Abstract
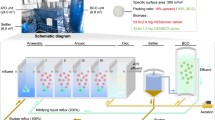
Biological nitrogen removal is a main contributor to the carbon footprint of municipal wastewater treatment plants (MWWTPs). Anaerobic ammonium oxidation (anammox) is key to decarbonizing MWWTPs. However, anammox technology has long been developed following the ‘hotspot’ concept, which spatially confines anammox to specific zones in MWWTPs, leaving its potential underexploited in mainstream treatment. Here we propose a ‘multi-zonal anammox’ concept to expand the anammox process throughout the whole MWWTP mainstream. It allows the implementation of anammox in existing MWWTPs with minimal reconstruction requirements. We evaluated the feasibility of this concept in an anaerobic–anoxic–oxic (A2O) biosystem, which is one of the most widely used processes in MWWTPs. During a 2-year-long operation, anammox bacteria highly self-enriched and contributed to nitrogen removal throughout the mainstream, with relative abundances reaching 4.8%, 5.7–10.0% and 0.2–1.0% in anaerobic, anoxic and oxic zones, respectively. The multi-zonal anammox enhanced nitrogen removal efficiency by 20%, reduced aerating electricity input by 20%, mitigated nitrous oxide emissions by 66% and eliminated the need for supplemental organic carbon. Overall, this work opens a promising avenue for implementing the anammox process, paving the way towards sustainable wastewater management.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Raw sequencing data are available in the NCBI Sequence Read Archive (SRA) with the accession codes PRJNA1178615 and PRJNA1355498. Source data are provided with this paper.
References
Sachs, J. D. et al. Six Transformations to achieve the Sustainable Development Goals. Nat. Sustain. 2, 805–814 (2019).
Schwabe, K., Nemati, M., Amin, R., Tran, Q. & Jassby, D. Unintended consequences of water conservation on the use of treated municipal wastewater. Nat. Sustain. 3, 628–635 (2020).
Tortajada, C. Reused water as a source of clean water and energy. Nat. Water 2, 102–103 (2024).
Winkler, M. K. & Straka, L. New directions in biological nitrogen removal and recovery from wastewater. Curr. Opin. Biotechnol. 57, 50–55 (2019).
McCarty, P. L. What is the best biological process for nitrogen removal: when and why?. Environ. Sci. Technol. 52, 3835–3841 (2018).
Du, W. et al. Spatiotemporal pattern of greenhouse gas emissions in China’s wastewater sector and pathways towards carbon neutrality. Nat. Water 1, 166–175 (2023).
Lotti, T. et al. Anammox growth on pretreated municipal wastewater. Environ. Sci. Technol. 48, 7874–7880 (2014).
Kartal, B., Kuenen, J. G. & van Loosdrecht, M. C. M. Sewage treatment with anammox. Science 328, 702–703 (2010).
Kuenen, J. G. Anammox bacteria: from discovery to application. Nat. Rev. Microbiol. 6, 320–326 (2008).
Ponce-Jahen, S. J., Cercado, B., Estrada-Arriaga, E. B., Rangel-Mendez, J. R. & Cervantes, F. J. Anammox with alternative electron acceptors: perspectives for nitrogen removal from wastewaters. Biodegradation 35, 47–70 (2024).
Dalsgaard, T., Canfield, D. E., Petersen, J., Thamdrup, B. & Acuña-González, J. N2 production by the anammox reaction in the anoxic water column of Golfo Dulce, Costa Rica. Nature 422, 606–608 (2003).
Prokopenko, M. G. et al. Nitrogen losses in anoxic marine sediments driven by Thioploca–anammox bacterial consortia. Nature 500, 194–198 (2013).
Joss, A. et al. Full-scale nitrogen removal from digester liquid with partial nitritation and anammox in one SBR. Environ. Sci. Technol. 43, 5301–5306 (2009).
Zhao, Q. et al. Pilot-scale implementation of mainstream anammox for municipal wastewater treatment against cold temperature. Nat. Commun. 15, 10314 (2024).
Zhao, Q. et al. Carbon-restricted anoxic zone as an overlooked anammox hotspot in municipal wastewater treatment plants. Environ. Sci. Technol. 57, 21767–21778 (2023).
Laureni, M. et al. Activity and growth of anammox biomass on aerobically pre-treated municipal wastewater. Water Res. 80, 325–336 (2015).
Lackner, S. et al. Full-scale partial nitritation/anammox experiences—an application survey. Water Res. 55, 292–303 (2014).
Joss, A. et al. Combined nitritation–anammox: advances in understanding process stability. Environ. Sci. Technol. 45, 9735–9742 (2011).
Cao, Y. et al. The influence of dissolved oxygen on partial nitritation/anammox performance and microbial community of the 200,000 m3/d activated sludge process at the Changi water reclamation plant (2011 to 2016). Water Sci. Technol. 78, 634–643 (2018).
Nsenga Kumwimba, M., Lotti, T., Şenel, E., Li, X. & Suanon, F. Anammox-based processes: how far have we come and what work remains? A review by bibliometric analysis. Chemosphere 238, 124627 (2020).
Cervantes, F., Monroy, O. & Gomez, J. Influence of ammonium on the performance of a denitrifying culture under heterotrophic conditions. Appl. Biochem. Biotechnol. 81, 13–21 (1999).
Cervantes, F. J., De la Rosa, D. A. & Gómez, J. Nitrogen removal from wastewaters at low C/N ratios with ammonium and acetate as electron donors. Bioresour. Technol. 79, 165–170 (2001).
Du, R. et al. Partial denitrification providing nitrite: opportunities of extending application for anammox. Environ. Int. 131, 105001 (2019).
Li, J. et al. Quantify the contribution of anammox for enhanced nitrogen removal through metagenomic analysis and mass balance in an anoxic moving bed biofilm reactor. Water Res. 160, 178–187 (2019).
Nejidat, A. et al. Abundance and diversity of anammox bacteria in a mainstream municipal wastewater treatment plant. Appl. Microbiol. Biotechnol. 102, 6713–6723 (2018).
Anja, K. & Ottosen, L. D. M. Anammox and partial nitritation in the mainstream of a wastewater treatment plant in a temperate region (Denmark). Water Sci. Technol. 79, 1397–1405 (2019).
Park, H., Brotto, A. C., van Loosdrecht, M. C. M. & Chandran, K. Discovery and metagenomic analysis of an anammox bacterial enrichment related to Candidatus ‘Brocadia caroliniensis’ in a full-scale glycerol-fed nitritation-denitritation separate centrate treatment process. Water Res. 111, 265–273 (2017).
James, S. N. & Vijayanandan, A. Recent advances in simultaneous nitrification and denitrification for nitrogen and micropollutant removal: a review. Biodegradation 34, 103–123 (2023).
Roothans, N. et al. Long-term multi-meta-omics resolves the ecophysiological controls of seasonal N2O emissions during wastewater treatment. Nat. Water 3, 590–604 (2025).
Song, C. et al. Oversimplification and misestimation of nitrous oxide emissions from wastewater treatment plants. Nat. Sustain. 7, 1348–1358 (2024).
Lawson, C. E. et al. Metabolic network analysis reveals microbial community interactions in anammox granules. Nat. Commun. 8, 15416 (2017).
Speth, D. R., In T Zandt, M. H., Guerrero-Cruz, S., Dutilh, B. E. & Jetten, M. S. M. Genome-based microbial ecology of anammox granules in a full-scale wastewater treatment system. Nat. Commun. 7, 11172 (2016).
Huelsmann, M. & Ackermann, M. Community instability in the microbial world. Science 378, 29–30 (2022).
Okabe, S., Shafdar, A. A., Kobayashi, K., Zhang, L. & Oshiki, M. Glycogen metabolism of the anammox bacterium ‘Candidatus Brocadia sinica’. ISME J. 15, 1287–1301 (2021).
Puyol, D., Carvajal-Arroyo, J. M., Garcia, B., Sierra-Alvarez, R. & Field, J. A. Kinetic characterization of Brocadia spp.-dominated anammox cultures. Bioresour. Technol. 139, 94–100 (2013).
Ahn, Y. Sustainable nitrogen elimination biotechnologies: a review. Process Biochem. 41, 1709–1721 (2006).
Strous, M. et al. Deciphering the evolution and metabolism of an anammox bacterium from a community genome. Nature 440, 790–794 (2006).
Trojanowicz, K., Trela, J. & Plaza, E. Possible mechanism of efficient mainstream partial nitritation/anammox (PN/A) in hybrid bioreactors (IFAS). Environ. Technol. 42, 1023–1037 (2021).
Wade, M. J. & Wolkowicz, G. S. K. Bifurcation analysis of an impulsive system describing partial nitritation and anammox in a hybrid reactor. Environ. Sci. Technol. 55, 2099–2109 (2021).
Zhao, Q. et al. From hybrid process to pure biofilm anammox process: suspended sludge biomass management contributing to high-level anammox enrichment in biofilms. Water Res. 236, 119959 (2023).
Alvarado, V., Ying, L., Asghari, V., Hsu, S. & Lee, P. Modeling of a mainstream partial nitrification/anammox process through a hybrid theoretical-machine learning approach. ACS EST Water 5, 1469–1480 (2025).
Liu, Q. et al. Mechanism of suspended sludge impact on anammox enrichment in anoxic biofilm through long term operation and microbial analysis. Water Res. 229, 119412 (2023).
Philipp, L., Bühler, K., Ulber, R. & Gescher, J. Beneficial applications of biofilms. Nat. Rev. Microbiol. 22, 276–290 (2024).
Chen, S. et al. Decoupling wastewater-related greenhouse gas emissions and water stress alleviation across 300 cities in China is challenging yet plausible by 2030. Nat. Water 1, 534–546 (2023).
Christou, A. et al. Sustainable wastewater reuse for agriculture. Nat. Rev. Earth Environ. 5, 504–521 (2024).
Zhang, S. et al. Full-scale mainstream partial nitritation/anammox process: a three-year demonstration for municipal wastewater treatment. Water Res. X 29, 100419 (2025).
Li, Z., Wu, Y., Chen, T., Yan, B. & Wei, C. Deciphering organic substrate impacts in anammox systems: a machine learning driven framework for predictive classification and process mechanism analysis. Environ. Int. 202, 11 (2025).
Song, C., Zhu, J., Yuan, Z., van Loosdrecht, M. C. M. & Ren, Z. J. Defining and achieving net-zero emissions in the wastewater sector. Nat. Water 2, 927–935 (2024).
Cao, S. et al. Toward mainstream anammox by integrating sidestream treatment. Environ. Sci. Technol. 56, 10553–10556 (2022).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (grant nos. 52131004, Y.P.; U23A20675, L.Z. and Y.P.), the Higher Education Discipline Innovation Project (111 Project, D16003, Y.P.), the Funding Projects of Beijing Municipal Commission of Education (Y.P.), the Postdoctoral Fellowship Program and China Postdoctoral Science Foundation (grant nos. BX20250323, Q.Z.; 2025M781157, Q.Z.) and the Shanghai Tongji Gaotingyao Environmental Science & Technology Development Foundation (Q.Z.).
Author information
Authors and Affiliations
Contributions
The project was conceived and led by Y.P. Q.Z., L.Z., Y.Z., J.L., L.W. and X.L. contributed to the chemical, microbial and statistical analyses. Q.Z. wrote the manuscript, and Y.P. and L.Z. contributed to its commenting and revision. All authors discussed and interpreted the results and contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Fangang Meng and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–14, Tables 1–5 and Supplementary Figs. 1–8.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 1 (download ZIP )
Please note the source data for the Supplementary Information have been uploaded as a zipped folder, which includes eight separate Excel files corresponding to eight supplementary figures. The zipped folder is named ‘Source_Data_for_Supplementary _Information.zip’.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Q., Zhang, L., Zhao, Y. et al. Multi-zonal anaerobic ammonium oxidation for mainstream municipal wastewater treatment. Nat Sustain (2026). https://doi.org/10.1038/s41893-026-01786-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41893-026-01786-y