 2012).
Pethrick, R. A. Design and ageing of adhesives for structural adhesive bonding - a review. Proc. Inst. Mech. Eng. L 229, 349–379 (2015).
Ma, Y. et al. Research advances in bio-based adhesives. Int. J. Adhes. Adhes. 126, 103444 (2023).
Pereira, J. R. et al. Demonstration of the adhesive properties of the medium-chain-length polyhydroxyalkanoate produced by Pseudomonas chlororaphis subsp. aurantiaca from glycerol. Int. J. Biol. Macromol. 122, 1144–1151 (2019).
Yang, G., Gong, Z., Luo, X., Chen, L. & Shuai, L. Bonding wood with uncondensed lignins as adhesives. Nature 621, 511–515 (2023).
Lv, Z. et al. Bio-based hot-melt adhesive from xylan. Nat. Sustain. 8, 827–836 (2025).
Zhou, Y. et al. High strength and flame retardant soybean polysaccharide-based wood adhesive produced by borate chemistry and crosslinking strategy. Eur. Polym. J. 164, 110973 (2022).
Zhang, Z. et al. Stereomicrostructure-regulated biodegradable adhesives. Science 387, 297–303 (2025).
Westerman, C. R., McGill, B. C. & Wilker, J. J. Sustainably sourced components to generate high-strength adhesives. Nature 621, 306–311 (2023).
Zhao, Y. et al. Chemically recyclable polyolefin-like multiblock polymers. Science 382, 310–314 (2023).
Liu, X. et al. Catalytic closed-loop recycling of polyethylene-like materials produced by acceptorless dehydrogenative polymerization of bio-derived diols. Nat. Chem. 17, 500–506 (2025).
Zhao, Y. et al. Tailoring the properties of chemically recyclable polyethylene-like multiblock polymers by modulating the branch structure. Angew. Chem. Int. Ed. 137, e202415707 (2025).
Han, S. & Wu, J. Recent advances of poly(ester amide)s-based biomaterials. Biomacromolecules 23, 1892–1919 (2022).
Rodriguez-Galan, A., Franco, L. & Puiggali, J. Degradable poly(ester amide)s for biomedical applications. Polymers 3, 65–99 (2011).
Winnacker, M. & Rieger, B. Poly(ester amide)s: recent insights into synthesis, stability and biomedical applications. Polym. Chem. 7, 7039–7046 (2016).
He, J., Ding, Y., Jiang, F. & Wang, Z. Poly(ester amide)s derived from low-value plant oil as reusable low-temperature tolerant adhesive. Eur. Polym. J. 198, 112387 (2023).
Yoshinaka, Y. & Miller, S. A. Bio-oil derived polyesteramides as water-degradable replacements for polyethylene. Green Chem. 27, 4152–4164 (2025).
Zeng, H. & Guan, Z. Direct synthesis of polyamides via catalytic dehydrogenation of diols and diamines. J. Am. Chem. Soc. 133, 1159–1161 (2011).
Filonenko, G. A., Putten, R., Hensen, E. J. M. & Pidko, E. A. Catalytic (de)hydrogenation promoted by non-precious metals – Co, Fe and Mn: recent advances in an emerging field. Chem. Soc. Rev. 47, 1459–1483 (2018).
Gunanathan, C. & Milstein, D. Bond activation and catalysis by ruthenium pincer complexes. Chem. Rev. 114, 12024–12087 (2014).
Li, H., Wang, X., Huang, F., Jiang, J. & Wang, Z. Computational study on the catalytic role of pincer ruthenium (II)-PNN complex in directly synthesizing amide from alcohol and amine: the origin of selectivity of amide over ester and imine. Organometallics 30, 5233–5247 (2011).
Beyer, V. P., Kim, J. & Becer, C. R. Synthetic approaches for multiblock copolymers. Polym. Chem. 11, 1271–1291 (2020).
Dau, H. et al. Linear block copolymer synthesis. Chem. Rev. 122, 14471–14553 (2022).
Pingen, D. et al. Diamines for polymer materials via direct amination of lipid- and lignocellulose-based alcohols with NH3. ChemCatChem 10, 3027–3033 (2018).
Tuominen, J. & Seppälä, J. V. Synthesis and characterization of lactic acid based poly(ester−amide). Macromolecules 33, 3530–3535 (2000).
Regaño, C., Alla, A., Ilarduya, A. M. & Muñoz-Guerra, S. Poly(ester amide)s derived from L-malic acid. Macromolecules 37, 2067–2075 (2004).
Mahadas, N. A. et al. High molecular weight biobased long-chain aliphatic polyesters with degradability: insights into mimicking polyethylene. Macromolecules 58, 4070–4081 (2025).
Wolfgang, G. & Ruestem, D. Nylon-12-preparation, properties, and applications. Ind. Eng. Chem. 62, 16–22 (1970).
Luckachan, E. G. & Pillai, C. K. S. Random multiblock poly(ester amide)s containing poly(L-lactide) and cycloaliphatic amide segments: synthesis and biodegradation studies. J. Polym. Sci. A Polym. Chem. 44, 3250–3260 (2006).
Michell, R. M. et al. Effect of sequence distribution on the morphology, crystallization, melting, and biodegradation of poly(ε-caprolactone-co-ε-caprolactam) copolymers. Macromolecules 42, 6671–6681 (2009).
Hamley, I. W. et al. Crystallization in poly(l-lactide)-b-poly(ε-caprolactone) double crystalline diblock copolymers: a study using X-ray scattering, differential scanning calorimetry, and polarized optical microscopy. Macromolecules 38, 463–472 (2005).
Marques, E. A. S., da Silva, L. F. M., Banea, M. D. & Carbas, R. J. C. Adhesive joints for low- and high-temperature use: an overview. J. Adhes. 91, 556–585 (2015).
Viana, G., Costa, M., Banea, M. D. & da silva, L. F. M. A review on the temperature and moisture degradation of adhesively bonded joints. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 231, 488–501 (2017).
Ellis, L. D. et al. Chemical and biological catalysis for plastics recycling and upcycling. Nat. Catal. 4, 539–556 (2021).
Khopade, K. V., Chikkali, S. H. & Barsu, N. Metal-catalyzed plastic depolymerization. Cell Rep Phys Sci 4, 101341 (2023).
Kumar, A. et al. Hydrogenative depolymerization of nylons. J. Am. Chem. Soc. 142, 14267–14275 (2020).
Acknowledgements
This work was supported by the National Institutes of Health under grant no. R35GM144356 and RePLACE (Redesigning Polymers to Leverage a Circular Economy) funded by the Office of Science of the US Department of Energy through grant no. DE-SC0022290. The authors thank the Analytical Resources Core (RRID: SCR_021759) at Colorado State University for instrument access. G.M.M. acknowledges support from the Dr. Robert Williams Professorship in Organic Chemistry at Colorado State University. Funding for J.M. and N.A.R. was provided by the US Department of Energy, Office of Energy Efficiency and Renewable Energy, Advanced Materials and Manufacturing Technologies Office (AMMTO) and Bioenergy Technologies Office (BETO) as part of the BOTTLE Consortium, supported by AMMTO and BETO under contract no. DE-AC36-08GO28308 with the National Renewable Energy Laboratory, operated by Alliance for Sustainable Energy, LLC.
Author information
Authors and Affiliations
Contributions
X.L. and G.M.M. conceived the idea. X.L. designed and conducted experiments and analysed results. X.L., Y.Z., K.L.H., E.M.R., J.M. and N.A.R. performed characterization and analysed results. X.L. wrote the initial manuscript and supplemental materials. All authors read and edited the manuscript. G.M.M. directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Feng Peng and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–29, Tables 1–44 and Discussion.
Source data
Source Data Fig. 2 (download XLSX )
Raw data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Raw data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Raw data for Fig. 4.
Source Data Table 1 (download XLSX )
Raw data for Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Harry, K.L., Zhao, Y. et al. Strong and recyclable bio-derived poly(ester amide) hot-melt adhesive. Nat Sustain 9, 450–458 (2026). https://doi.org/10.1038/s41893-026-01776-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41893-026-01776-0