Abstract
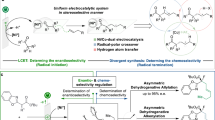
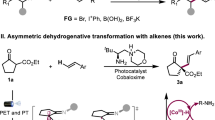
Enzymes are emerging as promising catalysts for selective radical transformations. However, non-natural radical-type enzymatic catalysis is currently limited to utilizing C-, N- and S-centred radical species. Alkoxy radicals are recognized as versatile intermediates with high reactivity, typically engaging in reactivity modes such as hydrogen atom transfer, β-scission processes and intramolecular addition to alkenes. Enantioselective intermolecular alkoxy radical addition to alkenes remained unknown. Here we develop a biocatalytic strategy based on engineered ene-reductases that facilitate the radical hydroalkoxylation of oxygen-centred radicals with alkenes. A single, ground-state ene-reductase adeptly controls the biocompatible generation of O-radicals, the follow-up intermolecular O-radical addition to alkenes and the final prochiral C-radical termination, achieving high chemo- and enantioselectivity (both enantiomers are obtained separately with different enzymes). Mechanistic experiments, including computational simulations, reveal that the radical enzymatic reaction initiates via a ground-state single-electron transfer and elucidate the origins of enantiodiscrimination of the overall reaction.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
HPLC spectra and NMR spectra are available in the Supplementary Information. The PDB code of the GluER used in this work is 6O08. The structural data (including all QM calculation coordinates, QM region coordinates from QM/MM calculations and the initial and final configurations of molecular dynamics trajectories) are available via Zenodo at https://doi.org/10.5281/zenodo.14851048 (ref. 67).
Code availability
The codes for building the QSAR model are available via GitHub at https://github.com/ld139/QSAR_enz.
References
Chang, L., An, Q., Duan, L., Feng, K. & Zuo, Z. Alkoxy radicals see the light: new paradigms of photochemical synthesis. Chem. Rev. 122, 2429–2486 (2022).
Wang, C., Harms, K. & Meggers, E. Catalytic asymmetric C–H functionalization under photoredox conditions by radical translocation and stereocontrolled alkene addition. Angew. Chem. Int. Ed. 55, 13495–13498 (2016).
Zhang, J., Li, Y., Zhang, F., Hu, C. & Chen, Y. Generation of alkoxyl radicals by photoredox catalysis enables selective C(sp3)–H functionalization under mild reaction conditions. Angew. Chem. Int. Ed. 55, 1872–1875 (2016).
Hu, A., Guo, J.-J., Pan, H. & Zuo, Z. Selective functionalization of methane, ethane, and higher alkanes by cerium photocatalysis. Science 361, 668–672 (2018).
Vasilopoulos, A., Krska, S. W. & Stahl, S. S. C(sp3)–H methylation enabled by peroxide photosensitization and Ni-mediated radical coupling. Science 372, 398–403 (2021).
Yayla, H. G., Wang, H., Tarantino, K. T., Orbe, H. S. & Knowles, R. R. Catalytic ring-opening of cyclic alcohols enabled by PCET activation of strong O–H bonds. J. Am. Chem. Soc. 138, 10794–10797 (2016).
Hartung, J., Stowasser, R., Vitt, D. & Bringmann, G. 5-exo or 6-endo? Exploring transition state structures in cyclizations of 4-penten-1-oxyl radicals. Angew. Chem. Int. Ed. Engl. 35, 2820–2823 (1996).
Tsui, E., Metrano, A. J., Tsuchiya, Y. & Knowles, R. R. Catalytic hydroetherification of unactivated alkenes enabled by proton-coupled electron transfer. Angew. Chem. Int. Ed. 59, 11845–11849 (2020).
Barthelemy, A. L., Tuccio, B., Magnier, E. & Dagousset, G. Alkoxyl radicals generated under photoredox catalysis: a strategy for anti-Markovnikov alkoxylation reactions. Angew. Chem. Int. Ed. 57, 13790–13794 (2018).
Yuan, F. et al. Photoredox-catalyzed multicomponent cyclization of 2-vinyl phenols, N-alkoxypyridinium salts, and sulfur ylides for synthesis of dihydrobenzofurans. ChemCatChem 13, 543–547 (2021).
Capaldo, L. & Ravelli, D. Alkoxy radicals generation: facile photocatalytic reduction of N-alkoxyazinium or azolium salts. Chem. Commun. 55, 3029–3032 (2019).
Ali, M., Sewell, S., Li, J. & Wang, T. Recent advances in application of alkoxy radical in organic synthesis. Organics 4, 459–489 (2023).
Li, P. et al. Modulating electron transfer via cerium photocatalysis for alkoxy radical-mediated selective hydroetherification. Angew. Chem. Int. Ed. 64, e202501949 (2025).
Mao, R. et al. Biocatalytic, enantioenriched primary amination of tertiary C–H bonds. Nat. Catal. 7, 585–592 (2024).
Emmanuel, M. A., Greenberg, N. R., Oblinsky, D. G. & Hyster, T. K. Accessing non-natural reactivity by irradiating nicotinamide-dependent enzymes with light. Nature 540, 414–417 (2016).
Peng, Y. et al. Photoinduced promiscuity of cyclohexanone monooxygenase for the enantioselective synthesis of α-fluoroketones. Angew. Chem. Int. Ed. 61, e202211199 (2022).
Zhou, Q., Chin, M., Fu, Y., Liu, P. & Yang, Y. Stereodivergent atom-transfer radical cyclization by engineered cytochromes P450. Science 374, 1612–1616 (2021).
Rui, J. et al. Directed evolution of nonheme iron enzymes to access abiological radical-relay C(sp3)–H azidation. Science 376, 869–874 (2022).
Jain, S., Ospina, F. & Hammer, S. C. A new age of biocatalysis enabled by generic activation modes. JACS Au 4, 2068–2080 (2024).
Toogood, H. S. & Scrutton, N. S. Discovery, characterization, engineering, and applications of ene-reductases for industrial biocatalysis. ACS Catal. 8, 3532–3549 (2018).
Baker Dockrey, S. A. & Narayan, A. R. H. Flavin-dependent biocatalysts in synthesis. Tetrahedron 75, 1115–1121 (2019).
Kumar Roy, T., Sreedharan, R., Ghosh, P., Gandhi, T. & Maiti, D. Ene-reductase: a multifaceted biocatalyst in organic synthesis. Chem. Eur. J. 28, e202103949 (2022).
Stephenson, C. R. J., Yoon, T. P. & MacMillan, D. W. C. Visible Light Photocatalysis in Organic Chemistry (Wiley, 2018).
Mondal, S. et al. Enantioselective radical reactions using chiral catalysts. Chem. Rev. 122, 5842–5976 (2022).
Harrison, W., Huang, X. & Zhao, H. Photobiocatalysis for abiological transformations. Acc. Chem. Res. 55, 1087–1096 (2022).
Fu, H. & Hyster, T. K. From ground-state to excited-state activation modes: flavin-dependent “ene”-reductases catalyzed non-natural radical reactions. Acc. Chem. Res. 57, 1446–1457 (2024).
Biegasiewicz, K. F. et al. Photoexcitation of flavoenzymes enables a stereoselective radical cyclization. Science 364, 1166–1169 (2019).
Liu, Y. et al. Asymmetric synthesis of α-chloroamides via photoenzymatic hydroalkylation of olefins. J. Am. Chem. Soc. 146, 7191–7197 (2024).
Huang, X. et al. Photoenzymatic enantioselective intermolecular radical hydroalkylation. Nature 584, 69–74 (2020).
Li, M., Harrison, W., Zhang, Z., Yuan, Y. & Zhao, H. Remote stereocontrol with azaarenes via enzymatic hydrogen atom transfer. Nat. Chem. 16, 277–284 (2024).
Duan, X. et al. A photoenzymatic strategy for radical-mediated stereoselective hydroalkylation with diazo compounds. Angew. Chem. Int. Ed. 62, e202214135 (2023).
Zhu, C. et al. Photoenzymatic enantioselective synthesis of oxygen-containing benzo-fused heterocycles. Angew. Chem. Int. Ed. 62, e202311762 (2023).
Crisenza, G. E. M., Mazzarella, D. & Melchiorre, P. Synthetic methods driven by the photoactivity of electron donor–acceptor complexes. J. Am. Chem. Soc. 142, 5461–5476 (2020).
Zhao, B. et al. Direct visible-light-excited flavoproteins for redox-neutral asymmetric radical hydroarylation. Nat. Catal. 6, 996–1004 (2023).
Yu, J. et al. Repurposing visible-light-excited ene-reductases for diastereo- and enantioselective lactones synthesis. Angew. Chem. Int. Ed. 63, e202402673 (2024).
Ye, Y. et al. Using enzymes to tame nitrogen-centred radicals for enantioselective hydroamination. Nat. Chem. 15, 206–212 (2023).
Zhang, Z. et al. Photoenzymatic enantioselective intermolecular radical hydroamination. Nat. Catal. 6, 687–694 (2023).
Harrison, W. et al. Photoenzymatic asymmetric hydroamination for chiral alkyl amine synthesis. J. Am. Chem. Soc. 146, 10716–10722 (2024).
Shi, F. et al. Enantioselective biosynthesis of vicinal diamines enabled by synergistic photo/biocatalysis consisting of an ene-reductase and a green-light-excited organic dye. Chin. J. Catal. 68, 223–229 (2025).
Chen, X. et al. Photoenzymatic hydrosulfonylation for the stereoselective synthesis of chiral sulfones. Angew. Chem. Int. Ed. 62, e202218140 (2023).
Jiang, L. et al. Photoenzymatic redox-neutral radical hydrosulfonylation initiated by FMN. ACS Catal. 14, 6710–6716 (2024).
Shi, Q. et al. Single-electron oxidation-initiated enantioselective hydrosulfonylation of olefins enabled by photoenzymatic catalysis. J. Am. Chem. Soc. 146, 2748–2756 (2024).
Xu, Y. et al. A light-driven enzymatic enantioselective radical acylation. Nature 625, 74–78 (2024).
Chen, B. et al. Modular access to chiral amines via imine reductase-based photoenzymatic catalysis. J. Am. Chem. Soc. 146, 14278–14286 (2024).
Zhang, J. et al. Photoenzymatic conversion of enamides to enantioenriched benzylic amines enabled by visible-light-induced single-electron reduction. ACS Catal. 13, 15682–15690 (2023).
Gould, I. R., Shukla, D., Giesen, D. & Farid, S. Energetics of electron-transfer reactions of photoinitiated polymerization: dye-sensitized fragmentation of N-alkoxypyridinium salts. Helv. Chim. Acta 84, 2796–2812 (2001).
Stewart, R. C. & Massey, V. Potentiometric studies of native and flavin-substituted old yellow enzyme. J. Biol. Chem. 260, 13639–13647 (1985).
Sandoval, B. A., Meichan, A. J. & Hyster, T. K. Enantioselective hydrogen atom transfer: discovery of catalytic promiscuity in flavin-dependent ‘ene’-reductases. J. Am. Chem. Soc. 139, 11313–11316 (2017).
Duan, X. et al. Ground-state flavin-dependent enzymes catalyzed enantioselective radical trifluoromethylation. Nat. Commun. 16, 1225 (2025).
Xu, J. et al. Stereodivergent protein engineering of a lipase to access all possible stereoisomers of chiral esters with two stereocenters. J. Am. Chem. Soc. 141, 7934–7945 (2019).
Bao, Y., Xu, Y. & Huang, X. Focused rational iterative site-specific mutagenesis (FRISM): a powerful method for enzyme engineering. Mol. Catal. 553, 113755 (2024).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Trott, O. & Olson, A. J. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31, 455–461 (2010).
Maier, J. A. et al. ff14SB: improving the accuracy of protein side chain and backbone parameters from ff99SB. J. Chem. Theory Comput. 11, 3696–3713 (2015).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Becke, A. D. Density‐functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926–935 (1983).
Case, D. A. et al. AMBER 2018 (Univ. of California, 2018).
Metz, S., Kästner, J., Sokol, A. A., Keal, T. W. & Sherwood, P. ChemShell—a modular software package for QM/MM simulations. WIREs Comput. Mol. Sci. 4, 101–110 (2014).
Lu, Y. et al. Multiscale QM/MM modelling of catalytic systems with ChemShell. Phys. Chem. Chem. Phys. 25, 21816–21835 (2023).
Furche, F. et al. Turbomole. WIREs Comput. Mol. Sci. 4, 91–100 (2014).
Smith, W. & Forester, T. R. DL_POLY_2.0: a general-purpose parallel molecular dynamics simulation package. J. Mol. Graph. 14, 136–141 (1996).
Smith, W., Yong, C. W. & Rodger, P. M. DL_POLY: application to molecular simulation. Mol. Simul. 28, 385–471 (2002).
Bakowies, D. & Thiel, W. Hybrid models for combined quantum mechanical and molecular mechanical approaches. J. Phys. Chem. 100, 10580–10594 (1996).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Zhang, Q. Steering oxygen-centred radicals with ground-state ene-reductases for enantioselective intermolecular hydroalkoxylations. Zenodo https://doi.org/10.5281/zenodo.14851048 (2025).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (grant number 2022YFA0913000 to X.H.), the National Natural Science Foundation of China (grant numbers 22277053 to X.H., 22121001 to B.W. and 224B2705 to B.Z.), the Fundamental Research Funds for the Central Universities (grant numbers 0205/14380346 and 0205/14380351 to X.H.) and the Excellent Research Program of Nanjing University (grant number ZYJH004), the State Key Laboratory of Microbial Technology Open Projects Fund (M2023-01 to B.W.).
Author information
Authors and Affiliations
Contributions
B.C. envisioned and developed the reaction. B.Z. assisted with synthetic experiments. J.Y. and R.G. created mutations and expressed proteins. Q.Z., D.L. and B.W. conducted computational studies. Z.Z. reproduced selected examples. B.C., Q.Z., B.W. and X.H. wrote the paper with input from all authors. X.H. coordinated and conceived the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Maciej Szaleniec and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–36, Tables 1–18 and methods.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, B., Zhang, Q., Yu, J. et al. Steering oxygen-centred radicals with ground-state ene-reductases for enantioselective intermolecular hydroalkoxylations. Nat Catal 8, 740–748 (2025). https://doi.org/10.1038/s41929-025-01372-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41929-025-01372-z