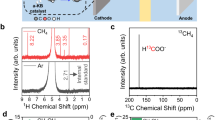
Abstract
The electrochemical conversion of methane into value-added chemicals offers a sustainable solution for utilizing abundant methane resources, yet achieving high selectivity for partial oxidation remains challenging. Here we demonstrate that employing an IrO2 catalyst with CO32− as an oxygen source enables efficient and selective electrochemical methane-to-methanol conversion at room temperature. Adsorption and dissociation of CO32− on IrO2(110) surfaces generates abundant active oxygen species, facilitating methane activation through surface-bound methoxy intermediates and thereby substantially enhancing methanol selectivity. Optimal conditions for methanol production are achieved within a potential range where interference from the competing oxygen evolution reaction is minimized, reaching a maximum methanol production rate of approximately 11.1 mmol gcat−1 h−1 at 1.50 versus the reversible hydrogen electrode under continuous operation. Process modelling indicates an approximately 50% reduction in carbon emissions compared to conventional methanol production methods, emphasizing the sustainability and practical potential of this electrochemical methane oxidation approach.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are included in the Article and its Supplementary Information. Source data are provided with this paper. All other supporting data are available from the corresponding authors upon reasonable request.
References
Li, Z. et al. Direct methane activation by atomically thin platinum nanolayers on two-dimensional metal carbides. Nat. Catal. 4, 882–891 (2021).
Li, X., Wang, C. & Tang, J. Methane transformation by photocatalysis. Nat. Rev. Mater 7, 617–632 (2022).
Liu, J., Yang, Y., Goetjen, T. A. & Hupp, J. T. Carbon-efficient conversion of natural gas and natural-gas condensates to chemical products and intermediate feedstocks via catalytic metal–organic framework (MOF) chemistry. Energy Environ. Sci. 15, 2819–2842 (2022).
Mao, J. et al. Direct conversion of methane with O2 at room temperature over edge-rich MoS2. Nat. Catal. 6, 1052–1061 (2023).
Cruchade, H. et al. Catalytic routes for direct methane conversion to hydrocarbons and hydrogen: current state and opportunities. ACS Catal. 12, 14533–14558 (2022).
Schüth, F. Making more from methane. Science 363, 1282–1283 (2019).
Liu, F., Yan, Y., Chen, G. & Wang, D. Recent advances in ambient electrochemical methane conversion to oxygenates using metal oxide electrocatalysts. Green. Chem. 26, 655–677 (2024).
Meng, X. et al. Direct methane conversion under mild condition by thermo-, electro-, or photocatalysis. Chem 5, 2296–2325 (2019).
Yuan, S. et al. Conversion of methane into liquid fuels—bridging thermal catalysis with electrocatalysis. Adv. Energy Mater. 10, 2002154 (2020).
Min, H. et al. Co-Enriched high entropy oxides for efficient continuous electrochemical methane conversion: catalytic performance and sustainability insights. Adv. Mater. 37, 2418767 (2025).
Bagherzadeh Mostaghimi, A. H., Al-Attas, T. A., Kibria, M. G. & Siahrostami, S. A review on electrocatalytic oxidation of methane to oxygenates. J. Mater. Chem. A 8, 15575–15590 (2020).
Prajapati, A., Sartape, R., Kani, N. C., Gauthier, J. A. & Singh, M. R. Chloride-promoted high-rate ambient electrooxidation of methane to methanol on patterned Cu–Ti bimetallic oxides. ACS Catal. 12, 14321–14329 (2022).
Boghosian Patricio, N. et al. Assembling bifunctional ceria-zirconia electrocatalyst for efficient electrochemical conversion of methane at room temperature. Chem. Eng. J. 488, 150951 (2024).
Jiang, H. et al. Electrocatalytic methane direct conversion to methanol in electrolyte of ionic liquid. Electrochim. Acta 445, 142065 (2023).
Martin, R., Kim, M., Asthagiri, A. & Weaver, J. F. Alkane activation and oxidation on late-transition-metal oxides: challenges and opportunities. ACS Catal. 11, 4682–4703 (2021).
Dummer, N. F. et al. Methane oxidation to methanol. Chem. Rev. 123, 6359–6411 (2023).
Yang, L. et al. Metal–organic framework-derived IrO2/CuO catalyst for selective oxidation of methane to methanol. ACS Energy Lett. 4, 2945–2951 (2019).
Liu, Y.-C. et al. In situ spectroscopic and theoretical investigation of methane activation on IrO2 nanoparticles: Role of Ir oxidation state on C-H activation. J. Catal. 385, 265–273 (2020).
Liang, Z., Li, T., Kim, M., Asthagiri, A. & Weaver Jason, F. Low-temperature activation of methane on the IrO2(110) surface. Science 356, 299–303 (2017).
Latimer, A. A. et al. Understanding trends in C–H bond activation in heterogeneous catalysis. Nat. Mater. 16, 225–229 (2017).
Kim, H. W., Shim, S. H., Myung, J. H. & Lee, C. Annealing effects on the structural properties of IrO2 thin films. Vacuum 82, 1400–1403 (2008).
Chen, T.-S. et al. Stability of reactive DC-sputtered Ir and IrO2 thin films in various ambients. Integr. Ferroelectr. 16, 191–198 (1997).
Martin, R. et al. Isothermal reduction of IrO2(110) films by methane investigated using in situ X-ray photoelectron spectroscopy. ACS Catal. 11, 5004–5016 (2021).
Matz, O. & Calatayud, M. Periodic DFT study of rutile IrO2: surface reactivity and catechol adsorption. J. Phys. Chem. C. 121, 13135–13143 (2017).
Escalera-López, D. et al. Phase- and surface composition-dependent electrochemical stability of Ir-Ru nanoparticles during oxygen evolution reaction. ACS Catal. 11, 9300–9316 (2021).
Mehmood, A., Chae, S. Y. & Park, E. D. Low-temperature electrochemical oxidation of methane into alcohols. Catalysts 14, 58 (2024).
Song, Y. et al. High-pressure electro-Fenton driving CH4 conversion by O2 at room temperature. J. Am. Chem. Soc. 146, 5834–5842 (2024).
Huang, Y. S. et al. Raman spectrum of IrO2. Solid State Commun. 70, 517–522 (1989).
Moriau, L. et al. Ir metal nanoparticles and IrO2 for acidic oxygen evolution reaction: insight from Raman spectroscopy. Sustain. Mater. Technol. 40, e00901 (2024).
Kim, M. et al. Adsorption and oxidation of CH4 on oxygen-rich IrO2(110). J. Phys. Chem. C. 123, 27603–27614 (2019).
Bian, Y., Kim, M., Li, T., Asthagiri, A. & Weaver, J. F. Facile dehydrogenation of ethane on the IrO2(110) Surface. J. Am. Chem. Soc. 140, 2665–2672 (2018).
Kim, C. et al. NiO/ZnO heterojunction nanorod catalyst for high-efficiency electrochemical conversion of methane. Appl. Catal. B 323, 122129 (2023).
Lee, J., Yang, J. & Moon, J. H. Solar cell-powered electrochemical methane-to-methanol conversion with CuO/CeO2 catalysts. ACS Energy Lett. 6, 893–899 (2021).
Xing, Z., Hu, X. & Feng, X. Tuning the microenvironment in gas-diffusion electrodes enables high-rate CO2 electrolysis to formate. ACS Energy Lett. 6, 1694–1702 (2021).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Yang, Y. & Li, F. Reactor design for electrochemical CO2 conversion toward large-scale applications. Curr. Opin. Green. Sustain. Chem. 27, 100419–100425 (2021).
Ma, M., Kim, S., Chorkendorff, I. & Seger, B. Role of ion-selective membranes in the carbon balance for CO2 electroreduction via gas diffusion electrode reactor designs. Chem. Sci. 11, 8854–8861 (2020).
Fang, W. et al. Durable CO2 conversion in the proton-exchange membrane system. Nature 626, 86–91 (2024).
Banat, F. A., Abu Al-Rub, F., Jumah, R. & Shannag, M. On the effect of inert gases in breaking the formic acid–water azeotrope by gas-gap membrane distillation. Chem. Eng. J. 73, 37–42 (1999).
Ewell, R., Harrison, J. & Berg, L. Azeotropic distillation. Ind. Eng. Chem. 36, 871–875 (1944).
Woods, M., Kuehn, N., Shah, V. & Goellner, J. F. Baseline Analysis of Crude Methanol Production From Coal and Natural Gas (National Energy Technology Laboratory, 2014).
Schorn, F. et al. Methanol as a renewable energy carrier: An assessment of production and transportation costs for selected global locations. Adv. Appl. Energy 3, 100050 (2021).
Egli, F., Steffen, B. & Schmidt, T. S. A dynamic analysis of financing conditions for renewable energy technologies. Nat. Energy 3, 1084–1092 (2018).
Colella, W. G., James, B. & Moton, J. M. Hydrogen Pathways Analysis for Polymer Electrolyte Membrane (PEM) Electrolysis (Strategic Anal. Inc, 2014).
Lee, W. H. et al. New strategies for economically feasible CO2 electroreduction using a porous membrane in zero-gap configuration. J. Mater. Chem. A 9, 16169–16177 (2021).
Kumaravel, V., Bartlett, J. & Pillai, S. C. Photoelectrochemical conversion of carbon dioxide (CO2) into fuels and value-added products. ACS Energy Lett. 5, 486–519 (2020).
Belsa, B. et al. Materials challenges on the path to gigatonne CO2 electrolysis. Nat. Rev. Mater. 9, 535–549 (2024).
Rochelle, G. T. Amine scrubbing for CO2 capture. Science 325, 1652–1654 (2009).
Zoelle, A. J. et al. Cost and Performance Baseline for Fossil Energy Plants Vol. 1, Revision 4 (National Energy Technology Laboratory, 2018).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Hammer, B., Hansen, L. B. & Nørskov, J. K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B 59, 7413–7421 (1999).
Moellmann, J. & Grimme, S. DFT-D3 study of some molecular crystals. J. Phys. Chem. C. 118, 7615–7621 (2014).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) under grant numbers 2024-00466477 and 2025-00559443.
Author information
Authors and Affiliations
Contributions
C.K. contributed to the methodology, formal analysis, validation and data curation. J.L. contributed to investigation and data curation. S.L. performed the computational work. W.J. conducted process analysis and drafted the corresponding sections. H.M. performed investigations and contributed to formal analysis and data curation. J.C. contributed to validation and data curation. S.K. contributed to validation. Y.T.K. provided resources. J.L. provided resources and supervised the project. J.S.Y. supervised the computational analysis and edited the corresponding section. J.H.M. acquired funding, contributed to conceptualization, supervised the study, proposed the computational strategy, contributed to formal analysis and was responsible for writing the original draft and reviewing and editing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–22, Tables 1–13, Notes 1–5 and Refs. 1–46.
Supplementary Data 1 (download TXT )
Atomic coordinates of optimized slab models.
Source data
Source Data Fig. 1 (download XLSX )
Raw numerical data.
Source Data Fig. 2 (download XLSX )
Raw numerical data.
Source Data Fig. 3 (download XLSX )
Raw numerical data.
Source Data Fig. 4 (download XLSX )
Raw numerical data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, C., Lee, J., Lee, S. et al. High-selectivity room-temperature partial oxidation of methane to methanol enabled by electrochemical oxygen promotion on IrO2 catalysts. Nat Catal 8, 688–696 (2025). https://doi.org/10.1038/s41929-025-01363-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41929-025-01363-0
This article is cited by
-
Tethered oxygen turns methane into methanol
Nature Catalysis (2025)