Abstract
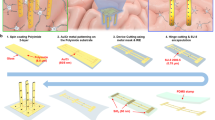
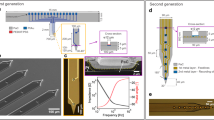
Cognition and behaviour rely on coordinated activity from neural circuits distributed across three dimensions. However, typical probes for recording neural activity in the brain are limited to two-dimensional interfacing due to the planar semiconductor fabrication process. Here we report a rolling-of-soft-electronics approach to create monolithic three-dimensional (3D) neural probes with high scalability and design flexibility. Compared with previous stacking or assembly methods, the approach directly transforms a planar device into a 3D probe by leveraging the softness of flexible electrodes. The electrode shanks are initially fabricated in a single plane and then connected to a flexible spacer. By varying the features of planar design, such as shank pitch and spacer layer thickness, the device can then be deterministically rolled into versatile 3D probe designs containing hundreds of electrodes. With the system, we demonstrate single-unit spike recording in vivo in rodent and non-human primate models. We also show that the probe can provide microscopy-like 3D spatiotemporal mapping of spike activities in the rodent visual cortex, with five-week-long recording stability and promising 3D decoding performance of visual orientation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Animal electrophysiology data that support the findings of this study are available via Zenodo at https://doi.org/10.5281/zenodo.15498935 (ref. 81). All other datasets generated during and/or analysed during the study are available from the corresponding authors upon reasonable request. Source data are provided with this paper.
Code availability
The custom Python scripts used to analyse the neural signals and visualize the spike activities are available via GitHub at https://github.com/qiangy0819/ROSE_process.git. Code for extended analyses is available upon reasonable request.
References
Lynn, C. W. & Bassett, D. S. The physics of brain network structure, function and control. Nat. Rev. Phys. 1, 318–332 (2019).
Hong, G. & Lieber, C. M. Novel electrode technologies for neural recordings. Nat. Rev. Neurosci. 20, 330–345 (2019).
Chung, J. E. et al. High-density, long-lasting, and multi-region electrophysiological recordings using polymer electrode arrays. Neuron 101, 21–31.e25 (2019).
Park, Y. et al. Three-dimensional, multifunctional neural interfaces for cortical spheroids and engineered assembloids. Sci. Adv. 7, eabf9153 (2021).
Liu, L. D. et al. Accurate localization of linear probe electrode arrays across multiple brains. eNeuro 8, ENEURO.0241-21.2021 (2021).
Cabrera, L., Sadle, C. & Purcell, E. Neuroethical considerations of high-density electrode arrays. Nat. Biomed. Eng. 3, 586–589 (2019).
Maynard, E. M., Nordhausen, C. T. & Normann, R. A. The Utah intracortical electrode array: a recording structure for potential brain-computer interfaces. Electroencephalogr. Clin. Neurophysiol. 102, 228–239 (1997).
Hoogerwerf, A. C. & Wise, K. D. A three-dimensional microelectrode array for chronic neural recording. IEEE Trans. Biomed. Eng. 41, 1136–1146 (1994).
Bai, Q., Wise, K. D. & Anderson, D. J. A high-yield microassembly structure for three-dimensional microelectrode arrays. IEEE Trans. Biomed. Eng. 47, 281–289 (2000).
Yao, Y., Gulari, M. N., Wiler, J. A. & Wise, K. D. A microassembled low-profile three-dimensional microelectrode array for neural prosthesis applications. J. Microelectromech. Syst. 16, 977–988 (2007).
Du, J., Blanche, T. J., Harrison, R. R., Lester, H. A. & Masmanidis, S. C. Multiplexed, high density electrophysiology with nanofabricated neural probes. PLoS ONE 6, e26204 (2011).
Merriam, S. M. E., Srivannavit, O., Gulari, M. N. & Wise, K. D. A three-dimensional 64-site folded electrode array using planar fabrication. J. Microelectromech. Syst. 20, 594–600 (2011).
John, J., Li, Y., Zhang, J., Loeb, J. A. & Xu, Y. Microfabrication of 3D neural probes with combined electrical and chemical interfaces. J. Micromech. Microeng. 21, 105011 (2011).
Shobe, J. L., Claar, L. D., Parhami, S., Bakhurin, K. I. & Masmanidis, S. C. Brain activity mapping at multiple scales with silicon microprobes containing 1,024 electrodes. J. Neurophysiol. 114, 2043–2052 (2015).
Rios, G., Lubenov, E. V., Chi, D., Roukes, M. L. & Siapas, A. G. Nanofabricated neural probes for dense 3-D recordings of brain activity. Nano Lett. 16, 6857–6862 (2016).
Barz, F. et al. Versatile, modular 3D microelectrode arrays for neuronal ensemble recordings: from design to fabrication, assembly, and functional validation in non-human primates. J. Neural Eng. 14, 036010 (2017).
Wang, X. et al. Freestanding 3D mesostructures, functional devices, and shape-programmable systems based on mechanically induced assembly with shape memory polymers. Adv. Mater. 31, 1805615 (2019).
Yan, Z. et al. Three-dimensional mesostructures as high-temperature growth templates, electronic cellular scaffolds, and self-propelled microrobots. Proc. Natl Acad. Sci. USA 114, E9455–E9464 (2017).
Zhao, Z. et al. Ultraflexible electrode arrays for months-long high-density electrophysiological mapping of thousands of neurons in rodents. Nat. Biomed. Eng. 7, 520–532 (2023).
Musk, E. An integrated brain-machine interface platform with thousands of channels. J. Med. Internet Res. 21, e16194 (2019).
Obaid, A. et al. Massively parallel microwire arrays integrated with CMOS chips for neural recording. Sci. Adv. 6, eaay2789 (2020).
Wang, P. et al. Direct-print three-dimensional electrodes for large-scale, high-density, and customizable neural interfaces. Preprint at bioRxiv https://doi.org/10.1101/2023.05.30.542925 (2023).
Polikov, V. S., Tresco, P. A. & Reichert, W. M. Response of brain tissue to chronically implanted neural electrodes. J. Neurosci. Methods 148, 1–18 (2005).
Chen, R., Canales, A. & Anikeeva, P. Neural recording and modulation technologies. Nat. Rev. Mater. 2, 16093 (2017).
Sharafkhani, N. et al. Neural tissue-microelectrode interaction: brain micromotion, electrical impedance, and flexible microelectrode insertion. J. Neurosci. Methods 365, 109388 (2022).
Lacour, S. P., Wagner, S., Huang, Z. & Suo, Z. Stretchable gold conductors on elastomeric substrates. Appl. Phys. Lett. 82, 2404–2406 (2003).
Rogers, J. A., Someya, T. & Huang, Y. Materials and mechanics for stretchable electronics. Science 327, 1603–1607 (2010).
Singh, S. et al. Modeling the insertion mechanics of flexible neural probes coated with sacrificial polymers for optimizing probe design. Sensors 16, 330 (2016).
Yang, C., Xie, Y., Liu, S. & Sun, D. Force modeling, identification, and feedback control of robot-assisted needle insertion: a survey of the literature. Sensors 18, 561 (2018).
Pomfret, R., Miranpuri, G. & Sillay, K. The substitute brain and the potential of the gel model. Ann. Neurosci. 20, 118–122 (2013).
Lecomte, A., Descamps, E. & Bergaud, C. A review on mechanical considerations for chronically-implanted neural probes. J. Neural Eng. 15, 031001 (2018).
Casanova, F., Carney, P. R. & Sarntinoranont, M. In vivo evaluation of needle force and friction stress during insertion at varying insertion speed into the brain. J. Neurosci. Methods 237, 79–89 (2014).
Obaid, A. et al. Ultra-sensitive measurement of brain penetration mechanics and blood vessel rupture with microscale probes. Preprint at bioRxiv https://doi.org/10.1101/2020.09.21.306498 (2020).
Le Floch, P. et al. 3D spatiotemporally scalable in vivo neural probes based on fluorinated elastomers. Nat. Nanotechnol. 19, 319–329 (2024).
Zhao, S. et al. Tracking neural activity from the same cells during the entire adult life of mice. Nat. Neurosci. 26, 696–710 (2023).
Seo, K. J. et al. A soft, high-density neuroelectronic array. NPJ Flex. Electron. 7, 40 (2023).
Takeuchi, S., Ziegler, D., Yoshida, Y., Mabuchi, K. & Suzuki, T. Parylene flexible neural probes integrated with microfluidic channels. Lab Chip 5, 519–523 (2005).
Kil, D. et al. Dextran as a resorbable coating material for flexible neural probes. Micromachines 10, 61 (2019).
Cointe, C. et al. Scalable batch fabrication of ultrathin flexible neural probes using a bioresorbable silk layer. Microsyst. Nanoeng. 8, 21 (2022).
Kozai, T. D. et al. Comprehensive chronic laminar single-unit, multi-unit, and local field potential recording performance with planar single shank electrode arrays. J. Neurosci. Methods 242, 15–40 (2015).
Oke, O. O. et al. High‐frequency gamma oscillations coexist with low-frequency gamma oscillations in the rat visual cortex in vitro. Eur. J. Neurosci. 31, 1435–1445 (2010).
Cody, P. A., Eles, J. R., Lagenaur, C. F., Kozai, T. D. & Cui, X. T. Unique electrophysiological and impedance signatures between encapsulation types: an analysis of biological Utah array failure and benefit of a biomimetic coating in a rat model. Biomaterials 161, 117–128 (2018).
Black, B. J. et al. Chronic recording and electrochemical performance of Utah microelectrode arrays implanted in rat motor cortex. J. Neurophysiol. 120, 2083–2090 (2018).
Nordhausen, C. T., Maynard, E. M. & Normann, R. A. Single unit recording capabilities of a 100 microelectrode array. Brain Res. 726, 129–140 (1996).
Sahasrabuddhe, K. et al. The Argo: a high channel count recording system for neural recording in vivo. J. Neural Eng. 18, 015002 (2021).
Deku, F. et al. Amorphous silicon carbide ultramicroelectrode arrays for neural stimulation and recording. J. Neural Eng. 15, 016007 (2018).
Zhao, Z. et al. Parallel, minimally-invasive implantation of ultra-flexible neural electrode arrays. J. Neural Eng. 16, 035001 (2019).
Fiáth, R. et al. A silicon-based neural probe with densely-packed low-impedance titanium nitride microelectrodes for ultrahigh-resolution in vivo recordings. Biosens. Bioelectron. 106, 86–92 (2018).
Raducanu, B. C. et al. Time multiplexed active neural probe with 1,356 parallel recording sites. Sensors 17, 2388 (2017).
Fang, H. et al. Capacitively coupled arrays of multiplexed flexible silicon transistors for long-term cardiac electrophysiology. Nat. Biomed. Eng. 1, 0038 (2017).
Xu, W., Huang, X., Takagaki, K. & Wu, J.-Y. Compression and reflection of visually evoked cortical waves. Neuron 55, 119–129 (2007).
Apollo, N. V. et al. Gels, jets, mosquitoes, and magnets: a review of implantation strategies for soft neural probes. J. Neural Eng. 17, 041002 (2020).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates: Hard Cover Edition (Elsevier, 2006).
Liu, X. et al. Stability of the interface between neural tissue and chronically implanted intracortical microelectrodes. IEEE Trans. Rehabil. Eng. 7, 315–326 (1999).
McConnell, G. C. et al. Implanted neural electrodes cause chronic, local inflammation that is correlated with local neurodegeneration. J. Neural Eng. 6, 056003 (2009).
Kozai, T. D., Jaquins-Gerstl, A. S., Vazquez, A. L., Michael, A. C. & Cui, X. T. Brain tissue responses to neural implants impact signal sensitivity and intervention strategies. ACS Chem. Neurosci. 6, 48–67 (2015).
Franch, M. et al. Visuo-frontal interactions during social learning in freely moving macaques. Nature 627, 174–181 (2024).
Schoonover, C. E., Ohashi, S. N., Axel, R. & Fink, A. J. Representational drift in primary olfactory cortex. Nature 594, 541–546 (2021).
Gallego, J. A., Perich, M. G., Chowdhury, R. H., Solla, S. A. & Miller, L. E. Long-term stability of cortical population dynamics underlying consistent behavior. Nat. Neurosci. 23, 260–270 (2020).
Wagner, M. J. et al. A neural circuit state change underlying skilled movements. Cell 184, 3731–3747.e21 (2021).
Sponheim, C. et al. Longevity and reliability of chronic unit recordings using the Utah, intracortical multi-electrode arrays. J. Neural Eng. 18, 066044 (2021).
Ghodrati, M., Alwis, D. S. & Price, N. S. Orientation selectivity in rat primary visual cortex emerges earlier with low‐contrast and high‐luminance stimuli. Eur. J. Neurosci. 44, 2759–2773 (2016).
Girman, S. V., Sauvé, Y. & Lund, R. D. Receptive field properties of single neurons in rat primary visual cortex. J. Neurophysiol. 82, 301–311 (1999).
Juavinett, A. L. & Callaway, E. M. Pattern and component motion responses in mouse visual cortical areas. Curr. Biol. 25, 1759–1764 (2015).
Ohki, K. & Reid, R. C. Specificity and randomness in the visual cortex. Curr. Opin. Neurobiol. 17, 401–407 (2007).
White, L. E. & Fitzpatrick, D. Vision and cortical map development. Neuron 56, 327–338 (2007).
Fahey, P. G. et al. A global map of orientation tuning in mouse visual cortex. Preprint at bioRxiv https://doi.org/10.1101/745323 (2019).
Stringer, C., Michaelos, M., Tsyboulski, D., Lindo, S. E. & Pachitariu, M. High-precision coding in visual cortex. Cell 184, 2767–2778.e15 (2021).
Kamitani, Y. & Tong, F. Decoding the visual and subjective contents of the human brain. Nat. Neurosci. 8, 679–685 (2005).
Schneider, S., Lee, J. H. & Mathis, M. W. Learnable latent embeddings for joint behavioural and neural analysis. Nature 617, 360–368 (2023).
Fiscella, M. et al. Visual coding with a population of direction-selective neurons. J. Neurophysiol. 114, 2485–2499 (2015).
Liu, J. in Biomimetics Through Nanoelectronics: Development of Three Dimensional Macroporous Nanoelectronics for Building Smart Materials, Cyborg Tissues and Injectable Biomedical Electronics Ch. 5 (Springer, 2018).
Rousche, P. J. et al. Flexible polyimide-based intracortical electrode arrays with bioactive capability. IEEE Trans. Biomed. Eng. 48, 361–371 (2001).
Kim, B. J. et al. 3D parylene sheath neural probe for chronic recordings. J. Neural Eng. 10, 045002 (2013).
Byun, D., Cho, S. J. & Kim, S. Fabrication of a flexible penetrating microelectrode array for use on curved surfaces of neural tissues. J. Micromech. Microeng. 23, 125010 (2013).
Seo, K. J. et al. Transparent, flexible, penetrating microelectrode arrays with capabilities of single-unit electrophysiology. Adv. Biosyst. 3, 1800276 (2019).
Wang, X. et al. A parylene neural probe array for multi-region deep brain recordings. J. Microelectromech. Syst. 29, 499–513 (2020).
Shin, Y. et al. Array-wide uniform PEDOT: PSS electroplating from potentiostatic deposition. Biosens. Bioelectron. 261, 116418 (2024).
Woeppel, K., Dhawan, V., Shi, D. & Cui, X. T. Nanotopography-enhanced biomimetic coating maintains bioactivity after weeks of dry storage and improves chronic neural recording. Biomaterials 302, 122326 (2023).
Chung, J. E. et al. A fully automated approach to spike sorting. Neuron 95, 1381–1394.e6 (2017).
Qiang, Y. 3D electrophysiology data in rodents and NHP brain. Zenodo https://doi.org/10.5281/zenodo.15498935 (2025).
Acknowledgements
We acknowledge support from NIH award nos. R21EY030710 and U01NS123668, NSF award no. 2347978 and funds from Dartmouth College. S. Wang acknowledges support from the NSF CAREER award no. CMMI-1847062 and the Oklahoma Center for Advancement of Science & Technology grant no. HR18-085. X.T.C. acknowledges NINDS R01NS136622, R01 NS102725 and U01 NS113279. C.C. is supported by an NIH/NINDS K99/R00 NS092972, NIH/NINDS R01 NS122969, the Brain and Behavior Research Foundation, the Moorman-Simon Interdisciplinary Career Development Professorship and the Whitehall Foundation. T.L.M. acknowledges financial support from NIH/NIA R01 AG068168 and NIH/NINDS R56 NS112207.
Author information
Authors and Affiliations
Contributions
H.F., S. Wang and X.T.C. supervised this work. W.G., K.J.S., Y.Q. and H.F. conceived the original concept of ROSE. W.G. and K.J.S. conducted the preliminary device optimization, fabrication and ROSE assembly. Y.Q. and T.B. designed and prototyped the MagMatrix board. Y.S., Y.Q. and J.R. further optimized the device design and fabrication for in vivo experiments. W.G., Y.Q., Y.S., G.L. and D.J. performed the device characterizations, mathematical modelling (offsets) and insertion dynamics testing. Y.Q., D.J., D.S. and W.G. performed the in vivo electrophysiology. Y.Q. and D.J. conducted the neural data processing and decoding analysis. D.J., D.S., S. Wu, A.I., J.-Y.L., D.L.R., M.M., T.L.M., A.N.K., R.K., P.A. and C.C. performed the animal surgeries and experiments. D.S. and V.D. performed the histological studies through immunohistochemistry and confocal fluorescence imaging. S.V., G.L., W.G. and S. Wang performed the structural and mechanical modelling of the ROSE shanks, including insertion force dynamics (S.V. and S. Wang), micromotion and twisting analysis (G.L.), and maximum principal strain (W.G.). Y.Q., W.G., D.S., K.J.S., D.J., Y.S., S. Wang and H.F. co‑wrote the manuscript. Y.Q., W.G. and D.S. prepared the figures. W.G. led the research in the original submission of the manuscript, and Y.Q. led the revision work. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
H.F., W.G., Y.Q. and K.J.S. are inventors on patent application US 19/058,657 filed by Dartmouth College that covers the 3D ROSE probe technology reported in this manuscript. H.F., Y.Q. and K.J.S. are inventors on patent US 11417987B2 covering the MagMatrix connector technology reported in this manuscript. The other authors declare no competing interests.
Peer review
Peer review information
Nature Electronics thanks Joonsoo Jeong, Chi Hwan Lee and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–40, Tables 1–6 and Notes 1–8.
Supplementary Video 1 (download MOV )
ROSE probe insertion process and synchronized force dynamic.
Supplementary Video 2 (download MP4 )
3D SU firing map under one visual stimulation trial.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiang, Y., Gu, W., Jang, D. et al. Monolithic three-dimensional neural probes from deterministic rolling of soft electronics. Nat Electron 8, 721–737 (2025). https://doi.org/10.1038/s41928-025-01431-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41928-025-01431-0